Demethylation
Demethylation is the chemical process resulting in the removal of a methyl group (CH3) from a molecule.[1][2] A common way of demethylation is the replacement of a methyl group by a hydrogen atom, resulting in a net loss of one carbon and two hydrogen atoms.
The counterpart of demethylation is methylation.
In biochemistry

Dicamba, a widely used herbicide, biodegrades by demethylation to give 3,6-dichlorosalicylic acid, catalyzed by a dioxygenase enzyme.[3]
Demethylation is relevant to
- R2N-CH3 + O → R2N-H + CH2O
One family of such oxidative enzymes is the
Demethylation of some sterols are steps in the biosynthesis of testosterone and cholesterol. Methyl groups are lost as formate.[7]
Biomass processing
Methoxy groups heavily decorate the biopolymer lignin. Much interest has been shown in converting this abundant form of biomass into useful chemicals (aside from fuel). One step in such processing is demethylation. [8][9] The demethylation of vanillin, a derivative of lignin, requires 250 °C (482 °F) and strong base.[10] Pulp and paper industry]] digests lignin using aqueous sodium sulfide, which partially depolymerizes the lignin. Delignification is accompanied by extensive O-demethylation, yielding methanethiol, which is emitted by paper mills.[11]
In organic chemistry
Demethylation often refers to cleavage of
Historically, aryl methyl ethers, including natural products such as

Another classical (but, again, harsh) method for the removal of the methyl group of an aryl methyl ether is to heat the ether in a solution of hydrogen bromide or hydrogen iodide sometimes also with acetic acid.[17] The cleavage of ethers by hydrobromic or hydroiodic acid proceeds by protonation of the ether, followed by displacement by bromide or iodide. A slightly milder set of conditions uses cyclohexyl iodide (CyI, 10.0 equiv) in N,N-dimethylformamide to generate a small amount of hydrogen iodide in situ.[18]
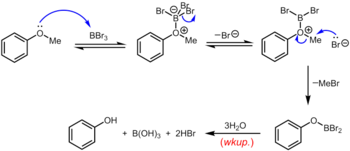
Boron tribromide, which can be used at room temperature or below, is a more specialized reagent for the demethylation of aryl methyl ethers. The mechanism of ether dealkylation proceeds via the initial reversible formation of a Lewis acid-base adduct between the strongly Lewis acidic BBr3 and the Lewis basic ether. This Lewis adduct can reversibly dissociate to give a dibromoboryl oxonium cation and Br–. Rupture of the ether linkage occurs through the subsequent nucleophilic attack on the oxonium species by Br– to yield an aryloxydibromoborane and methyl bromide. Upon completion of the reaction, the phenol is liberated along with boric acid (H3BO3) and hydrobromic acid (aq. HBr) upon hydrolysis of the dibromoborane derivative during aqueous workup.[19]
Stronger
Aromatic methyl ethers, particularly those with an adjacent carbonyl group, can be regioselectively demethylated using magnesium iodide etherate.[22] An example of this being used is in the synthesis of the natural product Calphostin A,[23] as seen below.

Methyl esters also are susceptible to demethylation, which is usually achieved by saponification. Highly specialized demethylations are abundant, such as the Krapcho decarboxylation:
A mixture of anethole, KOH, and alcohol was heated in an autoclave. Although the product of this reaction was the expected anol, a highly reactive dimerization product in the mother liquors called dianol was also discovered by Charles Dodds.
N-demethylation
N-demethylation of 3° amines is by the
The N-demethylation of imipramine gives desipramine.
See also
- Methylation, the addition of a methyl group to a substrate
References
- ISBN 978-0-19-850346-0.
- ISBN 978-0-471-72091-1
- PMID 19616011.
- PMID 20863703.
- ISBN 978-0-470-01672-5.
- PMID 24153300.
- PMID 31402327.
- .
- PMID 14503002.
- ^ Irwin A. Pearl (1949). "Protocatechulic Acid". Organic Syntheses. 29: 85; Collected Volumes, vol. 3, p. 745.
- PMID 13904415.
- .
- PMID 833817.
- OCLC 190810761.
- ^ ISSN 0009-2665.
- )
- OCLC 52836313.
- .
- ^ J. F. W. McOmie, D. E. West (1969). "3,3'-Dihydroxybiphenyl". Organic Syntheses. 49: 13; Collected Volumes, vol. 5, p. 412.
- ISBN 978-0471264224.
- .
- .
- PMID 23335811.


