Decalin
| |
| Names | |
|---|---|
| Preferred IUPAC name
Decahydronaphthalene[1] | |
| Other names
Bicyclo[4.4.0]decane[1]
Decalin | |
| Identifiers | |
3D model (
JSmol ) |
|
| 878165 | |
| ChEBI |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.001.861 |
| EC Number |
|
| 185147 | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1147 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H18 | |
| Molar mass | 138.25 g/mol |
| Appearance | colorless liquid |
| Density | 0.896 g/cm3 |
| Melting point | trans: −30.4 °C (−22.7 °F, 242.7 K) cis: −42.9 °C (−45.2 °F, 230.3 K)[2] |
| Boiling point | trans: 187 °C (369 °F) cis: 196 °C (384 °F) |
| Insoluble | |
| |
Refractive index (nD)
|
1.481 |
| Hazards | |
| GHS labelling: | |
     
| |
| Danger | |
| H226, H302, H305, H314, H331, H332, H410, H411 | |
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P271, P273, P280, P301+P310, P301+P330+P331, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P311, P312, P321, P331, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
| Flash point | 57 °C (135 °F; 330 K) |
| 250 °C (482 °F; 523 K) | |
| Safety data sheet (SDS) | Decalin MSDS |
| Related compounds | |
Related compounds
|
Naphthalene; Tetralin |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Decalin (decahydronaphthalene, also known as bicyclo[4.4.0]decane and sometimes decaline),
Isomers
Decalin occurs in
-
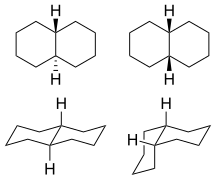
1: trans (left) and cis (right) isomers
-
2:ball-and-stick model of cis-decalin
-
3:trans-decalin
-
4:cis-decalin ring-flip
trans-Decalin
The only possible way to join the two six-membered rings in the trans position means the second ring needs to start from two equatorial bonds (blue) of the first ring. A six-membered ring does not offer sufficient space to start out on an axial position (upwards), and reach the axial position of the neighboring carbon atom, which then will be on the downwards side of the molecule (see the model of cyclohexane in figure 5). The structure is conformationally frozen. It does not have the ability to undergo a chair flip as in the cis isomer. In biology this fixation is widely used in the steroid skeleton to construct molecules (such as figure 6) that play a key role in the signalling between distantly separated cells.
Reactions
Oxygenation of decalin gives the tertiary hydroperoxide, which rearranges to cyclodecenone, a precursor to sebacic acid.[5]
Decalin is the saturated analog of
Derivation
Treatment of naphthalene in a fused state with hydrogen in the presence of a copper or nickel catalyst. [7]
Occurrence
Decalin itself is rare in nature but several decalin derivatives are known. They arise via terpene-derived precursors or polyketides.[8]
Safety
Decalin easily forms explosive[9] hydroperoxides upon storage in the presence of air.[10][11]
See also
References
- ^ ISBN 978-0-85404-182-4.
- ISBN 978-1-43982077-3.
- ^ "Dictionary.com".
- ^ "Fuel Additive Product". Archived from the original on 2009-03-12.
- ISBN 978-3527306732.
- .
- ^ [Hawley's Condensed Chemical Dictionary]
- PMID 24984916.
- ^ "PDF – Surrogate JP-8 Aviation Fuel Study – Alessandro Agosta Thesis Drexel University" (PDF). Archived from the original (PDF) on 2010-06-19.
- ^ "Inchem.org Data".
- ^ "MSDS Sheet – JT Baker".