Diatomic carbon
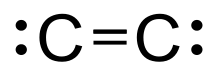
| |

| |
| Names | |
|---|---|
| IUPAC name
Diatomic carbon
| |
| Systematic IUPAC name
Ethenediylidene (substitutive) Dicarbon(C—C) (additive) | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
| 196 | |
PubChem CID
|
|
| |
| |
| Properties | |
| C2 | |
| Molar mass | 24.022 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diatomic carbon (systematically named dicarbon and 1λ2,2λ2-ethene), is a green, gaseous
Properties
C2 is a component of carbon vapor. One paper estimates that carbon vapor is around 28% diatomic,[2] but theoretically this depends on the temperature and pressure.
Electromagnetic properties
The electrons in diatomic carbon are distributed among the molecular orbitals according to the Aufbau principle to produce unique quantum states, with corresponding energy levels. The state with the lowest energy level, or ground state, is a singlet state (1Σ+
g), which is systematically named ethene-1,2-diylidene or dicarbon(0•). There are several excited singlet and triplet states that are relatively close in energy to the ground state, which form significant proportions of a sample of dicarbon under ambient conditions. When most of these excited states undergo photochemical relaxation, they emit in the infrared region of the electromagnetic spectrum. However, one state in particular emits in the green region. That state is a triplet state (3Πg), which is systematically named ethene-μ,μ-diyl-μ-ylidene or dicarbon(2•). In addition, there is an excited state somewhat further in energy from the ground state, which only form a significant proportion of a sample of dicarbon under mid-ultraviolet irradiation. Upon relaxation, this excited state fluoresces in the violet region and phosphoresces in the blue region. This state is also a singlet state (1Πg), which is also named ethene-μ,μ-diyl-μ-ylidene or dicarbon(2•).
State Excitation
enthalpy
(kJ mol−1)Relaxation
transitionRelaxation
wavelengthRelaxation EM-region X1Σ+
g0 – – – a3Π
u8.5 a3Π
u→X1Σ+
g14.0 μm Long-wavelength infrared b3Σ−
g77.0 b3Σ−
g→a3Π
u1.7 μm Short-wavelength infrared A1Π
u100.4 A1Π
u→X1Σ+
g
A1Π
u→b3Σ−
g1.2 μm
5.1 μmNear infrared
Mid-wavelength infraredB1Σ+
g? B1Σ+
g→A1Π
u
B1Σ+
g→a3Π
u?
??
?c3Σ+
u159.3 c3Σ+
u→b3Σ−
g
c3Σ+
u→X1Σ+
g
c3Σ+
u→B1Σ+
g1.5 μm
751.0 nm
?Short-wavelength infrared
Near infrared
?d3Π
g239.5 d3Π
g→a3Π
u
d3Π
g→c3Σ+
u
d3Π
g→A1Π
u518.0 nm
1.5 μm
860.0 nmGreen
Short-wavelength infrared
Near infraredC1Π
g409.9 C1Π
g→A1Π
u
C1Π
g→a3Π
u
C1Π
g→c3Σ+
u386.6 nm
298.0 nm
477.4 nmViolet
Mid-ultraviolet
Blue
In certain forms of crystalline carbon, such as diamond and graphite, a saddle point or "hump" occurs at the bond site in the charge density. The triplet state of C2 does follow this trend. However, the singlet state of C2 acts more like silicon or germanium; that is, the charge density has a maximum at the bond site.[6]
Reactions
Diatomic carbon will react with acetone and acetaldehyde to produce acetylene by two different pathways.[2]
- Triplet C2 molecules will react through an intermolecular pathway, which is shown to exhibit diradical character. The intermediate for this pathway is the ethylene radical. Its abstraction is correlated with bond energies.[2]
- vinylidene. The singlet reaction can happen through a 1,1-diabstraction or a 1,2-diabstraction. This reaction is insensitive to isotope substitution. The different abstractions are possibly due to the spatial orientations of the collisions rather than the bond energies.[2]
- Singlet C2 will also react with alkenes. Acetylene is a main product; however, it appears C2 will insert into carbon-hydrogen bonds.
- C2 is 2.5 times more likely to insert into a methyl group as into methylene groups.[7]
- There is a disputed possible room-temperature chemical synthesis via alkynyl-λ3-iodane.[8][9]
History

The light of gas-rich comets mainly originates from the emission of diatomic carbon. An example is C/2014 Q2 (Lovejoy), where there are several lines of C2 light, mostly in the visible spectrum[10] , forming the Swan bands.[11] C/2022 E3 (ZTF), visible in early 2023, also exhibits green color due to the presence of diatomic carbon.[12]
See also
- Acetylide – a related chemical with the formula C2−
2
References
- JSTOR 29775475. Archived from the original(PDF) on 2023-03-21. Retrieved 2017-07-22.
- ^ .
- ^ PMID 29896370.
- PMID 22354433.
- PMID 22354425.
- PMID 9975266.
- .
- PMID 32358541.
- PMID 33623013.
- .
- ^ Mikuz, Herman; Dintinjana, Bojan (1994). "CCD Photometry of Comets". International Comet Quarterly. Retrieved October 26, 2006.
- ^ Georgiou, Aristos (2023-01-10). "What makes the green comet green?". Newsweek. Archived from the original on 2023-01-25. Retrieved 2023-01-25.
