Copper(I) oxide

| |
 | |
Crystal packing
| |
| Names | |
|---|---|
| IUPAC name
Copper(I) oxide
| |
| Other names | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.013.883 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cu2O | |
| Molar mass | 143.09 g/mol |
| Appearance | brownish-red solid |
| Density | 6.0 g/cm3 |
| Melting point | 1,232 °C (2,250 °F; 1,505 K) |
| Boiling point | 1,800 °C (3,270 °F; 2,070 K) |
| Insoluble | |
| Solubility in acid | Soluble |
| Band gap | 2.137 eV |
| −20×10−6 cm3/mol | |
| Structure | |
| cubic | |
| Pn3m, #224 | |
a = 4.2696
| |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
93 J·mol−1·K−1 |
Std enthalpy of (ΔfH⦵298)formation |
−170 kJ·mol−1 |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H318, H332, H410 | |
| P273, P305+P351+P338[1] | |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu)[2] |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu)[2] |
IDLH (Immediate danger) |
TWA 100 mg/m3 (as Cu)[2] |
| Safety data sheet (SDS) | SIRI.org |
| Related compounds | |
Other anions
|
Copper(II) sulfide
Copper(I) selenide |
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Copper(I) oxide or cuprous oxide is the
Preparation
Copper(I) oxide may be produced by several methods.[4] Most straightforwardly, it arises via the oxidation of copper metal:
- 4 Cu + O2 → 2 Cu2O
Additives such as water and acids affect the rate of this process as well as the further oxidation to copper(II) oxides. It is also produced commercially by reduction of copper(II) solutions with sulfur dioxide.
Alternatively, it may be prepared via the reduction of Copper (II) Hydroxide with hydrogen peroxide during "a complex mechanism with the participation of three different reaction pathways is involved." with the final reducing reaction being:[5]
2 CuOOH+ → 2Cu(I) + H2O2 + O2
2Cu 2+ + OH- + H2O2 → 2Cu + + O2 + H2O [6]
Reactions
Aqueous cuprous chloride solutions react with base to give the same material. In all cases, the color is highly sensitive to the procedural details.
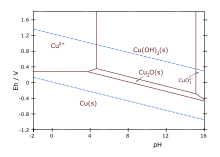
Formation of copper(I) oxide is the basis of the
It forms on silver-plated copper parts exposed to moisture when the silver layer is porous or damaged. This kind of corrosion is known as red plague.
Little evidence exists for copper(I) hydroxide CuOH, which is expected to rapidly undergo dehydration. A similar situation applies to the hydroxides of gold(I) and silver(I).
Properties
The solid is
Copper(I) oxide dissolves in concentrated
2. Dilute sulfuric acid and nitric acid produce copper(II) sulfate and copper(II) nitrate, respectively.[7]
Cu2O degrades to copper(II) oxide in moist air.
Structure
Cu2O crystallizes in a cubic structure with a lattice constant al = 4.2696 Å. The copper atoms arrange in a fcc sublattice, the oxygen atoms in a bcc sublattice. One sublattice is shifted by a quarter of the body diagonal. The space group is Pn3m, which includes the point group with full octahedral symmetry.
Semiconducting properties
In the history of semiconductor physics, Cu2O is one of the most studied materials, and many experimental semiconductor applications have been demonstrated first in this material:
- Semiconductor
- Semiconductor diodes[8]
- Phonoritons ("a coherent superposition of exciton, photon, and phonon")[9][10]
The lowest excitons in Cu2O are extremely long lived; absorption lineshapes have been demonstrated with neV linewidths, which is the narrowest bulk exciton resonance ever observed.[11] The associated quadrupole polaritons have low group velocity approaching the speed of sound. Thus, light moves almost as slowly as sound in this medium, which results in high polariton densities. Another unusual feature of the
Applications
Cuprous oxide is commonly used as a pigment, a fungicide, and an antifouling agent for marine paints. Rectifier diodes based on this material have been used industrially as early as 1924, long before silicon became the standard. Copper(I) oxide is also responsible for the pink color in a positive Benedict's test.
In December 2021,
Similar compounds
An example of natural copper(I,II) oxide is the mineral paramelaconite, Cu4O3 or CuI
2CuII
2O3.[15][16]
See also
- Copper(II) oxide
- Copper(III) oxide
References
- ^ https://www.nwmissouri.edu/naturalsciences/sds/c/Copper%20I%20oxide.pdf [dead link]
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH).
- ^ N. N. Greenwood, A. Earnshaw, Chemistry of the Elements, 2nd ed., Butterworth-Heinemann, Oxford, UK, 1997.
- PMID 14987843.
- ^ Yener, Ersin (September 2017). "Improvement of Stability of Hydrogen Peroxide using Ethylene Glycol".
- ^ D. Nicholls, Complexes and First-Row Transition Elements, Macmillan Press, London, 1973.
- ^ L. O. Grondahl, Unidirectional current carrying device, Patent, 1927
- .
- ISBN 9781483276014.
- PMID 18233254.
- ^ J. P. Wolfe and A. Mysyrowicz: Excitonic Matter, Scientific American 250 (1984), No. 3, 98.
- ISSN 0031-899X.
- ^ Bellini, Emiliano (2021-12-22). "Toshiba claims 8.4% efficiency for transparent cuprous oxide solar cell". pv magazine. Retrieved 2021-12-22.
- ^ "Paramelaconite".
- ^ "List of Minerals". 21 March 2011.

