Cyclic adenosine monophosphate
| |
| |
| Names | |
|---|---|
| IUPAC name
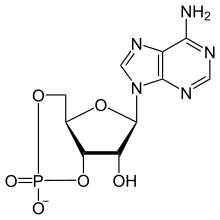
Adenosine 3′,5′-(hydrogen phosphate)
| |
| Systematic IUPAC name
(4aR,6R,7R,7aS)-6-(6-Amino-9H-purin-9-yl)-2,7-dihydroxytetrahydro-2H,4H-2λ5-furo[3,2-d][1,3,2]dioxaphosphinin-2-one | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.000.448 |
IUPHAR/BPS |
|
| KEGG | |
| MeSH | Cyclic+AMP |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H11N5O6P | |
| Molar mass | 329.206 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |


Cyclic adenosine monophosphate (cAMP, cyclic AMP, or 3',5'-cyclic
History
Synthesis
Cyclic
cAMP decomposition into AMP is catalyzed by the enzyme phosphodiesterase.
Functions
cAMP is a
Role in eukaryotic cells
cAMP is associated with kinases function in several biochemical processes, including the regulation of glycogen, sugar, and lipid metabolism.[2]
In eukaryotes, cyclic AMP works by activating protein kinase A (PKA, or
Cyclic AMP binds to specific locations on the regulatory units of the protein kinase, and causes dissociation between the regulatory and catalytic subunits, thus enabling those catalytic units to
The active subunits catalyze the transfer of phosphate from ATP to specific serine or threonine residues of protein substrates. The phosphorylated proteins may act directly on the cell's ion channels, or may become activated or inhibited enzymes. Protein kinase A can also phosphorylate specific proteins that bind to promoter regions of DNA, causing increases in transcription. Not all protein kinases respond to cAMP. Several classes of protein kinases, including protein kinase C, are not cAMP-dependent.
Further effects mainly depend on
Still, there are some minor PKA-independent functions of cAMP, e.g., activation of
However, the view that the majority of the effects of cAMP are controlled by PKA is an outdated one. In 1998 a family of cAMP-sensitive proteins with guanine nucleotide exchange factor (GEF) activity was discovered. These are termed Exchange proteins activated by cAMP (Epac) and the family comprises Epac1 and Epac2.[3] The mechanism of activation is similar to that of PKA: the GEF domain is usually masked by the N-terminal region containing the cAMP binding domain. When cAMP binds, the domain dissociates and exposes the now-active GEF domain, allowing Epac to activate small Ras-like GTPase proteins, such as Rap1.
Additional role of secreted cAMP in social amoebae
In the species Dictyostelium discoideum, cAMP acts outside the cell as a secreted signal. The chemotactic aggregation of cells is organized by periodic waves of cAMP that propagate between cells over distances as large as several centimetres. The waves are the result of a regulated production and secretion of extracellular cAMP and a spontaneous biological oscillator that initiates the waves at centers of territories.[4]
Role in bacteria
In
cAMP, for example, is involved in the positive regulation of the lac operon. In an environment with a low glucose concentration, cAMP accumulates and binds to the allosteric site on CRP (cAMP receptor protein), a transcription activator protein. The protein assumes its active shape and binds to a specific site upstream of the lac promoter, making it easier for RNA polymerase to bind to the adjacent promoter to start transcription of the lac operon, increasing the rate of lac operon transcription. With a high glucose concentration, the cAMP concentration decreases, and the CRP disengages from the lac operon.
Pathology
Since cyclic AMP is a second messenger and plays vital role in cell signalling, it has been implicated in various disorders but not restricted to the roles given below:
Role in human carcinoma
Some research has suggested that a deregulation of cAMP pathways and an aberrant activation of cAMP-controlled genes is linked to the growth of some cancers.[5][6][7]
Role in prefrontal cortex disorders
Recent research suggests that cAMP affects the function of higher-order thinking in the
cAMP is involved in activation of trigeminocervical system leading to neurogenic inflammation and causing migraine. [9]
Role in infectious disease agents' pathogenesis
Disrupted functioning of cAMP has been noted as one of the mechanisms of several bacterial exotoxins.
They can be subgrouped into two distinct categories:[10]
- Toxins that interfere with enzymes ADP-ribosyl-transferases, and
- invasive adenylate cyclases.
- Cholera toxin is an AB toxin that has five B subunints and one A subunit. The toxin acts by the following mechanism: First, the B subunit ring of the cholera toxin binds to GM1 gangliosides on the surface of target cells. If a cell lacks GM1 the toxin most likely binds to other types of glycans, such as Lewis Y and Lewis X, attached to proteins instead of lipids.[11][12][13][10]
Uses
Forskolin is commonly used as a tool in biochemistry to raise levels of cAMP in the study and research of cell physiology.[14]
See also
- Cyclic guanosine monophosphate (cGMP)
- 8-Bromoadenosine 3',5'-cyclic monophosphate (8-Br-cAMP)
- Acrasin specific to chemotactic use in Dictyostelium discoideum.
- phosphodiesterase 4 (PDE 4) which degrades cAMP
References
- PMID 24324443.
- PMID 27178543.
- PMID 17084085.
- ISBN 978-1-4899-0921-3.
- S2CID 14047485.
- PMID 17018604.
- ^ Simpson, B. J.; Ramage, A. D.; Hulme, M. J.; Burns, D. J.; Katsaros, D.; Langdon, S. P.; Miller, W. R. (January 1996). "American Association for Cancer Research (cAMP-binding Proteins' Presence in Tumors)". Clinical Cancer Research. 2 (1): 201–206.
- ^ "ScienceDaily ::Brain Networks Strengthened By Closing Ion Channels, Research Could Lead To ADHD Treatment".
- PMID 35052756.
- ^ S2CID 33162709. Retrieved February 26, 2022.
- .
- ^ Cervin J, Wands AM, Casselbrant A, Wu H, Krishnamurthy S, Cvjetkovic A, et al. (2018) GM1 ganglioside-independent intoxication by Cholera toxin. PLoS Pathog 14(2): e1006862. https://doi.org/10.1371/journal.ppat.1006862
- ^ Fucosylated Molecules Competitively Interfere with Cholera Toxin Binding to Host Cells; Amberlyn M. Wands, Jakob Cervin, He Huang, Ye Zhang, Gyusaang Youn, Chad A. Brautigam, Maria Matson Dzebo, Per Björklund, Ville Wallenius, Danielle K. Bright, Clay S. Bennett, Pernilla Wittung-Stafshede, Nicole S. Sampson, Ulf Yrlid, and Jennifer J. Kohler; ACS Infectious Diseases Article ASAP, DOI: 10.1021/acsinfecdis.7b00085
- ^
Alasbahi, RH; Melzig, MF (January 2012). "Forskolin and derivatives as tools for studying the role of cAMP". Die Pharmazie. 67 (1): 5–13. PMID 22393824.
