Diabetes medication
Drugs used in diabetes treat
Several drug classes are indicated for use in type 2 diabetes and are often used in combination. Therapeutic combinations may include several insulin isoforms or varying classes of oral antihyperglycemic agents. As of 2020, 23 unique antihyperglycemic drug combinations were approved by the FDA.[1] The first triple combination of oral anti-diabetics was approved in 2019, consisting of metformin, saxagliptin, and dapagliflozin. Another triple combination approval for metformin, linagliptin, and empagliflozin followed in 2020.[1]
Mechanisms of action
Diabetes medications have four main mechanisms of action:[citation needed]
- Insulin sensitization: Increased sensitivity of insulin receptors on cells leading to decreasedinsulin resistance, and higher effects of insulin on blood glucose levels.
- Stimulation of beta cells: This stimulation increases insulin secretion from beta cells of pancreas.
- alpha-glucosidaseenzyme decreases the rate at which glucose is absorbed from the gastrointestinal tract.
- SGLT2 inhibition: Inhibition of sodium-glucose transport protein 2 (SGLT2) decreases glucose reabsorption in the renal tubules of nephrons, thus increasing the amount of glucose excreted in urine.
Insulin
Insulin is usually given subcutaneously, either by injections or by an insulin pump. In acute care settings, insulin may also be given intravenously. Insulins are typically characterized by the rate at which they are metabolized by the body, yielding different peak times and durations of action.[2] Faster-acting insulins peak quickly and are subsequently metabolized, while longer-acting insulins tend to have extended peak times and remain active in the body for more significant periods.[3]
Examples of rapid-acting insulins (peak at ~1 hour) are:
- Insulin lispro (Humalog)
- Insulin aspart (Novolog)
- Insulin glulisine (Apidra)
Examples of short-acting insulins (peak 2–4 hours) are:
- Regular insulin (Humulin R, Novolin R)
- Prompt insulin zinc (Semilente)
Examples of intermediate-acting insulins (peak 4–10 hours) are:
- Isophane insulin, neutral protamine Hagedorn (NPH) (Humulin N, Novolin N)
- Insulin zinc (Lente)
Examples of long-acting insulins (duration 24 hours, often without peak) are:
- Extended insulin zinc insulin (Ultralente)
- Insulin glargine (Lantus)
- Insulin detemir (Levemir)
- Insulin degludec (Tresiba)
Insulin degludec is sometimes classed separately as an "ultra-long" acting insulin due to its duration of action of about 42 hours, compared with 24 hours for most other long-acting insulin preparations.[3]
As a systematic review of studies comparing insulin detemir, insulin glargine, insulin degludec and NPH insulin did not show any clear benefits or serious
Most oral anti-diabetic agents are contraindicated in pregnancy, in which case insulin is preferred.[5]
Insulin is not administered by other routes, although this has been studied. An inhaled form was briefly licensed but was subsequently withdrawn.[citation needed]
Sensitizers
Insulin sensitizers address the core problem in type 2 diabetes – insulin resistance.
Biguanides
Biguanides reduce hepatic glucose output and increase uptake of glucose by the periphery, including skeletal muscle. Although it must be used with caution in patients with impaired liver or kidney function, Metformin, a biguanide, has become the most commonly used agent for type 2 diabetes in children and teenagers. Among common diabetic drugs, Metformin is the only widely used oral drug that does not cause weight gain.[citation needed]
Typical reduction in glycated hemoglobin (A1C) values for Metformin is 1.5–2.0%
- contrast, as patients are at an increased risk of lactic acidosis.
- Phenformin (DBI) was used from 1960s through 1980s, but was withdrawn due to lactic acidosis risk.[7]
- Buformin also was withdrawn due to lactic acidosis risk.[8]
Metformin is a first-line medication used for treatment of type 2 diabetes. It is generally prescribed at initial diagnosis in conjunction with exercise and weight loss, as opposed to the past, where it was prescribed after diet and exercise had failed. There is an immediate-release as well as an extended-release formulation, typically reserved for patients experiencing
Thiazolidinediones
Typical reductions in glycated hemoglobin (A1C) values are 1.5–2.0%. Some examples are:
- Rosiglitazone (Avandia): the European Medicines Agency recommended in September 2010 that it be suspended from the EU market due to elevated cardiovascular risks.[10]
- Pioglitazone (Actos): remains on the market but has also been associated with increased cardiovascular risks.[11]
- Troglitazone (Rezulin): used in 1990s, withdrawn due to hepatitis and liver damage risk.[12]
Multiple retrospective studies have resulted in a concern about rosiglitazone's safety, although it is established that the group, as a whole, has beneficial effects on diabetes. The greatest concern is an increase in the number of severe cardiac events in patients taking it. The ADOPT study showed that initial therapy with drugs of this type may prevent the progression of disease,[13] as did the DREAM trial.[14] The American Association of Clinical Endocrinologists (AACE), which provides clinical practice guidelines for management of diabetes, retains thiazolidinediones as recommended first, second, or third line agents for type 2 diabetes mellitus, as of their 2019 executive summary, over sulfonylureas and α-glucosidase inhibitors. However, they are less preferred than GLP-1 agonists or SGLT2 inhibitors, especially in patients with cardiovascular disease (which liraglutide, empagliflozin, and canagliflozin are all FDA approved to treat).[15]
Concerns about the safety of rosiglitazone arose when a retrospective meta-analysis was published in
In contrast, at least one large prospective study, PROactive 05, has shown that pioglitazone may decrease the overall incidence of cardiac events in people with type 2 diabetes who have already had a heart attack.[19]
LYN Kinase Activators
The LYN kinase activator Tolimidone has been reported to potentiate insulin signaling in a manner that is distinct from the glitazones.[20] The compound has demonstrated positive results in a Phase 2a clinical study involving 130 diabetic subjects.[21]
Secretagogues
Secretagogues are drugs that increase output from a gland, in the case of insulin from the pancreas.
Sulfonylureas
Sulfonylureas were the first widely used oral anti-hyperglycemic medications. They are insulin secretagogues, triggering insulin release by inhibiting the KATP channel of the pancreatic beta cells. Eight types of these pills have been marketed in North America, but not all remain available. The "second-generation" sulfonylureas are now more commonly used. They are more effective than first-generation drugs and have fewer side-effects. All may cause weight gain.
Current clinical practice guidelines from the AACE rate sulfonylureas (as well as glinides) below all other classes of antidiabetic drugs in terms of suggested use as first, second, or third line agents - this includes Bromocriptine, the bile acid sequestrant Colesevelam, α-glucosidase inhibitors, Thiazolidinediones (glitazones), and DPP-4 inhibitors (gliptins).[15] The low cost of most sulfonylureas, however, especially when considering their significant efficacy in blood glucose reduction, tends to keep them as a more feasible option in many patients - neither SGLT2 inhibitors nor GLP-1 agonists, the classes most favored by the AACE guidelines after metformin, are currently available as generics.
Sulfonylureas bind strongly to
A Cochrane systematic review from 2011 showed that treatment with Sulfonylureas did not improve control of glucose levels more than insulin at 3 nor 12 months of treatment.[23] This same review actually found evidence that treatment with Sulfonylureas could lead to earlier insulin dependence, with 30% of cases requiring insulin at 2 years.[23] When studies measured fasting C-peptide, no intervention influenced its concentration, but insulin maintained concentration better compared to Sulphonylurea.[23] Still, it is important to highlight that the studies available to be included in this review presented considerable flaws in quality and design.[23]
Typical reductions in glycated hemoglobin (A1C) values for second-generation sulfonylureas are 1.0–2.0%.
- First-generation agents
- Second-generation agents
- glipizide
- glyburide or glibenclamide
- glimepiride
- gliclazide
- glyclopyramide
- gliquidone
Meglitinides
Meglitinides help the pancreas produce insulin and are often called "short-acting secretagogues." They act on the same potassium channels as sulfonylureas, but at a different binding site.[24] By closing the potassium channels of the pancreatic beta cells, they open the calcium channels, thereby enhancing insulin secretion.[25]
They are taken with or shortly before meals to boost the insulin response to each meal. If a meal is skipped, the medication is also skipped.
Typical reductions in glycated hemoglobin (A1C) values are 0.5–1.0%.
Adverse reactions include weight gain and hypoglycemia.
Alpha-glucosidase inhibitors
Typical reductions in glycated hemoglobin (A1C) values are 0.5–1.0%.
These medications are rarely used in the United States because of the severity of their side-effects (flatulence and bloating). They are more commonly prescribed in Europe. They do have the potential to cause weight loss by lowering the amount of sugar metabolized.
Peptide analogs
This section needs additional citations for verification. (January 2016) |
Injectable incretin mimetics
Injectable glucagon-like peptide analogs and agonists
Glucagon-like peptide (GLP) agonists bind to a membrane GLP receptor.[25] As a consequence, insulin release from the pancreatic beta cells is increased. Endogenous GLP has a half-life of only a few minutes, thus an analogue of GLP would not be practical. As of 2019, the AACE lists GLP-1 agonists, along with SGLT2 inhibitors, as the most preferred anti-diabetic agents after metformin. Liraglutide in particular may be considered first-line in diabetic patients with cardiovascular disease, as it has received FDA approval for reduction of risk of major adverse cardiovascular events in patients with type 2 diabetes.[15][26] In a 2011 Cochrane review, GLP-1 agonists showed approximately a 1% reduction in HbA1c when compared to placebo.[22] GLP-1 agonists also show improvement of beta-cell function, but this effect does not last after treatment is stopped.[22] Due to shorter duration of studies, this review did not allow for long-term positiver or negative effects to be assessed.[22]
- Exenatide (also Exendin-4, marketed as Byetta) is the first GLP-1 agonist approved for the treatment of type 2 diabetes. Exenatide is not an analogue of GLP but rather a GLP agonist.[27][28] Exenatide has only 53% homology with GLP, which increases its resistance to degradation by DPP-4 and extends its half-life.[29] A 2011 Cochrane review showed a HbA1c reduction of 0.20% more with Exenatide 2 mg compared to insulin glargine, exenatide 10 µg twice daily, sitagliptin and pioglitazone.[22] Exenatide, together with liraglutide, led to greater weight loss than glucagon-like peptide analogues.[22]
- U.S. Food and Drug Administration (FDA) on January 25, 2010.[30][31][32][33][34][35] A 2011 Cochrane review showed a HbA1c reduction of 0.24% more with liraglutide 1.8 mg compared to insulin glargine, 0.33% more than exenatide 10 µg twice daily, sitagliptin and rosiglitazone.[22] Liraglutide, together with exenatide, led to greater weight loss than glucagon-like peptide analogues.[22]
- Hoffman-La Roche.
- Lixisenatide (Lyxumia) Sanofi Aventis
- Semaglutide (Ozempic) (oral version is Rybelsus)
- Trulicity) - once weekly
- Albiglutide (Tanzeum) - once weekly
These agents may also cause a decrease in gastric motility, responsible for the common side-effect of nausea, which tends to subside with time.[22]
Gastric inhibitory peptide analogs
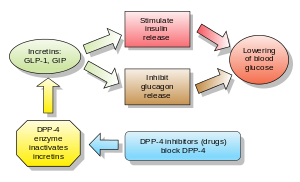
Dipeptidyl peptidase-4 inhibitors
GLP-1 analogs resulted in weight loss and had more gastrointestinal side-effects, while in general dipeptidyl peptidase-4 (DPP-4) inhibitors were weight-neutral and are associated with increased risk for infection and headache. Both classes appear to present an alternative to other antidiabetic drugs. However, weight gain and/or hypoglycemia have been observed when dipeptidyl peptidase-4 inhibitors were used with sulfonylureas; effects on long-term health and morbidity rates are still unknown.[36]
DPP-4 inhibitors increase blood concentration of the incretin GLP-1 by inhibiting its degradation by DPP-4.
Examples are:
- vildagliptin (Galvus) EU Approved 2008
- sitagliptin (Januvia) FDA approved Oct 2006
- saxagliptin (Onglyza) FDA Approved July 2009
- linagliptin (Tradjenta) FDA Approved May 2, 2011
- alogliptin
- septagliptin
- teneligliptin
- gemigliptin (Zemiglo)
DPP-4 inhibitors lowered hemoglobin A1C values by 0.74%, comparable to other antidiabetic drugs.[37]
A result in one RCT comprising 206 patients aged 65 or older (mean baseline HgbA1c of 7.8%) receiving either 50 or 100 mg/d of sitagliptin was shown to reduce HbA1c by 0.7% (combined result of both doses).[38] A combined result of 5 RCTs enlisting a total of 279 patients aged 65 or older (mean baseline HbA1c of 8%) receiving 5 mg/d of saxagliptin was shown to reduce HbA1c by 0.73%.[39] A combined result of 5 RCTs enlisting a total of 238 patients aged 65 or older (mean baseline HbA1c of 8.6%) receiving 100 mg/d of vildagliptin was shown to reduce HbA1c by 1.2%.[40] Another set of 6 combined RCTs involving alogliptin (approved by FDA in 2013) was shown to reduce HbA1c by 0.73% in 455 patients aged 65 or older who received 12.5 or 25 mg/d of the medication.[41]
Injectable amylin analogues
SGLT2 inhibitors
Examples include:
- Dapagliflozin
- Canagliflozin
- Empagliflozin
- Remogliflozin
The side effects of SGLT2 inhibitors are derived directly from their mechanism of action; these include an increased risk of: ketoacidosis, urinary tract infections, candidal vulvovaginitis, and hypoglycemia.[46]
Comparison
The following table compares some common anti-diabetic agents, generalizing classes, although there may be substantial variation in individual drugs of each class. When the table makes a comparison such as "lower risk" or "more convenient" the comparison is with the other drugs on the table.
| Comparison of anti-diabetic medication[47][48] | ||||
|---|---|---|---|---|
| Drug class[48] | Mechanism of action[5] | Advantages[48] | Disadvantages[48] | |
glyburide, glimepiride, glipizide )
|
Stimulating insulin release by pancreatic beta cells by inhibiting the KATP channel |
|
| |
| Metformin | Acts on the liver to reduce gluconeogenesis and causes a decrease in insulin resistance via increasing AMPK signalling. |
|
| |
| Alpha-glucosidase inhibitors (acarbose, miglitol, voglibose) | Inhibit carbohydrate digestion in the small intestine by inhibiting enzymes that break down polysaccharides |
|
| |
Thiazolidinediones (Pioglitazone, Rosiglitazone )
|
Reduce insulin resistance by activating PPAR-γ in fat and muscle |
|
| |
SGLT2 inhibitors
| ||||
Generics
Many anti-diabetes drugs are available as generics. These include:[49]
- Sulfonylureas– glimepiride, glipizide, glyburide
- Biguanides– metformin
- Thiazolidinediones(Tzd) – pioglitazone, Actos generic
- Alpha-glucosidase inhibitors – Acarbose
- Meglitinides – nateglinide
- Combination of sulfonylureas plus metformin – known by generic names of the two drugs
No generics are available for dipeptidyl peptidase-4 inhibitors (Onglyza), the glifozins, the incretins and various combinations. Sitagliptin patent expired in July 2022, leading to launch of generic sitagliptin[50] brands . This lowered the cost of therapy for type 2 diabetes using sitagliptin .
Alternative Medicine
The effect of Ayurvedic treatments has been researched, however due to methodological flaws of relevant studies and research, it has not been possible to draw conclusions regarding efficacy of these treatments and there is insufficient evidence to recommend them.[51]
References
- ^ PMID 35126141.
- ISBN 978-0071748896.
- ^ PMID 25905175. Retrieved November 16, 2019.
- ^ PMID 33662147.
- ^ ISBN 978-0-7817-7153-5.
- PMID 17761999.
- from the original on December 9, 2012.
- PMID 7202882.
- ^ "diabetesinsulinPPAR". www.healthvalue.net. Archived from the original on March 3, 2016. Retrieved May 6, 2018.
- ^ European Medicines Agency, "European Medicines Agency recommends suspension of Avandia, Avandamet and Avaglim" Archived February 3, 2014, at the Wayback Machine, EMA, 23 September 2009
- PMID 17848652.
- Wired News. Archivedfrom the original on December 4, 2008. Retrieved June 21, 2009.
- ^ Haffner SM (2007). "Expert Column – A Diabetes Outcome Progression Trial (ADOPT)". Medscape. Retrieved September 21, 2007.
- ^ Gagnon L (October 24, 2006). "DREAM: Rosiglitazone Effective in Preventing Diabetes". Medscape. Archived from the original on December 2, 2008. Retrieved September 21, 2007.
- ^ PMID 30742570.
- PMID 17517853.
- ^ Wood S (July 31, 2007). "FDA Advisory Panels Acknowledge Signal of Risk With Rosiglitazone, but Stop Short of Recommending Its Withdrawal". Heartwire. Archived from the original on March 18, 2014. Retrieved September 21, 2007.
- S2CID 73109231.
- PMID 17466227.
- PMID 10848597.
- ^ "Melior Pharmaceuticals Announces Positive Phase 2A Results in Type 2 Diabetes Study". businesswire.com. June 13, 2016. Archived from the original on August 12, 2017. Retrieved May 6, 2018.
- ^ PMID 21975753.
- ^ PMID 21901702.
- PMID 15647714.
- ^ a b "Helping the pancreas produce insulin". HealthValue. Archived from the original on September 27, 2007. Retrieved September 21, 2007.
- ^ "Victoza (liraglutide) is Approved to Reduce the Risk of Three Major Adverse Cardiovascular Events in Type 2 Diabetes Patients". Drugs.com. Retrieved November 16, 2019.
- S2CID 43740629.
- S2CID 8800490.
- S2CID 195691202.
- ^ "Novo Nordisk Files for Regulatory Approval of Liraglutide in Both the US and Europe". Archived from the original on December 15, 2017. Retrieved January 23, 2018. May 2008
- ^ "Liraglutide Provides Significantly Better Glucose Control Than Insulin Glargine in Phase 3 Study". Archived from the original on July 23, 2010. Retrieved February 9, 2010. "Liraglutide Provides Significantly Better Glucose Control Than Insulin Glargine In Phase 3 Study" June 2007
- ^ "Clinical Study Shows Liraglutide Reduced Blood Sugar, Weight, and Blood Pressure in Patients with Type 2 Diabetes". Archived from the original on February 5, 2009. Retrieved February 9, 2010. "Clinical Study Shows Liraglutide Reduced Blood Sugar, Weight, And Blood Pressure In Patients With Type 2 Diabetes" June 2008
- ^ "Liraglutide – Next-Generation Antidiabetic Medication". Archived from the original on June 18, 2010. Retrieved February 9, 2010.
- ^ "Quarterly R&D; Update - Novo Nordisk A/S". Archived from the original on January 9, 2010. Retrieved February 9, 2010. Oct 2008 Inc results of LEAD 6 extension
- ^ "Novo Nordisk Receives US Approval for Victoza(R) (Liraglutide) for the Treatment of Type 2 Diabetes". Archived from the original on January 29, 2010. Retrieved February 9, 2010. January 2009
- ^ National Prescribing Service (August 1, 2010). "Dipeptidyl peptidase-4 inhibitors ('gliptins') for type 2 diabetes mellitus". RADAR. Retrieved March 7, 2021.
- PMID 17622601.
- ^ Barzilei N, Mahoney EM, Guo H (2009). "Sitagliptin is well tolerated and leads to rapid improvement in blood glucose in the first days of monotherapy in patients aged 65 years and older with T2DM". Diabetes. 58: 587.
- S2CID 206965817.
- PMID 17878242.
- S2CID 28683917.
- PMID 24285921.
- ^ Center for Drug Evaluation and Research. "Drug Safety and Availability - Sodium-glucose Cotransporter-2 (SGLT2) Inhibitors". www.fda.gov. Archived from the original on November 29, 2016. Retrieved August 26, 2017.
- ^ "UpToDate". www.uptodate.com. Retrieved November 16, 2019.
- ^ Imamovic Kadric S, Kulo Cesic A, Dujic T. Pharmacogenetics of new classes of antidiabetic drugs. Bosn J of Basic Med Sci. 2021. DOI: https://doi.org/10.17305/bjbms.2021.5646
- ^ "SGLT2 Inhibitors (Gliflozins) – Drugs, Suitability, Benefits & Side Effects". Archived from the original on August 27, 2017. Retrieved August 26, 2017.
- PMID 17666560. adapted from table 2, which includes a list of issues
- ^ a b c d Consumer Reports Health Best Buy Drugs. "The Oral Diabetes Drugs: Treating Type 2 Diabetes" (PDF). Best Buy Drugs. Consumer Reports: 20. Archived (PDF) from the original on February 27, 2013. Retrieved September 18, 2012., which is citing
- Agency for Healthcare Research and Quality (March 2011). "Oral Diabetes Medications for Adults With Type 2 Diabetes. An Update" (PDF). Comparative Effectiveness Review (27). Archived from the original (PDF) on September 27, 2013. Retrieved November 28, 2012.
- Bennett WL, Maruthur NM, Singh S, Segal JB, Wilson LM, Chatterjee R, et al. (May 2011). "Comparative effectiveness and safety of medications for type 2 diabetes: an update including new drugs and 2-drug combinations". Annals of Internal Medicine. 154 (9): 602–613. PMID 21403054.
- ^ "The Oral Diabetes Drugs Treating Type 2 Diabetes Comparing Effectiveness, Safety, and Price" (PDF). Archived (PDF) from the original on June 15, 2013. Retrieved July 17, 2013.
- ^ "Sitagliptin Generic Alternatives". www.sastimedic.com. January 31, 2024. Retrieved January 31, 2024.
- PMID 22161426.
Further reading
- Lebovitz, Harold E. (2004). Therapy For Diabetes Mellitus and Related Disorders (4th ed.). Alexandria, VA: ISBN 978-1-58040-187-6.
- Adams, Michael Ian; Holland, Norman Norwood (2003). Core Concepts in Pharmacology. Englewood Cliffs, NJ: Prentice Hall. ISBN 978-0-13-089329-1.
