Coproporphyrinogen III oxidase
| CPOX | |||
|---|---|---|---|
Gene ontology | |||
| Molecular function | |||
| Cellular component | |||
| Biological process |
| ||
| Sources:Amigo / QuickGO | |||
Ensembl |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | n/a | Chr 16: 58.49 – 58.54 Mb | |||||||
| PubMed search | [2] | [3] | |||||||
| View/Edit Human | View/Edit Mouse |
| Coprogen oxidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
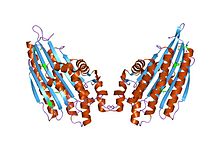 coproporphyrinogen iii oxidase from leishmania major | |||||||||
| Identifiers | |||||||||
| Symbol | Coprogen oxidase | ||||||||
| Pfam | PF01218 | ||||||||
| InterPro | IPR001260 | ||||||||
| PROSITE | PDOC00783 | ||||||||
| |||||||||
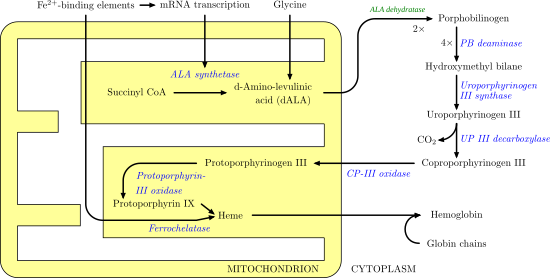
Coproporphyrinogen-III oxidase, mitochondrial (abbreviated as CPOX) is an enzyme that in humans is encoded by the CPOX gene.[4][5][6] A genetic defect in the enzyme results in a reduced production of heme in animals. The medical condition associated with this enzyme defect is called hereditary coproporphyria.[7][8]
CPOX, the sixth enzyme of the
Function
CPOX is an enzyme involved in the sixth step of
Structure
Gene
Human CPOX is a mitochondrial enzyme encoded by a 14 kb CPOX gene containing seven
Protein
CPOX is expressed as a 40 kDa
Clinical significance
To date, over 50 CPOX mutations causing HCP have been described.[17] Most of these mutations result in substitution of amino acid residues within the structural framework of CPOX.[18] At least 32 of these mutations are considered to be disease-causing mutations.[19] In terms of the molecular basis of HCP and harderoporphyria, mutations of CPOX in patients with harderoporphyria were demonstrated in the region of exon 6, where mutations in those with HCP were also identified.[20] As only patients with mutation in this region (K404E) would develop harderoporphyria, this mutation led to diminishment of the second step of the decarboxylation reaction during the conversion of coproporphyrinogen to protoporphyrinogen, implying that the active site of the enzyme involved in the second step of decarboxylation is located in exon 6.[17]
Interactions
CPOX has been shown to
References
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000022742 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 7757079.
- ^ PMID 8407975.
- ^ a b "Entrez Gene: CPOX coproporphyrinogen oxidase".
- ^ "Hereditary coproporphyria". Genetic and Rare Diseases Information Center. National Institutes of Health. Archived from the original on 7 August 2012. Retrieved 8 August 2011.
- ^ "CPOX". Genetics Home Reference. Retrieved 8 August 2011.
- PMID 13746277.
- PMID 3233772.
- S2CID 23011457.
- PMID 3516695.
- PMID 8159699.
- PMID 8012360.
- PMID 8286403.
- PMID 24078084.
- ^ PMID 21103937.
- PMID 16176984.
- PMID 31819097.
- PMID 16159891.
- PMID 16214298.
Further reading
- Fujita H, Kondo M, Taketani S, Nomura N, Furuyama K, Akagi R, Nagai T, Terajima M, Galbraith RA, Sassa S (October 1994). "Characterization and expression of cDNA encoding coproporphyrinogen oxidase from a patient with hereditary coproporphyria". Human Molecular Genetics. 3 (10): 1807–10. PMID 7849704.
- Cacheux V, Martasek P, Fougerousse F, Delfau MH, Druart L, Tachdjian G, Grandchamp B (November 1994). "Localization of the human coproporphyrinogen oxidase gene to chromosome band 3q12". Human Genetics. 94 (5): 557–9. S2CID 11997203.
- Delfau-Larue MH, Martasek P, Grandchamp B (August 1994). "Coproporphyrinogen oxidase: gene organization and description of a mutation leading to exon 6 skipping". Human Molecular Genetics. 3 (8): 1325–30. PMID 7987309.
- Martasek P, Nordmann Y, Grandchamp B (March 1994). "Homozygous hereditary coproporphyria caused by an arginine to tryptophane substitution in coproporphyrinogen oxidase and common intragenic polymorphisms". Human Molecular Genetics. 3 (3): 477–80. PMID 8012360.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. PMID 8125298.
- Martasek P, Camadro JM, Delfau-Larue MH, Dumas JB, Montagne JJ, de Verneuil H, Labbe P, Grandchamp B (April 1994). "Molecular cloning, sequencing, and functional expression of a cDNA encoding human coproporphyrinogen oxidase". Proceedings of the National Academy of Sciences of the United States of America. 91 (8): 3024–8. PMID 8159699.
- Lamoril J, Deybach JC, Puy H, Grandchamp B, Nordmann Y (1997). "Three novel mutations in the coproporphyrinogen oxidase gene". Human Mutation. 9 (1): 78–80. S2CID 45889945.
- Daimon M, Gojyou E, Sugawara M, Yamatani K, Tominaga M, Sasaki H (February 1997). "A novel missense mutation in exon 4 of the human coproporphyrinogen oxidase gene in two patients with hereditary coproporphyria". Human Genetics. 99 (2): 199–201. S2CID 1813242.
- Schreiber WE, Zhang X, Senz J, Jamani A (1997). "Hereditary coproporphyria: exon screening by heteroduplex analysis detects three novel mutations in the coproporphyrinogen oxidase gene". Human Mutation. 10 (3): 196–200. S2CID 32065580.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. PMID 9373149.
- Lamoril J, Puy H, Gouya L, Rosipal R, Da Silva V, Grandchamp B, Foint T, Bader-Meunier B, Dommergues JP, Deybach JC, Nordmann Y (February 1998). "Neonatal hemolytic anemia due to inherited harderoporphyria: clinical characteristics and molecular basis". Blood. 91 (4): 1453–7. PMID 9454777.
- Susa S, Daimon M, Kondo H, Kondo M, Yamatani K, Sasaki H (November 1998). "Identification of a novel mutation of the CPO gene in a Japanese hereditary coproporphyria family". American Journal of Medical Genetics. 80 (3): 204–6. PMID 9843038.
- Rosipal R, Lamoril J, Puy H, Da Silva V, Gouya L, De Rooij FW, Te Velde K, Nordmann Y, Martàsek P, Deybach JC (1999). "Systematic analysis of coproporphyrinogen oxidase gene defects in hereditary coproporphyria and mutation update". Human Mutation. 13 (1): 44–53. S2CID 2705450.
- Taketani S, Furukawa T, Furuyama K (March 2001). "Expression of coproporphyrinogen oxidase and synthesis of hemoglobin in human erythroleukemia K562 cells". European Journal of Biochemistry. 268 (6): 1705–11. PMID 11248690.
- Lamoril J, Puy H, Whatley SD, Martin C, Woolf JR, Da Silva V, Deybach JC, Elder GH (May 2001). "Characterization of mutations in the CPO gene in British patients demonstrates absence of genotype-phenotype correlation and identifies relationship between hereditary coproporphyria and harderoporphyria". American Journal of Human Genetics. 68 (5): 1130–8. PMID 11309681.
- Elkon H, Don J, Melamed E, Ziv I, Shirvan A, Offen D (June 2002). "Mutant and wild-type alpha-synuclein interact with mitochondrial cytochrome C oxidase". Journal of Molecular Neuroscience. 18 (3): 229–38. S2CID 42265181.
- Wiman A, Floderus Y, Harper P (2002). "Two novel mutations and coexistence of the 991C>T and the 1339C>T mutation on a single allele in the coproporphyrinogen oxidase gene in Swedish patients with hereditary coproporphyria". Journal of Human Genetics. 47 (8): 407–12. PMID 12181641.
External links
- Coproporphyrinogen+III+Oxidases at the U.S. National Library of Medicine Medical Subject Headings (MeSH)

