Tenofovir alafenamide
(Redirected from
Darunavir/cobicistat/emtricitabine/tenofovir alafenamide
)This article needs to be updated. (January 2021) |
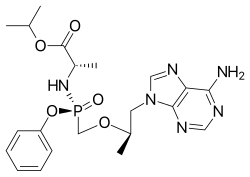 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌtəˈnoʊfəvɪər ˌæləˈfɛnəmaɪd/ |
| Trade names | Vemlidy Genvoya (with elvitegravir, cobicistat and emtricitabine) Odefsey (with emtricitabine and rilpivirine) Descovy (with emtricitabine) Symtuza (with darunavir, cobicistat, and emtricitabine) |
| Other names | GS-7340 |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | ~80%[6] |
| Elimination half-life | 0.51 hour |
| Excretion | Feces (31.7%), urine (<1%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
fumarate
, the salt used in drug formulationsTenofovir alafenamide, sold under the brand name Vemlidy, is an
antiviral medication used against hepatitis B and HIV. It is used for the treatment of chronic hepatitis B virus (HBV) infection in adults with compensated liver disease[8] and is given in combination with other medications for the prevention and treatment of HIV. It is taken by mouth.[6]
Tenofovir alafenamide is a
Fixed-dose combinations containing tenofovir alafenamide
- Elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya)[15] — approved both in the United States and in the European Union in November 2015[16][17][18][19] (compare elvitegravir/cobicistat/emtricitabine/tenofovir; (Stribild)[20][21][22][23])
- Emtricitabine/rilpivirine/tenofovir alafenamide (Odefsey)[24] — approved in the United States in March 2016, and in the European Union in June 2016[25][26][27] (compare Emtricitabine/rilpivirine/tenofovir; (Complera)[28][29][30])
- Emtricitabine/tenofovir alafenamide (Descovy)[31] — approved in the United States in April 2016 (compare emtricitabine/tenofovir; (Truvada)). In October 2019, Descovy was approved in the United States for HIV-1 pre-exposure prophylaxis (PrEP).[32][33]
- Bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy)[34] — approved in the United States in February 2018.
- Darunavir/cobicistat/emtricitabine/tenofovir alafenamide (Symtuza)[35] — approved in the European Union in September 2017, in the United States in July 2018, and in Australia in November 2019.[36][37][38][39][40]
- Dolutegravir/emtricitabine/tenofovir alafenamide.[41]
- Dolutegravir/lamivudine/tenofovir alafenamide.[42][43]
Research
Gilead announced a
elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil (Stribild) to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya), the results showed the newer drug's effects to be non-inferior to the established agent, but at much lower dosages and with lower incidence of adverse side effects such as impaired kidney function.[48][49][50] The FDA approved the TAF-based treatment regimen for treatment of HIV-1 in November 2015.[11] Genvoya is the first TAF-based regimen to receive approval.[11]
References
- ^ "Tenofovir alafenamide (Vemlidy) Use During Pregnancy". Drugs.com. 26 December 2018. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Product Monograph: Vemlidy (tenofovir alafenamide) tablets" (PDF). Government of Canada: The Drug and Health Product Register. 20 August 2020. Archived (PDF) from the original on 10 June 2022. Retrieved 7 June 2022.
- ^ "Liver disease". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Vemlidy 25 mg film coated tablets - Summary of Product Characteristics (SmPC)". (emc). 8 September 2020. Archived from the original on 11 July 2021. Retrieved 12 November 2020.
- ^ a b c "Vemlidy- tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Vemlidy EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 13 July 2021. Retrieved 12 November 2020.
- S2CID 52033420.
- S2CID 24652157.
- ^ Markowitz M, Zolopa A, Ruane P, Squires K, Zhong L, Kearney BP, et al. (March 2011). GS-7340 demonstrates greater declines in HIV-1 RNA than tenofovir disoproxil fumarate during 14 days of monotherapy in HIV-1 infected subjects (PDF). 18th Conference on Retroviruses and Opportunistic Infections. Archived from the original (PDF) on 25 April 2012.
- ^ a b c "U.S. Food and Drug Administration Approves Gilead's Single Tablet Regimen Genvoya (Elvitegravir, Cobicistat, Emtricitabine and Tenofovir Alafenamide) for Treatment of HIV-1 Infection" (Press release). Gilead. 5 November 2015. Archived from the original on 8 November 2015.
- ^ "FDA Approves Vemlidy (tenofovir alafenamide) for Chronic Hepatitis B in Adults". United States Department of Health and Human Services. 21 November 2016. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ "First Generic Drug Approvals 2023". U.S. Food and Drug Administration (FDA). 30 May 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Gilead settles 5 more Descovy patent feuds, pushing copycats to its PrEP successor out to 2031". Fierce Pharma. 12 September 2022. Archived from the original on 30 June 2023. Retrieved 27 March 2024.
- ^ "Genvoya- elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Genvoya (elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide) fixed-dose combination tablet". U.S. Food and Drug Administration (FDA). 8 December 2015. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Genvoya" (PDF). US Food and Drug Administration. 6 August 2012. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Genvoya EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 8 August 2020. Retrieved 28 July 2020.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Drug Approval Package: Stribild (elvitegravir, cobicistat, emtricitabine, tenofovir disoproxil fumarate) Fixed Dose". U.S. Food and Drug Administration (FDA). 10 October 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Stribild" (PDF). US Food and Drug Administration. 19 October 2015. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Stribild- elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 28 January 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Stribild EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Odefsey- emtricitabine, rilpivirine hydrochloride, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 6 December 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Odefsey (emtricitabine, rilpivirine, and tenofovir alafenamide) Tablets". U.S. Food and Drug Administration (FDA). 29 November 2016. Archived from the original on 13 April 2021. Retrieved 28 July 2020.
- ^ "Odefsey EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Immune system health". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Drug Approval Package: (emtricitabine/rilpivirine/tenofovir disoproxil fumarate) NDA #202123". U.S. Food and Drug Administration (FDA). 6 September 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Complera" (PDF). US Food and Drug Administration. 19 July 2011. Archived (PDF) from the original on 1 April 2021. Retrieved 29 July 2020.
- ^ "Complera- emtricitabine, rilpivirine hydrochloride, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 9 December 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Descovy- emtricitabine and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 13 January 2020. Archived from the original on 24 June 2021. Retrieved 18 April 2020.
- ^ "FDA approves second drug to prevent HIV infection as part of ongoing efforts to end the HIV epidemic". U.S. Food and Drug Administration (FDA). 3 October 2019. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ Mandavilli A (4 October 2019). "F.D.A. Approves New H.I.V.-Prevention Drug, but Not for Everyone". The New York Times. Archived from the original on 9 July 2021. Retrieved 11 October 2019.
- ^ "Biktarvy- bictegravir sodium, emtricitabine, and tenofovir alafenamide fumarate tablet". DailyMed. U.S. National Library of Medicine. 8 August 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Symtuza- darunavir, cobicistat, emtricitabine, and tenofovir alafenamide tablet, film coated". DailyMed. U.S. National Library of Medicine. 6 March 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Drug Approval Package: Symtuza (darunavir, cobicistat, emtricitabine, and tenofovir alafenamide)". U.S. Food and Drug Administration (FDA). 11 December 2018. Archived from the original on 9 July 2021. Retrieved 19 August 2020.
- ^ "Symtuza EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 10 July 2021. Retrieved 19 August 2020.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on 20 February 2022. Retrieved 20 February 2022.
- ^ "Symtuza 800/150/200/10 Tablets". NPS MedicineWise. 15 July 2021. Archived from the original on 20 February 2022. Retrieved 19 February 2022.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ "Drugs@FDA: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 9 July 2021. Retrieved 5 December 2020.
- ^ "Drugs@FDA: FDA-Approved Drugs". accessdata.fda.gov. Archived from the original on 2 April 2022. Retrieved 2 April 2022.
- ^ "Tentative Approval: Dolutegravir, Lamivudine, and Tenofovir Alafenamide Tablets" (PDF). US Food and Drug Administration. 30 March 2022. Archived (PDF) from the original on 7 May 2022. Retrieved 2 April 2022.
- ^ "Gilead Initiates Phase 3 Clinical Program for Tenofovir Alafenamide, a Novel Low-Dose Prodrug for the Treatment of HIV" (Press release). Gilead. 24 January 2013. Archived from the original on 11 October 2019.
- ^ "Gilead Sciences Finalizes Agreement with Tibotec Pharmaceuticals to Develop and Commercialize a Single-Tablet Regimen of Prezista with Emtriva, GS 7340 and Cobicistat". Gilead Sciences (Press release). 15 November 2011. Archived from the original on 11 October 2019. Retrieved 10 October 2019.
- ^ Horn T (15 March 2012). "GS-7340 Packs Greater HIV Punch, Potentially Better Safety, Versus Viread". AIDSmeds.com. Archived from the original on 8 September 2015.
- ^ "Pharmacokinetics of a Novel EVG/COBI/FTC/GS-7340 Single Tablet Regimen". 13th International Workshop on Clinical Pharmacology of HIV Therapy. Barcelona, Spain. April 2012. Archived from the original on 28 November 2020.
- ^ Once-Daily Tenofovir Prodrug Combo Pill as Effective as Stribild Archived 20 September 2015 at the Wayback Machine. AIDSmeds.com 1 Nov 2012.
- ^ CROI 2013: New Pro-drug Tenofovir Alafenamide Appears Equally Effective but Better Tolerated Archived 25 October 2020 at the Wayback Machine. Highleyman, Liz. HIVandHepatitis.com. 6 March 2013.
- ^ Horn T (13 June 2013). "Tenefovir Alafenamide Fumarate (TAF) Sign-On Letter to Gilead". Treatment Action Group. Archived from the original on 19 November 2019.
