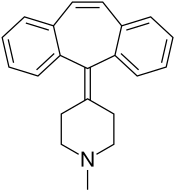
Cyproheptadine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌsaɪproʊˈhɛptədiːn/[1] |
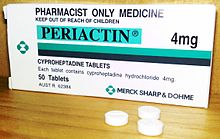
| Trade names | Periactin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682541 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Cyproheptadine, sold under the brand name Periactin among others, is a
It was patented in 1959 and came into medical use in 1961.[5] In 2021, it was the 280th most commonly prescribed medication in the United States, with more than 800,000 prescriptions.[6][7]
Medical uses

Cyproheptadine is used to treat allergic reactions (specifically
It is also used as a preventive treatment against migraine. In a 2013 study the frequency of migraine was dramatically reduced in patients within 7 to 10 days after starting treatment. The average frequency of migraine attacks in these patients before administration was 8.7 times per month, this was decreased to 3.1 times per month at 3 months after the start of treatment.[9][10] This use is on the label in the UK and some other countries.
It is also used off-label in the treatment of
Cyproheptadine is sometimes used off-label to improve akathisia in people on antipsychotic medications.[12]
It is used off-label to treat various dermatological conditions, including psychogenic itch[13] drug-induced hyperhidrosis (excessive sweating),[14] and prevention of blister formation for some people with epidermolysis bullosa simplex.[15]
One of the effects of the drug is increased appetite and weight gain, which has led to its use (off-label in the USA) for this purpose in children who are wasting as well as people with cystic fibrosis.[16][17][18][19]
It is also used off-label in the management of moderate to severe cases of
Cyproheptadine has sedative effects and can be used to treat insomnia similarly to other centrally-acting antihistamines.[22][23][24][25] The recommended dose for this use is 4 to 8 mg.[23]
Adverse effects
Adverse effects include:[3][4]
- Sedation and sleepiness (often transient)
- Dizziness
- Disturbed coordination
- Confusion
- Restlessness
- Excitation
- Nervousness
- Tremor
- Irritability
- Insomnia
- Paresthesias
- Neuritis
- Convulsions
- Euphoria
- Hallucinations
- Hysteria
- Faintness
- Allergic manifestation of rash and edema
- Diaphoresis
- Urticaria
- Photosensitivity
- Acute labyrinthitis
- Diplopia (seeing double)
- Vertigo
- Tinnitus
- Hypotension (low blood pressure)
- Palpitation
- Extrasystoles
- Anaphylactic shock
- Hemolytic anemia
- Blood dyscrasias such as leukopenia, agranulocytosis and thrombocytopenia
- Cholestasis
- Hepatic (liver) side effects such as:
- Hepatitis
- Jaundice
- Liver failure[26]
- Hepatic function abnormality
- Epigastricdistress
- Anorexia
- Nausea
- Vomiting
- Diarrhea
- Anticholinergic side effects such as:
- Blurred vision
- Constipation
- Xerostomia (dry mouth)
- Tachycardia (high heart rate)
- Urinary retention
- Difficulty passing urine
- Nasal congestion
- Nasal or throat dryness
- Urinary frequency
- Early menses
- Thickening of bronchial secretions
- Tightness of chest and wheezing
- Fatigue
- Chills
- Headache
- Increased appetite
- Weight gain
Overdose
Gastric decontamination measures such as
Pharmacology
Pharmacodynamics
| Site | Ki (nM)[a] | Action[b] | Species | Ref. |
|---|---|---|---|---|
| H1 | 0.06 | ↓ | Human | |
| H2 | ND | ND | ||
| H3 | >10,000 | Human | ||
| H4 | 202 | Human | ||
| M1 | 12 | ↓ | Human | |
| M2 | 7 | ↓ | Human | |
| M3 | 12 | ↓ | Human | |
| M4 | 8 | ↓ | Human | |
| M5 | 11.8 | ↓ | Human | |
| 5-HT1A | 59 | ↓ | Human | |
| 5-HT2A | 1.67 | ↓ | Human | |
| 5-HT2B | 1.54 | ↓ | Human | |
| 5-HT2C | 2.23 | ↓ | Human | |
| 5-HT3 | 228 | Mouse | ||
| 5-HT6 | 142 | Human | ||
| 5-HT7 | 123 | Human | ||
D1 |
117 | Human | ||
D2 |
112 | ↓ | Human | |
D3 |
8 | Human | ||
| SERT | 4,100 | Rat | ||
| NET | 290 | Rat | ||
| DAT | ND | ND | ||
| ||||
Cyproheptadine is a very potent
Pharmacokinetics
Cyproheptadine is well-absorbed following
Chemistry
Cyproheptadine is a tricyclic benzocycloheptene and is closely related to pizotifen and ketotifen as well as to tricyclic antidepressants.
Research
Cyproheptadine was studied in one small trial as an adjunct in people with schizophrenia whose condition was stable and were on other medication; while attention and verbal fluency appeared to be improved, the study was too small to draw generalizations from.[33] It has also been studied as an adjuvant in two other trials in people with schizophrenia, around fifty people overall, and did not appear to have an effect.[34]
There have been some trials to see if cyproheptadine could reduce sexual dysfunction caused by SSRI and antipsychotic medications.[35]
Cyproheptadine has been studied for the treatment of post-traumatic stress disorder.[34]
Veterinary use
Cyproheptadine is used in cats as an
Cyproheptadine is a second line treatment for pituitary pars intermedia dysfunction in horses.[40][41]
References
- ^ "Cyproheptadine". Dictionary.com Unabridged (Online). n.d.
- ^ S2CID 20196551.
- ^ a b c d "Cyproheptadine Hydrochloride tablet [Boscogen, Inc.]" (PDF). DailyMed. U.S. National Library of Medicine. November 2010. Retrieved 26 October 2013.
- ^ a b c d "Product Information: Periactin (cyproheptadine hydrochloride)" (PDF). Aspen Pharmacare Australia. Aspen Pharmacare Australia Pty Ltd. 17 November 2011. Archived from the original (PDF) on 29 October 2013. Retrieved 26 October 2013.
- ISBN 9783527607495.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Cyproheptadine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "Cyproheptadine". MedlinePlus Drug Information. U.S. National Library of Medicine.
- ^ S2CID 21185048.
- S2CID 34703034.
- PMID 27423188.
- ISBN 978-1-118-75457-3.
- PMID 27578081.
- PMID 11986151.
- PMID 20301543.
- ^ "Ciproheptadina, estimulante del apetito" [Cyproheptadine, appetite stimulant]. vademecum.es (in Spanish).
- ^ "Bioplex NF". Archived from the original on 18 April 2018. Retrieved 18 April 2018.
- S2CID 72333631.
- S2CID 237493456.
- ISBN 978-0-9805790-9-3.
- PMID 23145389.
- PMID 33632345.
- ^ S2CID 227131545.
- ProQuest 301570569.
- PMID 334082.
- PMID 25580444.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ S2CID 17640246.
- S2CID 19809259.
- PMID 9167527.
- PMID 9476091.
- ISBN 978-0-7295-3939-5. Retrieved 27 November 2011.
- S2CID 19202816.
- ^ S2CID 25769557.
- S2CID 23406005.
- S2CID 37126352.
- OCLC 794491298.
- ^ ISBN 978-0-911910-50-6. Retrieved on 26 October 2008.
- ISBN 978-0-911910-50-6. Retrieved on 26 October 2008.
- PMID 28190613.
- ^ Kritchevsky JE (July 2019). "Hirsutism Associated with Adenomas of the Pars Intermedia". Merck Vet Manual. Retrieved 24 April 2011.
