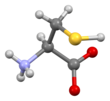
Cysteine
 Skeletal formula of L-cysteine
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Cysteine
| |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| Abbreviations | Cys, C | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.000.145 | ||
| EC Number |
| ||
| E number | E920 (glazing agents, ...) | ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
| UNII |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties[3] | |||
| C3H7NO2S | |||
| Molar mass | 121.15 g·mol−1 | ||
| Appearance | white crystals or powder | ||
| Melting point | 240 °C (464 °F; 513 K) decomposes | ||
277g/L (at 25 °C)
[1] | |||
| Solubility | 1.5g/100g ethanol 19 °C [2] | ||
Chiral rotation ([α]D)
|
+9.4° (H2O, c = 1.3) | ||
| Supplementary data page | |||
| Cysteine (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cysteine (symbol Cys or C;
The thiol is susceptible to oxidation to give the
When used as a food additive, cysteine has the E number E920.
Cysteine is
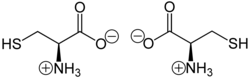
Structure
Like other amino acids (not as a residue of a protein), cysteine exists as a
Dietary sources
Cysteinyl is a residue in high-protein foods. Some foods considered rich in cysteine include poultry, eggs, beef, and whole grains. In high-protein diets, cysteine may be partially responsible for reduced blood pressure and stroke risk.[11] Although classified as a nonessential amino acid, in rare cases, cysteine may be essential for infants, the elderly, and individuals with certain metabolic diseases or who suffer from malabsorption syndromes. Cysteine can usually be synthesized by the human body under normal physiological conditions if a sufficient quantity of methionine is available.
Industrial sources
The majority of l-cysteine is obtained industrially by
Biosynthesis
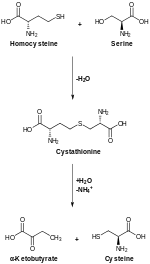
In animals, biosynthesis begins with the amino acid
Biological functions
The cysteine sulfhydryl group is
Precursor to the antioxidant glutathione
Due to the ability of thiols to undergo redox reactions, cysteine and cysteinyl residues have antioxidant properties. Its antioxidant properties are typically expressed in the tripeptide glutathione, which occurs in humans and other organisms. The systemic availability of oral glutathione (GSH) is negligible; so it must be biosynthesized from its constituent amino acids, cysteine, glycine, and glutamic acid. While glutamic acid is usually sufficient because amino acid nitrogen is recycled through glutamate as an intermediary, dietary cysteine and glycine supplementation can improve synthesis of glutathione.[19]
Precursor to iron-sulfur clusters
Cysteine is an important source of
Metal ion binding
Beyond the iron-sulfur proteins, many other metal cofactors in enzymes are bound to the thiolate substituent of cysteinyl residues. Examples include zinc in
Roles in protein structure
In the translation of messenger RNA molecules to produce polypeptides, cysteine is coded for by the UGU and UGC
Cysteine has traditionally been considered to be a
Most cysteine residues are covalently bonded to other cysteine residues to form

Disulfide bonds in proteins are formed by oxidation of the sulfhydryl group of cysteine residues. The other sulfur-containing amino acid, methionine, cannot form disulfide bonds. More aggressive oxidants convert cysteine to the corresponding sulfinic acid and sulfonic acid. Cysteine residues play a valuable role by crosslinking proteins, which increases the rigidity of proteins and also functions to confer proteolytic resistance (since protein export is a costly process, minimizing its necessity is advantageous). Inside the cell, disulfide bridges between cysteine residues within a polypeptide support the protein's tertiary structure. Insulin is an example of a protein with cystine crosslinking, wherein two separate peptide chains are connected by a pair of disulfide bonds.
Aside from its oxidation to cystine, cysteine participates in numerous
Applications
Cysteine, mainly the l-
In the field of personal care, cysteine is used for
Cysteine is a very popular target for site-directed labeling experiments to investigate biomolecular structure and dynamics.
Reducing toxic effects of alcohol
Cysteine has been proposed as a preventive or antidote for some of the negative effects of alcohol, including liver damage and hangover. It counteracts the poisonous effects of acetaldehyde.[32] Cysteine supports the next step in metabolism, which turns acetaldehyde into acetic acid.[citation needed]
In a
In 2020 an article was published that suggests L-cysteine might also work in humans.[34]
N-Acetylcysteine
Sheep
Cysteine is required by
Chemical reactions
Being multifunctional, cysteine undergoes a variety of reactions. Much attention has focused on protecting the sulfhydryl group.[37] Methylation of cysteine gives S-methylcysteine. Treatment with formaldehyde gives the thiazolidine thioproline. Cysteine forms a variety of coordination complexes upon treatment with metal ions.[38]
Safety
Relative to most other amino acids, cysteine is much more toxic.[39]
Dietary restrictions
The animal-originating sources of l-cysteine as a food additive are a point of contention for people following dietary restrictions such as kosher, halal, vegan, or vegetarian.[40] To avoid this problem, l-cysteine can also be sourced from microbial or other synthetic processes.
History
In 1884 German chemist
This section needs expansion. You can help by adding to it. (January 2023) |
See also
References
- ^ "PubChem data".
- ISBN 9783540699330.
- ISBN 0-8493-0462-8..
- ^ "cysteine - Definition of cysteine in English by Oxford Dictionaries". Oxford Dictionaries - English. Archived from the original on September 25, 2016. Retrieved 15 April 2018.
- ^ a b "The primary structure of proteins is the amino acid sequence". The Microbial World. University of Wisconsin-Madison Bacteriology Department. Archived from the original on 25 May 2013. Retrieved 16 September 2012.
- ISSN 0307-4412.
- ^ "Amber Workshop - Tutorial A1 - Section 1: Do some editing of the PDB file". ambermd.org. Archived from the original on 2022-05-22. Retrieved 2022-06-02.
- ^ S2CID 220796795.
- ^ "Amber Workshop - Tutorial A1 - Section 1: Do some editing of the PDB file". ambermd.org. Archived from the original on 2022-05-22. Retrieved 2022-06-02.
- S2CID 14895681.
- ^ "EU Chemical Requirements". Retrieved May 24, 2020.
...L-cysteine hydrochloride or hydrochloride monohydrate. Human hair may not be used as a source for this substance
- ^ "Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products". Retrieved July 28, 2021.
...ANNEX II LIST OF SUBSTANCES PROHIBITED IN COSMETIC PRODUCTS...416 Cells, tissues or products of human origin
- ^ "Questions About Food Ingredients: What is L-cysteine/cysteine/cystine?". Vegetarian Resource Group.
- .
- ISBN 978-3-527-30673-2.
- S2CID 2539629.
- PMID 9636038.
- PMID 21795440.

- PMID 16824008.
- ISBN 978-0-935702-73-6.[page needed]
- PMID 3298579.
- PMID 5650851.
- ^ "A Review of Amino Acids (tutorial)". Curtin University. Archived from the original on 2015-09-07. Retrieved 2015-09-09.
- S2CID 34980474.
- ^ Betts, M.J.; R.B. Russell (2003). "Hydrophobic amino acids". Amino Acid Properties and Consequences of Substitutions, In: Bioinformatics for Geneticists. Wiley. Retrieved 2012-09-16.
- ^ Gorga, Frank R. (1998–2001). "Introduction to Protein Structure--Non-Polar Amino Acids". Archived from the original on 2012-09-05. Retrieved 2012-09-16.
- ^ "Virtual Chembook--Amino Acid Structure". Elmhurst College. Archived from the original on 2012-10-02. Retrieved 2012-09-16.
- S2CID 2885059.
- ISBN 978-0-203-90808-2.
- ^ "Food Ingredients and Colors". U.S. Food and Drug Administration. November 2004. Archived from the original on 2009-05-12. Retrieved 2009-09-06.
- PMID 30925182.
- S2CID 5924137.
- PMID 32808029.
- S2CID 9209528.
- PMID 7569041.
- .
- .
- PMID 3309557.
- ^ "Kosher View of L-Cysteine". kashrut.com. May 2003.
- ^ Baumann, E. (1884). "Ueber Cystin und Cysteïn" [On cystine and cysteine]. Zeitschrift für physiologische Chemie (in German). 8: 299–305. From pp. 301-302: "Die Analyse der Substanz ergibt Werthe, welche den vom Cystin (C6H12N2S2O4) verlangten sich nähern, […] nenne ich dieses Reduktionsprodukt des Cystins: Cysteïn." (Analysis of the substance [cysteine] reveals values which approximate those [that are] required by cystine (C6H12N2S2O4), however the new base [cysteine] can clearly be recognized as a reduction product of cystine, to which the [empirical] formula C3H7NSO2, [which had] previously [been] ascribed to cystine, is [now] ascribed. In order to indicate the relationships of this substance to cystine, I name this reduction product of cystine: "cysteïne".) Note: Baumann's proposed structures for cysteine and cystine (see p.302) are incorrect: for cysteine, he proposed CH3CNH2(SH)COOH .
Further reading
- Nagano N, Ota M, Nishikawa K (September 1999). "Strong hydrophobic nature of cysteine residues in proteins". FEBS Lett. 458 (1): 69–71. S2CID 34980474.
External links
- Cysteine MS Spectrum
- International Kidney Stone Institute Archived 2019-05-13 at the Wayback Machine
- http://www.chemie.fu-berlin.de/chemistry/bio/aminoacid/cystein en.html Archived 2016-11-10 at the Wayback Machine
- 952-10-3056-9 Interaction of alcohol and smoking in the pathogenesis of upper digestive tract cancers - possible chemoprevention with cysteine
- Cystine Kidney Stones
- Kosher View of L-Cysteine