Copper(II) chloride
 | |
 Anhydrous
| |
 Dihydrate
| |
| Names | |
|---|---|
| IUPAC name
Copper(II) chloride
| |
| Other names
Cupric chloride
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| 8128168 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.028.373 |
| EC Number |
|
| 9300 | |
PubChem CID
|
|
RTECS number
|
|
| UNII |
|
| UN number | 2802 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CuCl2 | |
| Molar mass | 134.45 g/mol (anhydrous) 170.48 g/mol (dihydrate) |
| Appearance | yellow-brown solid (anhydrous) blue-green solid (dihydrate) |
| Odor | odorless |
| Density | 3.386 g/cm3 (anhydrous) 2.51 g/cm3 (dihydrate) |
| Melting point | 630 °C (1,166 °F; 903 K) (extrapolated) 100 °C (dehydration of dihydrate) |
| Boiling point | 993 °C (1,819 °F; 1,266 K) (anhydrous, decomposes) |
| 70.6 g/(100 mL) (0 °C) 75.7 g/(100 mL) (25 °C) 107.9 g/(100 mL) (100 °C) | |
| Solubility | methanol: 68 g/(100 mL) (15 °C)
|
| +1080·10−6 cm3/mol | |
| Structure[1][2] | |
orthorhombic (dihydrate)
| |
| C2/m (anhydrous) Pbmn (dihydrate) | |
a = 6.85 Å (anhydrous)
7.41 Å (dihydrate), b = 3.30 Å (anhydrous) 8.09 Å (dihydrate), c = 6.70 Å (anhydrous) 3.75 Å (dihydrate) | |
| Octahedral | |
| Hazards | |
| GHS labelling: | |
   
| |
| Danger | |
| H301, H302, H312, H315, H318, H319, H335, H410, H411 | |
| P261, P264, P270, P271, P273, P280, P301+P310, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 1 mg/m3 (as Cu)[3] |
REL (Recommended)
|
TWA 1 mg/m3 (as Cu)[3] |
IDLH (Immediate danger) |
TWA 100 mg/m3 (as Cu)[3] |
| Safety data sheet (SDS) | Fisher Scientific |
| Related compounds | |
Other anions
|
Copper(II) fluoride Copper(II) bromide |
Other cations
|
Copper(I) chloride Silver chloride Gold(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Copper(II) chloride, also known as cupric chloride, is an
Both the anhydrous and the dihydrate forms occur naturally as the rare minerals tolbachite and eriochalcite, respectively.
Structure

Anhydrous copper(II) chloride adopts a distorted
Copper(II) chloride is paramagnetic. Of historical interest, CuCl2·2H2O was used in the first electron paramagnetic resonance measurements by Yevgeny Zavoisky in 1944.[6][7]
Properties and reactions
Aqueous solutions prepared from copper(II) chloride contain a range of copper(II) complexes depending on concentration, temperature, and the presence of additional chloride ions. These species include the blue color of [Cu(H2O)6]2+ and the yellow or red color of the halide complexes of the formula [CuCl2+x]x−.[5]
Hydrolysis
When copper(II) chloride solutions are treated with a base, a precipitation of copper(II) hydroxide occurs:[8]
- CuCl2 + 2 NaOH → Cu(OH)2 + 2 NaCl
Partial hydrolysis gives dicopper chloride trihydroxide, Cu2(OH)3Cl, a popular fungicide.[8] When an aqueous solution of copper(II) chloride is left in the air and isn't stabilized by a small amount of acid, it is prone to undergo slight hydrolysis.[5]
Redox and decomposition
Copper(II) chloride is a mild oxidant. It starts to decompose to copper(I) chloride and chlorine gas around 400 °C (752 °F) and is completely decomposed near 1,000 °C (1,830 °F):[8][9][10][11]
- 2 CuCl2 → 2 CuCl + Cl2
The reported melting point of copper(II) chloride of 498 °C (928 °F) is a melt of a mixture of copper(I) chloride and copper(II) chloride. The true melting point of 630 °C (1,166 °F) can be extrapolated by using the melting points of the mixtures of CuCl and CuCl2.[12][13] Copper(II) chloride reacts with several metals to produce copper metal or copper(I) chloride (CuCl) with oxidation of the other metal. To convert copper(II) chloride to copper(I) chloride, it can be convenient to reduce an aqueous solution with sulfur dioxide as the reductant:[8]
- 2 CuCl2 + SO2 + 2 H2O → 2 CuCl + 2 HCl + H2SO4
Coordination complexes
CuCl2 reacts with HCl or other chloride sources to form complex ions: the red [CuCl3]− (found in potassium trichloridocuprate(II) K[CuCl3]) (it is a dimer in reality, [Cu2Cl6]2−, a couple of tetrahedrons that share an edge), and the green or yellow [CuCl4]2− (found in potassium tetrachloridocuprate(II) K2[CuCl4]).[5][14][15]
- CuCl2 + Cl− ⇌ [CuCl3]−
- CuCl2 + 2 Cl− ⇌ [CuCl4]2−
Some of these complexes can be crystallized from aqueous solution, and they adopt a wide variety of structures.[14]
Copper(II) chloride also forms a variety of
- CuCl2 + 2 C5H5N → [CuCl2(C5H5N)2] (tetragonal)
- CuCl2 + 2 (C6H5)3P=O → [CuCl2((C6H5)3P=O)2] (tetrahedral)
However "soft" ligands such as phosphines (e.g., triphenylphosphine), iodide, and cyanide as well as some tertiary amines induce reduction to give copper(I) complexes.[5]
Preparation
Copper(II) chloride is prepared commercially by the action of
- Cu(s) + Cl2(g) → CuCl2(l)
A solution of copper(II) chloride is commercially produced by adding chlorine gas to a circulating mixture of hydrochloric acid and copper. From this solution, the dihydrate can be produced by evaporation.[8][10]
Although copper metal itself cannot be oxidized by hydrochloric acid, copper-containing bases such as the hydroxide, oxide, or copper(II) carbonate can react to form CuCl2 in an acid-base reaction which can subsequently be heated above 100 °C (212 °F) to produce the anhydrous derivative.[8][10]
Once prepared, a solution of CuCl2 may be purified by crystallization. A standard method takes the solution mixed in hot dilute hydrochloric acid, and causes the crystals to form by cooling in a calcium chloride (CaCl2) ice bath.[17][18]
There are indirect and rarely used means of using copper ions in solution to form copper(II) chloride. Electrolysis of aqueous sodium chloride with copper electrodes produces (among other things) a blue-green foam that can be collected and converted to the hydrate. While this is not usually done due to the emission of toxic chlorine gas, and the prevalence of the more general chloralkali process, the electrolysis will convert the copper metal to copper ions in solution forming the compound. Indeed, any solution of copper ions can be mixed with hydrochloric acid and made into a copper chloride by removing any other ions.[19]
Uses
Co-catalyst in Wacker process
A major industrial application for copper(II) chloride is as a co-catalyst with
- C2H4 + PdCl2 + H2O → CH3CHO + Pd + 2 HCl
- Pd + 2 CuCl2 → 2 CuCl + PdCl2
- 4 CuCl + 4 HCl + O2 → 4 CuCl2 + 2 H2O
The overall process is:[20]
- 2 C2H4 + O2 → 2 CH3CHO
In organic synthesis
Copper(II) chloride has some highly specialized applications in the
This reaction is performed in a polar solvent such as dimethylformamide, often in the presence of lithium chloride, which accelerates the reaction.[20]
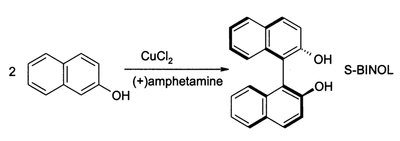
CuCl2, in the presence of
Such compounds are intermediates in the synthesis of BINAP and its derivatives.[20]
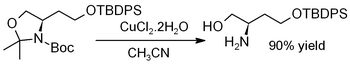
Copper(II) chloride dihydrate promotes the hydrolysis of
CuCl2 also catalyses the
Catalyst in production of chlorine
Copper(II) chloride is used as a
- 4 HCl + O2 → 2 Cl2 + 2 H2O
Copper(II) chloride catalyzes the chlorination in the production of vinyl chloride and dichloromethane.[8]
Copper(II) chloride is used in the copper–chlorine cycle where it reacts with steam into copper(II) oxide dichloride and hydrogen chloride and is later recovered in the cycle from the electrolysis of copper(I) chloride.[11]
Niche uses
Copper(II) chloride is used in pyrotechnics as a blue/green coloring agent. In a flame test, copper chlorides, like all copper compounds, emit green-blue light.[25]
In
Copper(II) chloride is used as a mordant in the textile industry, petroleum sweetener, wood preservative, and water cleaner.[8][28]
Natural occurrence

Copper(II) chloride occurs naturally as the very rare anhydrous mineral tolbachite and the dihydrate eriochalcite.[29] Both are found near fumaroles and in some copper mines.[30][31][32] Mixed oxyhydroxide-chlorides like atacamite (Cu2(OH)3Cl) are more common, arising among Cu ore beds oxidation zones in arid climates.[33]
Safety and biological impact
Copper(II) chloride can be toxic. Only concentrations below 1.3
Copper(II) chloride has been demonstrated to cause chromosomal aberrations and mitotic cycle disturbances within A. cepa (onion) cells.[37] Such cellular disturbances lead to genotoxicity. Copper(II) chloride has also been studied as a harmful environmental pollutant. Often present in irrigation-grade water, it can negatively affect water and soil microbes.[38] Specifically, denitrifying bacteria were found to be very sensitive to the presence of copper(II) chloride. At a concentration of 0.95 mg/L, copper(II) chloride was found to cause a 50% inhibition (IC50) of the metabolic activity of denitrifying microbes.[39]
See also
References
- .
- .
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0150". National Institute for Occupational Safety and Health (NIOSH).
- ISBN 0-19-855370-6.
- ^ ISBN 0-7506-3365-4.
- ISBN 978-3-540-74854-0.
- ISBN 978-0-470-25882-8.
- ^ ISBN 978-3-527-30673-2.
- S2CID 99924382.
- ^ ISBN 0471238961.
- ^ S2CID 93668361.
- .
- ISBN 9780080188607.
- ^ ISBN 978-0-470-13240-1.
- ^ ISBN 9781482277463.
- .
- ^ a b S. H. Bertz, E. H. Fairchild, in Handbook of Reagents for Organic Synthesis, Volume 1: Reagents, Auxiliaries and Catalysts for C-C Bond Formation, (R. M. Coates, S. E. Denmark, eds.), pp. 220–223, Wiley, New York, 1738.
- ISBN 978-1-85617-567-8.
- S2CID 95677720.
- ^ ISBN 9780470842898.
- .
- .
- PMID 12737588.
- .
- ^ Clark, Jim (August 2018). "Flame Tests". chemguide.co.uk. Archived from the original on November 27, 2020. Retrieved January 10, 2021.
- ^ US 20150300958 A1, Evan Koon Lun Yuuji Hajime, "Adjustable colorimetric moisture indicators", published 2015
- ^ "Cobalt dichloride". European Chemicals Agency. ECHA. Retrieved 30 May 2023.
- ISBN 9781845696955. Retrieved 2 June 2023.
- ^ Marlene C. Morris, Howard F. McMurdie, Eloise H. Evans, Boris Paretzkin, Harry S. Parker, and Nicolas C. Panagiotopoulos (1981) Copper chloride hydrate (eriochalcite), in Standard X-ray Diffraction Powder Patterns National Bureau of Standards, Monograph 25, Section 18; page 33.
- ^ "Tolbachite". mindat.org. Retrieved 24 August 2023.
- ^ "Eriochalcite". mindat.org. Retrieved 24 August 2023.
- ^ "The New IMA List of Minerals". Università degli studi di Trieste. International Mineralogical Association. Retrieved 24 August 2023.
- ^ "Atacamite". mindat.org. Retrieved 30 May 2023.
- ^ "National Primary Drinking Water Regulations". EPA. 30 November 2015. Retrieved 29 May 2023.
- ^ "Copper: Health Information Summary" (PDF). Environmental Fact Sheet. New Hampshire Department of Environmental Services. 2005. ARD-EHP-9. Archived from the original (PDF) on 20 January 2017.
- ^ "Safety Data Sheet". Sigma Aldrich. Retrieved 30 June 2023.
- S2CID 208649491.
- .
- PMID 22030247.
Further reading
- ISBN 978-0-08-037941-8.
- Lide, David R. (1990). CRC handbook of chemistry and physics: a ready-reference book of chemical and physical data. Boca Raton: CRC Press. ISBN 0-8493-0471-7.
- The Merck Index, 7th edition, Merck & Co, Rahway, New Jersey, USA, 1960.
- D. Nicholls, Complexes and First-Row Transition Elements, Macmillan Press, London, 1973.
- A. F. Wells, 'Structural Inorganic Chemistry, 5th ed., Oxford University Press, Oxford, UK, 1984.
- J. March, Advanced Organic Chemistry, 4th ed., p. 723, Wiley, New York, 1992.
- Fieser & Fieser Reagents for Organic Synthesis Volume 5, p158, Wiley, New York, 1975.
- D. W. Smith (1976). "Chlorocuprates(II)". Coordination Chemistry Reviews. 21 (2–3): 93–158. .
External links
- Copper Chloride at The Periodic Table of Videos(University of Nottingham)
- Copper (II) Chloride – Description and Pictures
- National Pollutant Inventory – Copper and compounds fact sheet