Copper(I) chloride

| |

| |
| Names | |
|---|---|
| IUPAC name
Copper(I) chloride
| |
| Other names
Cuprous chloride
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 8127933 | |
| ChEBI | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.028.948 |
| EC Number |
|
| 13676 | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CuCl | |
| Molar mass | 98.999 g/mol[1] |
| Appearance | white powder, slightly green from oxidized impurities |
| Density | 4.14 g/cm3[1] |
| Melting point | 423 °C (793 °F; 696 K)[1] |
| Boiling point | 1,490 °C (2,710 °F; 1,760 K) (decomposes)[1] |
| 0.047 g/L (20 °C)[1] | |
Solubility product (Ksp)
|
1.72×10−7 |
| Solubility | insoluble in NH4OH
|
| Band gap | 3.25 eV (300 K, direct)[2] |
| -40.0·10−6 cm3/mol[3] | |
Refractive index (nD)
|
1.930[4] |
| Structure | |
Zincblende, cF20
| |
| F43m, No. 216[5] | |
a = 0.54202 nm
| |
Lattice volume (V)
|
0.1592 nm3 |
Formula units (Z)
|
4 |
| Hazards | |
| GHS labelling: | |
 
| |
| Warning | |
| H302, H410 | |
| P264, P270, P273, P301+P312, P330, P391, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
140 mg/kg |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu)[6] |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu)[6] |
IDLH (Immediate danger) |
TWA 100 mg/m3 (as Cu)[6] |
| Safety data sheet (SDS) | JT Baker |
| Related compounds | |
Other anions
|
Copper(I) fluoride Copper(I) bromide Copper(I) iodide |
Other cations
|
Silver(I) chloride
Gold(I) chloride |
Related compounds
|
Copper(II) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear green due to the presence of copper(II) chloride (CuCl2).
History
Copper(I) chloride was first prepared by Robert Boyle in the mid-seventeenth century from mercury(II) chloride ("Venetian sublimate") and copper metal:[7]
- HgCl2 + 2 Cu → 2 CuCl + Hg
In 1799, J.L. Proust characterized the two different chlorides of copper. He prepared CuCl by heating CuCl2 at red heat in the absence of air, causing it to lose half of its combined chlorine followed by removing residual CuCl2 by washing with water.[8]
An acidic solution of CuCl was formerly used to analyze carbon monoxide content in gases, for example in Hempel's gas apparatus where the CuCl absorbs the carbon monoxide.[9] This application was significant during the nineteenth and early twentieth centuries when coal gas was widely used for heating and lighting.[10]
Synthesis
Copper(I) chloride is produced industrially by the direct combination of copper metal and chlorine at 450–900 °C:[11][12]
- 2 Cu + Cl2 → 2 CuCl
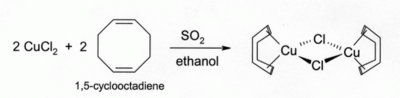
Copper(I) chloride can also be prepared by reducing copper(II) chloride with sulfur dioxide, or with ascorbic acid (vitamin C) that acts as a reducing sugar:[13][14]
- 2 CuCl2 + SO2 + 2 H2O → 2 CuCl + H2SO4 + 2 HCl
- 2 CuCl2 + C6H8O6 → 2CuCl + 2HCl + C6H6O6
Many other reducing agents can be used.[12]
-
White copper(I) chloride crystals on copper wire
-
Copper(I) chloride partially oxidized in air
Properties
Copper(I) chloride has the cubic
Copper(I) chloride is a
- CuCl + 1 P(C6H5)3 → 1/4 {CuCl[P(C6H5)3]}4
- CuCl + 2 P(C6H5)3 → CuCl[P(C6H5)3)]2
- CuCl + 3 P(C6H5)3 → CuCl[P(C6H5)3)]3
CuCl also forms complexes with halides. For example H3O+ CuCl2− forms in concentrated hydrochloric acid.[15] Chloride is displaced by CN− and S2O32−.[12]
Solutions of CuCl in
Upon contact with water, copper(I) chloride slowly undergoes disproportionation:[17]
- 2 CuCl → Cu + CuCl2
In part for this reason, samples in air assume a green coloration.[18]
Uses
The main use of copper(I) chloride is as a precursor to the
- Cu + CuCl2 → 2 CuCl
- 4 CuCl + O2 + 2 H2O → Cu3Cl2(OH)4 + CuCl2
Copper(I) chloride catalyzes a variety of
In organic synthesis
CuCl is used as a co-catalyst with
In the
The reaction has wide scope and usually gives good yields.[22]
Early investigators observed that copper(I) halides catalyse 1,4-addition of
This finding led to the development of
Niche uses
CuCl is used as a catalyst in atom transfer radical polymerization (ATRP). It is also used in pyrotechnics as a blue/green coloring agent. In a flame test, copper chlorides, like all copper compounds, emit green-blue.[26]
Natural occurrence
Natural form of CuCl is the rare mineral nantokite.[27][28]
See also
References
- ^ ISBN 1-4398-5511-0.
- .
- ISBN 1-4398-5511-0.
- ISBN 0-07-049439-8
- ^ PMID 9976955.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH).
- ^ Boyle, Robert (1666). Considerations and experiments about the origin of forms and qualities. Oxford. pp. 286–288.
- ^ Proust, J. L. (1799). "Recherches sur le Cuivre". Ann. Chim. Phys. 32: 26–54.
- ^ Martin, Geoffrey (1922). Industrial and Manufacturing Chemistry (Part 1, Organic ed.). London: Crosby Lockwood. p. 408.
- ^ Lewes, Vivian H. (1891). "The Analysis of Illuminationg Gases". Journal of the Society of Chemical Industry. 10: 407–413.
- ISBN 0471238961.
- ^ ISBN 978-3-527-30673-2.
- ^ Glemser, O.; Sauer, H. (1963). "Copper(I) Chloride". In Brauer, G. (ed.). Handbook of Preparative Inorganic Chemistry. Vol. 1 (2nd ed.). New York: Academic Press. p. 1005.
- S2CID 85408826.
- .
- ^ Nicholls, D. (1973) Complexes and First-Row Transition Elements, Macmillan Press, London.
- ISBN 978-0-08-037941-8.
- ^ Pastor, Antonio C. (1986) U.S. patent 4,582,579 "Method of preparing cupric ion free cuprous chloride" Section 2, lines 4–41.
- PMID 37083229.
- ISSN 0368-1769.
- ISBN 013033832X.
- ^ ISBN 978-0-470-46259-1
- .
- ISBN 9783527600083.
- ISBN 978-0-471-97924-1.
- ISSN 0370-1301.
- ^ "Nantokite".
- ^ "List of Minerals". 21 March 2011.
External links
- National Pollutant Inventory – Copper and compounds fact sheet
- The COPureSM Process for purifying CO utilizing a copper chloride complex