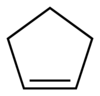
Cyclopentene
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cyclopentene | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.030 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H8 | |||
| Molar mass | 68.11 g/mol | ||
| Density | 0.771 g/cm3 | ||
| Melting point | −135 °C (−211 °F; 138 K) | ||
| Boiling point | 44 to 46 °C (111 to 115 °F; 317 to 319 K) | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −29 °C (−20 °F; 244 K) | ||
| Related compounds | |||
Related compounds
|
Cyclopentadiene Cyclobutene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cyclopentene is a
petrol-like odor. It has few applications, and thus is mainly used as a minor component of gasoline, present in concentrations of less than 1%.[1][2] It is one of the principal cycloalkenes
.
Production
Cyclopentene is produced industrially in large amounts by
vinylcyclopropane-cyclopentene rearrangement.[4]
It can also be produced by the
catalytic hydrogenation of cyclopentadiene.[5]
Reactions
The polymerization of cyclopentene by
Ziegler-Natta catalysts yields 1,3-linkages, not the more typical 1,2-linked polymer.[6]
hydrocarboxylation of cyclopentene gives cyclopentanecarboxylic acid:[7]
- C5H8 + CO + H2O → C5H9CO2H
References
- ISBN 978-3527306732.
- ^ "Hydrocarbon Composition of Gasoline Vapor Emissions from Enclosed Fuel Tanks". nepis.epa.gov. United States Environmental Protection Agency. 2011.
- .
- PMID 12683781.
- ^ D. Hönicke, R. Födisch, P. Claus, M. Olson: Cyclopentadiene and Cyclopentene, in: Ullmanns Enzyklopädie der Technischen Chemie 2002, Wiley-VCH, Weinheim.
- .
- S2CID 199466915.
External links
 Media related to Cyclopentene at Wikimedia Commons
Media related to Cyclopentene at Wikimedia Commons