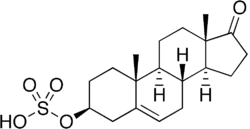
Dehydroepiandrosterone sulfate
| |
| |
| Names | |
|---|---|
| IUPAC name
17-Oxoandrost-5-en-3β-yl hydrogen sulfate
| |
| Systematic IUPAC name
(3aS,3bR,7S,9aR,9bS,11aS)-9a,11a-Dimethyl-1-oxo-2,3,3a,3b,4,6,7,8,9,9a,9b,10,11,11a-tetradecahydro-1H-cyclopenta[a]phenanthren-7-yl hydrogen sulfate | |
| Other names
Androstenolone sulfate; Prasterone sulfate; Androst-5-en-3β-ol-17-one 3β-sulfate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| Abbreviations | DHEA sulfate; DHEA-S; DHEAS |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C19H28O5S | |
| Molar mass | 368.49 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dehydroepiandrosterone sulfate, abbreviated as DHEA sulfate or DHEA-S, also known as androstenolone sulfate, is an
Biological activity
Neurosteroid activity
Similarly to other
Hormonal activity
Although DHEA-S itself is hormonally inert, it has been thought that it can be converted back into DHEA,[9] which is weakly androgenic and estrogenic, and that DHEA in turn can be transformed into more potent androgens like testosterone and dihydrotestosterone (DHT) as well as estrogens like estradiol.[2][1][10] As such, it has been thought that DHEA-S is a prohormone with the potential for androgenic and estrogenic effects.[2][1][10] However, a 2005 study found that DHEA could be converted into DHEA-S but found no evidence of conversion of DHEA-S into DHEA.[11]
Other activity
DHEA-S has also been found to inhibit the
Biochemistry
Biosynthesis
DHEA and DHEA-S are produced in the
Distribution
Unlike DHEA, which is weakly bound to
Whereas DHEA easily crosses the blood–brain barrier into the central nervous system,[23] DHEA-S poorly crosses the blood–brain barrier.[24]
Metabolism
DHEA-S can be converted back into DHEA via
The elimination half-life of DHEA-S is 7 to 10 hours, which is far longer than that of DHEA, which has an elimination half-life of only 15 to 30 minutes.[21]
Elimination
DHEA-S is excreted in the urine via the kidneys.[27]
Levels

DHEA and DHEA-S are the most abundant circulating steroids in the body.[29] Plasma levels of DHEA-S are 100 or more times higher than those of DHEA, 5 to 10 times higher than those of cortisol, 100 to 500 times those of testosterone, and 1,000 to 10,000 times higher than those of estradiol.[30][3]
Levels of DHEA and DHEA-S vary throughout life.[2][1] They remain low during childhood until adrenarche around 6 to 8 years of age, at which point they markedly increase,[31] eventually peaking at around 20 to 30 years of age.[2][1] From the third decade of life on, DHEA and DHEA-S levels gradually decrease.[29] By the age of 70, levels of DHEA and DHEA-S are 80 to 85% lower than those of young adults, and in people more than 80 years of age, DHEA and DHEA-S levels can reach 80 to 90% lower than those of younger individuals.[29]
DHEA-S levels are higher in men than in women.[2][29]
Reference ranges
Tanner stage and average age |
Lower limit | Upper limit | Unit | |
|---|---|---|---|---|
| Tanner stage I | >14 days | 16 | 96 | μg/dL |
| Tanner stage II | 10.5 years | 22 | 184 | |
| Tanner stage III | 11.6 years | <15 | 296 | |
| Tanner stage IV | 12.3 years | 17 | 343 | |
| Tanner stage V | 14.5 years | 44 | 332 | |
| 18–29 years | 44 | 332 | ||
| 30–39 years | 31 | 228 | ||
| 40–49 years | 18 | 244 | ||
| 50–59 years | <15 | 200 | ||
| > or =60 years | <15 | 157 | ||
Tanner stage and average age |
Lower limit | Upper limit | Unit | |
|---|---|---|---|---|
| Tanner stage I | >14 days | <15 | 120 | μg/dL |
| Tanner stage II | 11.5 years | <15 | 333 | |
| Tanner stage III | 13.6 years | <15 | 312 | |
| Tanner stage IV | 15.1 years | 29 | 412 | |
| Tanner stage V | 18.0 years | 89 | 457 | |
| 18–29 years | 89 | 457 | ||
| 30–39 years | 65 | 334 | ||
| 40–49 years | 48 | 244 | ||
| 50–59 years | 35 | 179 | ||
| > or =60 years | 25 | 131 | ||
Medical use
Deficiency
The Endocrine Society recommends against the therapeutic use of DHEA-S in both healthy women and those with adrenal insufficiency, as its role is not clear from studies performed so far.[33] The routine use of DHEA-S and other androgens is discouraged in the treatment of women with low androgen levels due to hypopituitarism, adrenal insufficiency, menopause due to ovarian surgery, glucocorticoid use, or other conditions associated with low androgen levels; this is because there are limited data supporting improvement in signs and symptoms with therapy and no long-term studies of risk.[33]
In otherwise elderly women, in whom an age-related fall of DHEA-S may be associated with menopausal symptoms and reduced libido, DHEA-S supplementation cannot currently be said to improve outcomes.[34]
Childbirth
As the
Diagnostic use
DHEA-S levels above 1890 μM or 700 to 800 μg/dL are highly suggestive of
Women with
Chemistry
DHEA-S, also known as androst-5-en-3β-ol-17-one 3β-sulfate, is a naturally occurring androstane steroid and the C3β sulfate ester of DHEA.
References
- ^ ISBN 978-0-444-51830-9.
- ^ PMID 26908835.
- ^ ISBN 978-3-540-88367-8.
Plasma DHEA-S levels in adult men and women are 100-500 times higher than those of testosterone and 1000-10000 times higher than those of estradiol.
- S2CID 30489004.
- ^ ISBN 978-1-4614-5559-2.
- PMID 21541365.
- PMID 25330101.
- S2CID 26914550.
- ^ ISBN 978-0-203-30121-0.
- ^ ISBN 978-1-4511-4847-3.
- PMID 15755854.
- ISSN 2002-4436.
- ^ PMID 17945481.
- PMID 26213785.
- ISBN 978-1-59259-832-8.
- ISBN 978-3-540-33713-3.
- ISBN 978-0-323-06986-1.
- ISBN 978-3-540-26861-1.
- ISBN 978-1-4471-0931-0.
- ISBN 978-0-7817-1750-2.
- ^ ISBN 978-0-323-08704-9.
- ISBN 978-0-8247-5504-1.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ISBN 978-1-4377-2333-5.
- ISBN 978-0-7216-6897-0.
Thus, the formation of DHEA-S occurs directly in the brain, particularly because DHEA-S does not cross the blood-brain barrier [...]
- PMID 18493132.
- PMID 30029732.
- ISBN 978-0-203-45043-7.
- ISBN 978-1-4557-5973-6.
- ^ ISBN 978-1-84184-952-2.
- ISBN 978-1-4020-6854-6.
- ISBN 978-1-4419-1436-1.
- ^ a b Dehydroepiandrosterone Sulfate (DHEA-S), Serum Archived 2018-03-14 at the Wayback Machine at Mayo Foundation For Medical Education And Research. Retrieved July 2012
- ^ PMID 25279570.
- PMID 25279571.
- ISBN 978-1-4757-2085-3.
- ISBN 978-0-8493-8217-8.
- ISBN 978-3-13-179525-0.
- ISBN 978-3-527-30247-5.
3β-Hydroxyandrost-5-en-17-one hydrogen sulfate = (3β)-3-(Sulfooxy)androst-5-en-17-one. R: Sodium salt (1099-87-2). S: Astenile, Dehydroepiandrosterone sulfate sodium, DHA-S, DHEAS, KYH 3102, Mylis, PB 005, Prasterone sodium sulfate, Teloin
- ^ Jianqiu Y (1992). "Clinical Application of Prasterone Sodium Sulfate". Chinese Journal of New Drugs. 5: 015.
- PMID 1403604.
- .
- S2CID 34029116.
- ^ "Polycystic Ovarian Syndrome Workup". eMedicine. 25 October 2011. Retrieved 19 November 2011.
- PMID 22649387.
- ^ PMID 20418968.
