DFMDA
Source: Wikipedia, the free encyclopedia.
Chemical compound
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
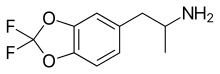
Difluoromethylenedioxyamphetamine (DFMDA, DiFMDA) is a substituted derivative of
alpha-methyldopamine, it was hoped that the difluoromethylenedioxy bioisostere would show increased metabolic stability and less toxicity.[1][2][3]
These compounds have not yet been tested in animals to verify whether they show similar pharmacological activity to the non-fluorinated parent compounds, although in vitro binding studies show DFMDA to have a SERT affinity in between that of MDA and MDMA.[4] It is also now generally accepted that MDMA neurotoxicity results from a variety of different causes and is not solely due to accumulation of alpha-methyldopamine,[5][6][7] making it unclear how much less neurotoxic DFMDA and related drugs would be in practice.
References
| |
|---|---|
| Phenylalkyl- amines (other than cathinones) |
|
| Cyclized phenyl- alkylamines | |
| Cathinones | |
| Tryptamines |
|
Chemical classes | |
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
Retrieved from "https://en.wikipedia.org/w/index.php?title=DFMDA&oldid=1215515702"
