Dendritic spine
| Dendritic spine | |
|---|---|
 Spiny dendrite of a striatal medium spiny neuron. | |
 Common types of dendritic spines. | |
| Details | |
| Identifiers | |
| Latin | gemmula dendritica |
| MeSH | D049229 |
| TH | H2.00.06.1.00036 |
| Anatomical terms of microanatomy | |
A dendritic spine (or spine) is a small membranous protrusion from a neuron's dendrite that typically receives input from a single axon at the synapse. Dendritic spines serve as a storage site for synaptic strength and help transmit electrical signals to the neuron's cell body. Most spines have a bulbous head (the spine head), and a thin neck that connects the head of the spine to the shaft of the dendrite. The dendrites of a single neuron can contain hundreds to thousands of spines. In addition to spines providing an anatomical substrate for memory storage and synaptic transmission, they may also serve to increase the number of possible contacts between neurons.[1] It has also been suggested that changes in the activity of neurons have a positive effect on spine morphology.[2]
Structure
Dendritic spines are small with spine head volumes ranging 0.01 μm3 to 0.8 μm3. Spines with strong synaptic contacts typically have a large spine head, which connects to the dendrite via a membranous neck. The most notable classes of spine shape are "thin", "stubby", "mushroom", and "bifurcated".
Distribution
Dendritic spines usually receive excitatory input from axons, although sometimes both inhibitory and excitatory connections are made onto the same spine head.[4] Excitatory axon proximity to dendritic spines is not sufficient to predict the presence of a synapse, as demonstrated by the Lichtman lab in 2015.[5]
Spines are found on the
Cytoskeleton and organelles
The cytoskeleton of dendritic spines is particularly important in their
In addition to their electrophysiological activity and their receptor-mediated activity, spines appear to be vesicularly active and may even translate
Morphogenesis
The morphogenesis of dendritic spines is critical to the induction of long-term potentiation (LTP).[7][8] The morphology of the spine depends on the states of actin, either in globular (G-actin) or filamentous (F-actin) forms. The role of Rho family of GTPases and its effects in the stability of actin and spine motility[9] has important implications for memory. If the dendritic spine is the basic unit of information storage, then the spine's ability to extend and retract spontaneously must be constrained. If not, information may be lost. Rho family of GTPases makes significant contributions to the process that stimulates actin polymerization, which in turn increases the size and shape of the spine.[10] Large spines are more stable than smaller ones and may be resistant to modification by additional synaptic activity.[11] Because changes in the shape and size of dendritic spines are correlated with the strength of excitatory synaptic connections and heavily depend on remodeling of its underlying actin cytoskeleton,[12] the specific mechanisms of actin regulation, and therefore the Rho family of GTPases, are integral to the formation, maturation, and plasticity of dendritic spines and to learning and memory.
RhoA pathway
One of the major Rho GTPases involved in spine morphogenesis is
A study conducted by Murakoshi et al. in 2011 implicated the Rho GTPases RhoA and Cdc42 in dendritic spine morphogenesis. Both GTPases were quickly activated in single dendritic spines of
Cdc42 pathway
Cdc42 has been implicated in many different functions including dendritic growth, branching, and branch stability.[14] Calcium influx into the cell through NMDA receptors binds to calmodulin and activates the Ca2+/calmodulin-dependent protein kinases II (CaMKII). In turn, CaMKII is activated and this activates Cdc42, after which no feedback signaling occurs upstream to calcium and CaMKII. If tagged with monomeric-enhanced green fluorescent protein, one can see that the activation of Cdc42 is limited to just the stimulated spine of a dendrite. This is because the molecule is continuously activated during plasticity and immediately inactivates after diffusing out of the spine. Despite its compartmentalized activity, Cdc42 is still mobile out of the stimulated spine, just like RhoA. Cdc42 activates PAK, which is a protein kinase that specifically phosphorylates and, therefore, inactivates ADF/cofilin.[15] Inactivation of cofilin leads to increased actin polymerization and expansion of the spine's volume. Activation of Cdc42 is required for this increase in spinal volume to be sustained.
Observed changes in structural plasticity

Murakoshi, Wang, and Yasuda (2011) examined the effects of Rho GTPase activation on the structural plasticity of single dendritic spines elucidating differences between the transient and sustained phases.[10]
Transient changes in structural plasticity
Applying a low-frequency train of two-photon glutamate uncaging in a single dendritic spine can elicit rapid activation of both RhoA and Cdc42. During the next two minutes, the volume of the stimulated spine can expand to 300 percent of its original size. However, this change in spine morphology is only temporary; the volume of the spine decreases after five minutes. Administration of C3 transferase, a Rho inhibitor, or glycyl-H1152, a Rock inhibitor, inhibits the transient expansion of the spine, indicating that activation of the Rho-Rock pathway is required in some way for this process.[10]
Sustained changes in structural plasticity
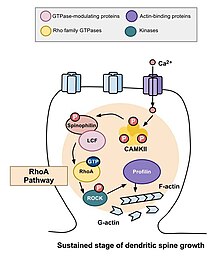
After the transient changes described above take place, the spine's volume decreases until it is elevated by 70 to 80 percent of the original volume. This sustained change in structural plasticity will last about thirty minutes. Once again, administration of C3 transferase and Glycyl-H1152 suppressed this growth, suggesting that the Rho-Rock pathway is necessary for more persistent increases in spinal volume. In addition, administration of the Cdc42 binding domain of Wasp or inhibitor targeting Pak1 activation-3 (IPA3) decreases this sustained growth in volume, demonstrating that the Cdc42-Pak pathway is needed for this growth in spinal volume as well. This is important because sustained changes in structural plasticity may provide a mechanism for the encoding, maintenance, and retrieval of memories. The observations made may suggest that Rho GTPases are necessary for these processes.[10]
Physiology
Receptor activity
Dendritic spines express
Glutamate receptors (GluRs) are localized to the postsynaptic density, and are anchored by cytoskeletal elements to the membrane. They are positioned directly above their signalling machinery, which is typically tethered to the underside of the plasma membrane, allowing signals transmitted by the GluRs into the
Signalling from GluRs is mediated by the presence of an abundance of proteins, especially kinases, that are localized to the postsynaptic density. These include
Spines are particularly advantageous to neurons by compartmentalizing biochemical signals. This can help to encode changes in the state of an individual synapse without necessarily affecting the state of other synapses of the same neuron. The length and width of the spine neck has a large effect on the degree of compartmentalization, with thin spines being the most biochemically isolated spines.
Plasticity
Dendritic spines are very "plastic", that is, spines change significantly in shape, volume, and number in small time courses. Because spines have a primarily actin cytoskeleton, they are dynamic, and the majority of spines change their shape within seconds to minutes because of the dynamicity of actin remodeling. Furthermore, spine number is very variable and spines come and go; in a matter of hours, 10-20% of spines can spontaneously appear or disappear on the pyramidal cells of the cerebral cortex, although the larger "mushroom"-shaped spines are the most stable.
Spine maintenance and plasticity is activity-dependent
Importance to learning and memory
Evidence of importance

Spine plasticity is implicated in motivation, learning, and memory.[18][19][20] In particular, long-term memory is mediated in part by the growth of new dendritic spines (or the enlargement of pre-existing spines) to reinforce a particular neural pathway. Because dendritic spines are plastic structures whose lifespan is influenced by input activity,[21] spine dynamics may play an important role in the maintenance of memory over a lifetime.
Age-dependent changes in the rate of spine turnover suggest that spine stability impacts developmental learning. In youth, dendritic spine turnover is relatively high and produces a net loss of spines.
Experience-induced changes in dendritic spine stability also point to spine turnover as a mechanism involved in the maintenance of long-term memories, though it is unclear how sensory experience affects neural circuitry. Two general models might describe the impact of experience on structural plasticity. On the one hand, experience and activity may drive the discrete formation of relevant synaptic connections that store meaningful information in order to allow for learning. On the other hand, synaptic connections may be formed in excess, and experience and activity may lead to the pruning of extraneous synaptic connections.[1]
In lab animals of all ages, environmental enrichment has been related to dendritic branching, spine density, and overall number of synapses.[1] In addition, skill training has been shown to lead to the formation and stabilization of new spines while destabilizing old spines,[18][24] suggesting that the learning of a new skill involves a rewiring process of neural circuits. Since the extent of spine remodeling correlates with success of learning, this suggests a crucial role of synaptic structural plasticity in memory formation.[24] In addition, changes in spine stability and strengthening occur rapidly and have been observed within hours after training.[18][19]
Conversely, while enrichment and training are related to increases in spine formation and stability, long-term sensory deprivation leads to an increase in the rate of spine elimination[1][22] and therefore impacts long-term neural circuitry. Upon restoring sensory experience after deprivation in adolescence, spine elimination is accelerated, suggesting that experience plays an important role in the net loss of spines during development.[22] In addition, other sensory deprivation paradigms—such as whisker trimming—have been shown to increase the stability of new spines.[25]
Research in neurological diseases and injuries shed further light on the nature and importance of spine turnover. After stroke, a marked increase in structural plasticity occurs near the trauma site, and a five- to eightfold increase from control rates in spine turnover has been observed.[26] Dendrites disintegrate and reassemble rapidly during ischemia—as with stroke, survivors showed an increase in dendritic spine turnover.[27] While a net loss of spines is observed in Alzheimer's disease and cases of intellectual disability, cocaine and amphetamine use have been linked to increases in dendritic branching and spine density in the prefrontal cortex and the nucleus accumbens.[28] Because significant changes in spine density occur in various brain and spinal cord diseases, this suggests a balanced state of spine dynamics in normal circumstances, which may be susceptible to disequilibrium under varying pathological conditions.[28][29]
There is also some evidence for loss of dendritic spines as a consequence of aging. One study using mice has noted a correlation between age-related reductions in spine densities in the hippocampus and age-dependent declines in hippocampal learning and memory.[30] Emerging evidence has also shown dendritic spine abnormalities in the pain processing regions of the spinal cord nociceptive system, including superficial and intermediate zones of the dorsal horn.[31][29][32][33]
Overall, the evidence suggests that dendritic spines are crucial for normal brain and spinal cord function. Alterations in spine morphology may not only influence synaptic plasticity and information processing but also have a key role in many neurological diseases. Furthermore, even subtle changes in dendritic spine densities or sizes can affect neuronal network properties,[34] which could lead to cognitive or mood alterations, impaired learning and memory, as well as pain hypersensitivity.[29] Moreover, the findings suggest that maintaining spine health through therapies such as exercise, cognitive stimulation and lifestyle modifications may be helpful in preserving neuronal plasticity and improving neurological symptoms.
Importance contested
Despite experimental findings that suggest a role for dendritic spine dynamics in mediating learning and memory, the degree of structural plasticity's importance remains debatable. For instance, studies estimate that only a small portion of spines formed during training actually contribute to lifelong learning.[24] In addition, the formation of new spines may not significantly contribute to the connectivity of the brain, and spine formation may not bear as much of an influence on memory retention as other properties of structural plasticity, such as the increase in size of spine heads.[35]
Modeling
Theoreticians have for decades hypothesized about the potential electrical function of spines, yet our inability to examine their electrical properties has until recently stopped theoretical work from progressing too far. Recent advances in imaging techniques along with increased use of two-photon glutamate uncaging have led to a wealth of new discoveries; we now suspect that there are voltage-dependent sodium,[36] potassium,[37] and calcium[38] channels in the spine heads.[39]
Cable theory provides the theoretical framework behind the most "simple" method for modelling the flow of electrical currents along passive neural fibres. Each spine can be treated as two compartments, one representing the neck, the other representing the spine head. The compartment representing the spine head alone should carry the active properties.
Baer and Rinzel's continuum model
To facilitate the analysis of interactions between many spines, Baer & Rinzel formulated a new cable theory for which the distribution of spines is treated as a continuum.[40] In this representation, spine head voltage is the local spatial average of membrane potential in adjacent spines. The formulation maintains the feature that there is no direct electrical coupling between neighboring spines; voltage spread along dendrites is the only way for spines to interact.
Spike-diffuse-spike model
The SDS model was intended as a computationally simple version of the full Baer and Rinzel model.[41] It was designed to be analytically tractable and have as few free parameters as possible while retaining those of greatest significance, such as spine neck resistance. The model drops the continuum approximation and instead uses a passive dendrite coupled to excitable spines at discrete points. Membrane dynamics in the spines are modelled using integrate and fire processes. The spike events are modelled in a discrete fashion with the wave form conventionally represented as a rectangular function.
Modeling spine calcium transients
Calcium transients in spines are a key trigger for synaptic plasticity.
Development
Dendritic spines can develop directly from dendritic shafts or from
Spines, however, require maturation after formation. Immature spines have impaired signaling capabilities, and typically lack "heads" (or have very small heads), only necks, while matured spines maintain both heads and necks.
Clinical significance
Emerging research has indicate abnormalities in spine density in anxiety disorders.[4]
Cognitive disorders such as
History
Dendritic spines were first described at the end of the 19th century by Santiago Ramón y Cajal on cerebellar neurons.[48] Ramón y Cajal then proposed that dendritic spines could serve as contacting sites between neurons. This was demonstrated more than 50 years later thanks to the emergence of electron microscopy.[49] Until the development of confocal microscopy on living tissues, it was commonly admitted that spines were formed during embryonic development and then would remain stable after birth. In this paradigm, variations of synaptic weight were considered as sufficient to explain memory processes at the cellular level. But since about a decade ago, new techniques of confocal microscopy demonstrated that dendritic spines are indeed motile and dynamic structures that undergo a constant turnover, even after birth.[50][51][44]
References
- ^ PMID 17280523.
- PMID 19519307.
- S2CID 234472935.
- ^ PMID 34702942.
- PMID 26232230.
- S2CID 12180359.
- PMID 10341235.
- PMID 10823894.
- S2CID 21100601.
- ^ PMID 21423166.
- S2CID 18436944.
- PMID 20457765.
- PMID 9605859.
- PMID 12716918.
- PMID 16443821.
- PMID 17517683.
- PMID 22492034.
- ^ PMID 19946267.
- ^ PMID 20164928.
- PMID 22405211.
- PMID 18788894.
- ^ S2CID 16232150.
- S2CID 13320649.
- ^ PMID 19946265.
- S2CID 4428322.
- PMID 17428988.
- S2CID 46267737.
- ^ PMID 19575680.
- ^ PMID 36461773.
- S2CID 30838296.
- PMID 32371602.
- PMID 28326929.
- PMID 26936986.
- PMID 19692517.
- PMID 12740121.
- PMID 17640908.
- S2CID 385712.
- S2CID 4271356.
- ^ PMID 25619656.
- PMID 2051208.
- S2CID 3058796.
- PMID 17065442.
- PMID 19074019.
- ^ S2CID 5054448.
- PMID 26051541.
- PMID 32019166.
- PMID 21346746.
- ^ Ramón y Cajal S (1888). "Estructura de los centros nerviosos de las aves". Rev. Trim. Histol. Norm. Pat. 1: 1–10.
- S2CID 4258584.
- PMID 8622128.
- S2CID 10183317.
Further reading
- Levitan IB, Kaczmarek LK (2002). The Neuron: Cell and Molecular Biology (Third ed.). New York: Oxford University Press. ISBN 978-0-19-514522-9.
- Sudhof TC, Stevens CF, Cowan WM (2001). Synapses. Baltimore: The Johns Hopkins University Press. ISBN 978-0-8018-6498-8.
- Kasai H, Matsuzaki M, Noguchi J, Yasumatsu N (October 2002). "[Dendritic spine structures and functions]". Nihon Shinkei Seishin Yakurigaku Zasshi = Japanese Journal of Psychopharmacology. 22 (5): 159–164. PMID 12451686.
- Lieshoff C, Bischof HJ (March 2003). "The dynamics of spine density changes". Behavioural Brain Research. 140 (1–2): 87–95. S2CID 2275781.
- Lynch G, Rex CS, Gall CM (January 2007). "LTP consolidation: substrates, explanatory power, and functional significance". Neuropharmacology. 52 (1): 12–23. S2CID 22652804.
- Matsuzaki M, Honkura N, Ellis-Davies GC, Kasai H (June 2004). "Structural basis of long-term potentiation in single dendritic spines". Nature. 429 (6993): 761–766. PMID 15190253.
- Nimchinsky EA, Sabatini BL, Svoboda K (2002). "Structure and function of dendritic spines". Annual Review of Physiology. 64: 313–353. PMID 11826272.
- Yuste R, Majewska A, Holthoff K (July 2000). "From form to function: calcium compartmentalization in dendritic spines". Nature Neuroscience. 3 (7): 653–659. S2CID 33466678.
