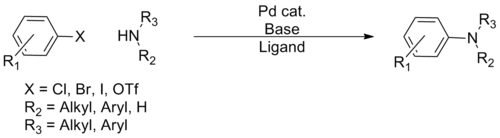Cross-coupling reaction
In
- R−M + R'−X → R−R' + MX (R, R' = organic fragments, usually aryl; M = main group center such as Li or MgX; X = halide)
These reactions are used to form carbon–carbon bonds but also carbon-heteroatom bonds.[1][2][3][4] Cross-coupling reaction are a subset of coupling reactions.
Mechanism
Many mechanisms exist reflecting the myriad types of cross-couplings, including those that do not require metal catalysts.[7] Often, however, cross-coupling refers to a metal-catalyzed reaction of a nucleophilic partner with an electrophilic partner.
In such cases, the
Catalysts

Catalysts are often based on palladium, which is frequently selected due to high
Copper-based catalysts are also common, especially for coupling involving heteroatom-C bonds.[10][11]
Iron-,[12] cobalt-,[13] and nickel-based[14] catalysts have been investigated.
Leaving groups
The
Carbon–carbon cross-coupling
Many cross-couplings entail forming carbon–carbon bonds.
| Reaction | Year | Reactant A | Reactant B | Catalyst | Remark | ||
|---|---|---|---|---|---|---|---|
| Cadiot–Chodkiewicz coupling | 1957 | RC≡CH | sp | RC≡CX | sp | Cu | requires base |
| Castro–Stephens coupling | 1963 | RC≡CH | sp | Ar-X | sp2 | Cu | |
| Corey–House synthesis | 1967 | R2CuLi or RMgX | sp3 | R-X | sp2, sp3 | Cu | Cu-catalyzed version by Kochi, 1971 |
| Kumada coupling | 1972 | RMgBr | sp2, sp3 | R-X | sp2 | Pd or Ni or Fe | |
| Heck reaction | 1972 | alkene | sp2 | Ar-X | sp2 | Pd or Ni | requires base |
| Sonogashira coupling | 1975 | ArC≡CH | sp | R-X | sp3 sp2 | Pd and Cu | requires base |
| Negishi coupling | 1977 | R-Zn-X | sp3, sp2, sp | R-X | sp3 sp2 | Pd or Ni | |
Stille cross coupling |
1978 | R-SnR3 | sp3, sp2, sp | R-X | sp3 sp2 | Pd or Ni | |
| Suzuki reaction | 1979 | R-B(OR)2 | sp2 | R-X | sp3 sp2 | Pd or Ni | requires base |
| Murahashi coupling[15] | 1979 | R-Li | sp2, sp3 | R-X | sp2 | Pd or Ru | |
| Hiyama coupling | 1988 | R-SiR3 | sp2 | R-X | sp3 sp2 | Pd | requires base |
| Fukuyama coupling | 1998 | R-Zn-I | sp3 | RCO(SEt) | sp2 | Pd or Ni | see Liebeskind–Srogl coupling, gives ketones |
| Liebeskind–Srogl coupling | 2000 | R-B(OR)2 | sp3, sp2 | RCO(SEt) Ar-SMe | sp2 | Pd | requires CuTC , gives ketones
|
| Cross dehydrogenative coupling | 2004 | R-H | sp, sp2, sp3 | R'-H | sp, sp2, sp3 | Cu, Fe, Pd etc. | requires oxidant or dehydrogenation |
| Decarboxylative cross-coupling | 2000s | R-CO2H | sp2 | R'-X | sp, sp2 | Cu, Pd | Requires little-to-no base |
The restrictions on carbon atom geometry mainly inhibit
Carbon–heteroatom coupling
Many cross-couplings entail forming carbon–heteroatom bonds (heteroatom = S, N, O). A popular method is the
-
(Eq.1)
| Reaction | Year | Reactant A | Reactant B | Catalyst | Remark | ||
|---|---|---|---|---|---|---|---|
| Ullmann-type reaction | 1905 | ArO-MM, ArNH2,RS-M,NC-M | sp3 | Ar-X (X = OAr, N(H)Ar, SR, CN) | sp2 | Cu | |
Buchwald–Hartwig reaction[17] |
1994 | R2N-H | sp3 | R-X | sp2 | Pd | N-C coupling, second generation free amine |
| Chan–Lam coupling[18] | 1998 | Ar-B(OR)2 | sp2 | Ar-NH2 | sp2 | Cu | |
Miscellaneous reactions
Palladium-catalyzes the cross-coupling of

Applications
Cross-coupling reactions are important for the production of pharmaceuticals,[4] examples being montelukast, eletriptan, naproxen, varenicline, and resveratrol.[20] with Suzuki coupling being most widely used.[21] Some polymers and monomers are also prepared in this way.[22]
Reviews
- Fortman, George C.; Nolan, Steven P. (2011). "N-Heterocyclic carbene (NHC) ligands and palladium in homogeneous cross-coupling catalysis: a perfect union". Chemical Society Reviews. 40 (10): 5151–69. PMID 21731956.
- Yin; Liebscher, Jürgen (2007). "Carbon−Carbon Coupling Reactions Catalyzed by Heterogeneous Palladium Catalysts". Chemical Reviews. 107 (1): 133–173. S2CID 36974481.
- Jana, Ranjan; Pathak, Tejas P.; Sigman, Matthew S. (2011). "Advances in Transition Metal (Pd,Ni,Fe)-Catalyzed Cross-Coupling Reactions Using Alkyl-organometallics as Reaction Partners". Chemical Reviews. 111 (3): 1417–1492. PMID 21319862.
- Molnár, Árpád (2011). "Efficient, Selective, and Recyclable Palladium Catalysts in Carbon−Carbon Coupling Reactions". Chemical Reviews. 111 (3): 2251–2320. PMID 21391571.
- .
- Roglans, Anna; Pla-Quintana, Anna; Moreno-Mañas, Marcial (2006). "Diazonium Salts as Substrates in Palladium-Catalyzed Cross-Coupling Reactions". Chemical Reviews. 106 (11): 4622–4643. S2CID 8128630.
- Korch, Katerina M.; Watson, Donald A. (2019). "Cross-Coupling of Heteroatomic Electrophiles". Chemical Reviews. 119 (13): 8192–8228. PMID 31184483.
- Cahiez, Gérard; Moyeux, Alban (2010). "Cobalt-Catalyzed Cross-Coupling Reactions". Chemical Reviews. 110 (3): 1435–1462. PMID 20148539.
- Yi, Hong; Zhang, Guoting; Wang, Huamin; Huang, Zhiyuan; Wang, Jue; Singh, Atul K.; Lei, Aiwen (2017). "Recent Advances in Radical C–H Activation/Radical Cross-Coupling". Chemical Reviews. 117 (13): 9016–9085. PMID 28639787.
References
- PMID 31184483.
- PMID 16836296.
- ISBN 978-1-84973-896-5
- ^ ISBN 978-3-540-01603-8.
- ^ "The Nobel Prize in Chemistry 2010 - Richard F. Heck, Ei-ichi Negishi, Akira Suzuki". NobelPrize.org. 2010-10-06. Retrieved 2010-10-06.
- S2CID 20582425.
- PMID 25184859.
- ^ Thayer, Ann (2005-09-05). "Removing Impurities". Chemical & Engineering News. Retrieved 2015-12-11.
- S2CID 36974481.
- PMID 16836296.
- PMID 18698737.
- PMID 25916260.
- PMID 20148539.
- PMID 21133429.
- ISSN 0022-3263.
- ^ Clayden, J.; Greeves, N.; Warren, S. Organic Chemistry, 2nd ed.; Oxford UP: Oxford, U.K., 2012. pp. 1069-1102.
- PMID 27689804.
- ISBN 9783527639328.
- PMID 16819868.
- ISBN 9783527328970.
- PMID 21504168.
- ISBN 1-891389-53-X