Denitrification
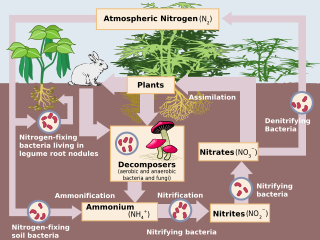
Denitrification is a microbially facilitated process where nitrate (NO3−) is reduced and ultimately produces molecular
The process is performed primarily by
Direct reduction from nitrate to ammonium, a process known as dissimilatory nitrate reduction to ammonium or DNRA,[6] is also possible for organisms that have the nrf-gene.[7][8] This is less common than denitrification in most ecosystems as a means of nitrate reduction. Other genes known in microorganisms which denitrify include nir (nitrite reductase) and nos (nitrous oxide reductase) among others;[3] organisms identified as having these genes include Alcaligenes faecalis, Alcaligenes xylosoxidans, many in the genus Pseudomonas, Bradyrhizobium japonicum, and Blastobacter denitrificans.[9]
Overview
Half reactions
Denitrification generally proceeds through some combination of the following half reactions, with the enzyme catalyzing the reaction in parentheses:
- NO3− + 2 H+ + 2 e− → NO
2− + H2O (Nitrate reductase) - NO
2− + 2 H+ + e− → NO + H2O (Nitrite reductase) - 2 NO + 2 H+ + 2 e− → N
2O + H2O (Nitric-oxide reductase) - N
2O + 2 H+ + 2 e− → N
2 + H2O (Nitrous-oxide reductase)
The complete process can be expressed as a net balanced
- 2 NO3− + 10 e− + 12 H+ → N2 + 6 H2O
Conditions of denitrification
In nature, denitrification can take place in both terrestrial and marine
Furthermore, denitrification can occur in oxic environments as well. High activity of denitrifiers can be observed in the intertidal zones, where the tidal cycles cause fluctuations of oxygen concentration in sandy coastal sediments.[14] For example, the bacterial species Paracoccus denitrificans engages in denitrification under both oxic and anoxic conditions simultaneously. Upon oxygen exposure, the bacteria is able to utilize nitrous oxide reductase, an enzyme that catalyzes the last step of denitrification.[15] Aerobic denitrifiers are mainly Gram-negative bacteria in the phylum Proteobacteria. Enzymes NapAB, NirS, NirK and NosZ are located in the periplasm, a wide space bordered by the cytoplasmic and the outer membrane in Gram-negative bacteria.[16]
A variety of environmental factors can influence the rate of denitrification on an ecosystem-wide scale. For example, temperature and pH have been observed to impact denitrification. In bacterial species Pseudomonas mandelii, expression of denitrifying genes was reduced at temperatures below 30 °C and a pH below 5, while activity was largely unaffected between a pH of 6-8.[17] Organic carbon as an electron donor is a common limiting nutrient for denitrification as observed in benthic sediments and wetlands.[18][19] Nitrate and oxygen can also be potential limiting factors for denitrification, although the latter only has an observed limiting effect in wet soils.[20] Oxygen likely affects denitrification in multiple ways—because most denitrifiers are facultative, oxygen can inhibit rates, but it can also stimulate denitrification by facilitating nitrification and the production of nitrate. In wetlands as well as deserts,[21] moisture is an environmental limiter on rates of denitrification.
Additionally, environmental factors can also influence whether denitrification proceeds to completion, characterized by the complete reduction of NO3- to N2 rather than releasing N2O as an end product. Soil pH and texture are both factors that can moderate denitrification, with higher pH levels driving the reaction more to completion.[22] Nutrient composition, particularly the ratio of carbon to nitrogen, is a strong contributor to complete denitrification,[23] with a 2:1 ratio of C:N being able to facilitate full nitrate reduction regardless of temperature or carbon source.[24] Copper, as a co-factor for nitrite reductase and nitrous-oxide reductase, also promoted complete denitrification when added as a supplement.[25] Besides nutrients and terrain, microbial community composition can also affect the ratio of complete denitrification, with prokaryotic phyla Actinomycetota and Thermoproteota being responsible for greater release of N2 than N2O compared to other prokaryotes.[26]
Denitrification can lead to a condition called isotopic fractionation in the soil environment. The two stable isotopes of nitrogen, 14N and 15N are both found in the sediment profiles. The lighter isotope of nitrogen, 14N, is preferred during denitrification, leaving the heavier nitrogen isotope, 15N, in the residual matter. This selectivity leads to the enrichment of 14N in the biomass compared to 15N.[27] Moreover, the relative abundance of 14N can be analyzed to distinguish denitrification apart from other processes in nature.
Use in wastewater treatment
Denitrification is commonly used to remove nitrogen from
Reduction under anoxic conditions can also occur through process called anaerobic ammonium oxidation (anammox):[33]
- NH4+ + NO2− → N2 + 2 H2O
In some
Aerobic denitrification, conducted by aerobic denitrifiers, may offer the potential to eliminate the need for separate tanks and reduce sludge yield. There are less stringent alkalinity requirements because alkalinity generated during denitrification can partly compensate for the alkalinity consumption in nitrification.[16]
Non-biological denitrification
A variety of non-biological methods can remove nitrate. These include methods that can destroy nitrogen compounds, such as chemical and electrochemical methods, and those that selectively transfer nitrate to a concentrated waste stream, such as ion exchange or reverse osmosis. Chemical removal of nitrate can occur through advanced oxidation processes, although it may produce hazardous byproducts.[38] Electrochemical methods can remove nitrate by via a voltage applied across electrodes, with degradation usually occurring at the cathode. Effective cathode materials include transition metals, post transition metals,[39] and semi-conductors like TiO2.[40] Electrochemical methods can often avoid requiring costly chemical additives, but their effectiveness can be constrained by the pH and ions present. Reverse osmosis is highly effective in removing small charged solutes like nitrate, but it may also remove desirable nutrients, create large volumes of wastewater, and require increased pumping pressures. Ion exchange can selectively remove nitrate from water without large waste streams,[41] but do require regeneration and may face challenges with absorption of undesired ions.
See also
- Aerobic denitrification
- Anaerobic respiration
- Bioremediation
- Climate change
- Hypoxia (environmental)
- Nitrogen fixation
- Simultaneous nitrification-denitrification
References
- PMID 6407395.
- S2CID 22428082.
- ^ PMID 9409151.
- ISBN 0-8053-0653-6
- PMID 33658719.
- .
- S2CID 3948918.
- ISBN 978-1-4020-3542-5.)
It is possible to encounter DNRA when your source of carbon is a fermentable substrate, as glucose, so if you wanna avoid DNRA use a non fermentable substrate
{{cite book}}: CS1 maint: multiple names: authors list (link - PMID 12788762.
- PMID 17205890.
- S2CID 20602996.
- .
- PMID 20562276.
- PMID 28463234.
- PMID 26568281.
- ^ S2CID 85744076.
- PMID 19376915.
- ISSN 0017-467X.
- ISSN 0925-8574.
- ISSN 0361-5995.
- ISSN 0361-5995.
- ISSN 2673-8619.
- PMID 22074905.
- ISSN 1369-703X.
- ISSN 2296-665X.
- ISSN 0038-0717.
- – via Copernicus Publications.
- .
- S2CID 96384737.
- S2CID 18518.
- PMID 25638338. (subscription required)
- S2CID 10397713. (subscription required)
- PMID 15862442.
- .
- S2CID 6445650.
- PMID 25078442.
- .
- ISSN 1385-8947.
- )
- S2CID 231818217.
- PMID 18143940.
