Diborane

| |

| |
| Names | |
|---|---|
| IUPAC name
Diborane(6)
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.039.021 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1911 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| B2H6 | |
| Molar mass | 27.67 g·mol−1 |
| Appearance | Colorless gas |
| Odor | repulsive and sweet |
| Density | 1.131 g/L[1] |
| Melting point | −164.85 °C (−264.73 °F; 108.30 K)[1] |
| Boiling point | −92.49 °C (−134.48 °F; 180.66 K)[1] |
| Reacts[2] | |
| Solubility in other solvents | Diethyl Ether,[3]
|
| Vapor pressure | 39.5 atm (16.6 °C)[2] |
| Structure | |
| Tetrahedral (for boron) | |
| see text | |
| 0 D | |
| Thermochemistry | |
Heat capacity (C)
|
56.7 J/(mol·K)[4] |
Std molar
entropy (S⦵298) |
232.1 J/(mol·K)[4] |
Std enthalpy of (ΔfH⦵298)formation |
36.4 kJ/mol[4] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic, highly flammable, reacts with water |
| GHS labelling: | |
   
| |
| Danger | |
| H220, H280, H314, H330, H370, H372 | |
| P210, P260, P264, P270, P271, P280, P284, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P307+P311, P310, P314, P320, P321, P363, P377, P381, P403, P403+P233, P405, P410+P403, P501 | |
| NFPA 704 (fire diamond) | |
| 38 °C (100 °F; 311 K) | |
Explosive limits
|
0.8–88%[2] |
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration)
|
40 ppm (rat, 4 h) 29 ppm (mouse, 4 h) 40–80 ppm (rat, 4 h) 159–181 ppm (rat, 15 min)[5] |
LCLo (lowest published)
|
125 ppm (dog, 2 h) 50 ppm (hamster, 8 h)[5] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.1 ppm (0.1 mg/m3)[2] |
REL (Recommended)
|
TWA 0.1 ppm (0.1 mg/m3)[2] |
IDLH (Immediate danger) |
15 ppm[2] |
| Safety data sheet (SDS) | ICSC 0432 |
| Related compounds | |
Related boron compounds
|
Decaborane BF3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diborane(6), commonly known as diborane, is the
Structure and bonding
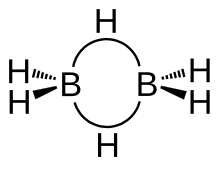
The structure of diborane has
The model determined by molecular orbital theory describes the bonds between boron and the terminal hydrogen atoms as conventional 2-center 2-electron covalent bonds. The bonding between the boron atoms and the bridging hydrogen atoms is, however, different from that in molecules such as hydrocarbons. Each boron uses two electrons in bonding to the terminal hydrogen atoms and has one valence electron remaining for additional bonding. The bridging hydrogen atoms provide one electron each. The B2H2 ring is held together by four electrons forming two 3-center 2-electron bonds. This type of bond is sometimes called a "banana bond".
B2H6 is
Of the other elements in group IIIA, gallium is known to form a similar compound digallane, Ga2H6. Aluminium forms a polymeric hydride, (AlH3)n; although unstable, Al2H6 has been isolated in solid hydrogen and is isostructural with diborane.[10]
Production and synthesis
Extensive studies of diborane have led to the development of multiple synthesis routes. Most preparations entail reactions of hydride donors with boron halides or alkoxides. The industrial synthesis of diborane involves the reduction of BF3 by sodium hydride (NaH), lithium hydride (LiH) or lithium aluminium hydride (LiAlH4):[11]
- 8 BF3 + 6 LiH → B2H6 + 6 LiBF4
Two laboratory methods start from boron trichloride with lithium aluminium hydride or from boron trifluoride ether solution with sodium borohydride. Both methods result in as much as 30% yield:
- 4 BCl3 + 3 LiAlH4 → 2 B2H6 + 3 LiAlCl4
- 4 BF3 + 3 NaBH4 → 2 B2H6 + 3 NaBF4
When heated with NaBH4, tin(II) chloride is reduced to elemental tin, forming diborane in the process:
- SnCl2 + 2NaBH4 → 2NaCl + Sn + B2H6 + H2
Older methods entail the direct reaction of borohydride salts with a non-oxidizing acid, such as phosphoric acid or dilute sulfuric acid.
- 2 BH4− + 2 H+ → 2 H2 + B2H6
Similarly, oxidation of borohydride salts has been demonstrated and remains convenient for small-scale preparations. For example, using iodine as an oxidizer:[13]
- 2 NaBH
4 + I
2 → 2 NaI + B
2H
6 + H
2
Another small-scale synthesis uses potassium hydroborate and phosphoric acid as starting materials.[14]
Reactions

Diborane is a highly reactive and versatile reagent.[16]
Air, water, oxygen
As a
and water:Diborane reacts violently with water to form hydrogen and boric acid:
- B2H6 + 6 H2O → 2 B(OH)3 + 6 H2 (ΔHr = −466 kJ/mol = −16.82 kJ/g)
Diborane also reacts with alcohols similarly. Methanol for example give hydrogen and
- B2H6 + 6 MeOH → 2 B(OMe)3 + 6 H2
Lewis acidity
One dominating reaction pattern involves formation of adducts with
With ammonia diborane forms the diammoniate of diborane, DADB with small quantities of ammonia borane as byproduct. The ratio depends on the conditions.
Hydroboration
In the
Other
Pyrolysis of diborane gives hydrogen and diverse boron hydride clusters. For example, pentaborane was first prepared by pyrolysis of diborane at about 200 °C.[18][19] Although this pyrolysis route is rarely employed, it ushered in a large research theme of borane cluster chemistry.
Treating diborane with sodium amalgam gives NaBH4 and Na[B3H8][17] When diborane is treated with lithium hydride in diethyl ether, lithium borohydride is formed:[17]
- B2H6 + 2 LiH → 2 LiBH4
Diborane reacts with anhydrous hydrogen chloride or hydrogen bromide gas to give a boron halohydride:[17]
- B2H6 + HX → B2H5X + H2 (X = Cl, Br)
Treating diborane with carbon monoxide at 470 K and 20 bar gives H3BCO.[17]
Reagent in organic synthesis
Diborane and its variants are central organic synthesis reagents for hydroboration. Alkenes add across the B–H bonds to give trialkylboranes, which can be further elaborated.[20] Diborane is used as a reducing agent roughly complementary to the reactivity of lithium aluminium hydride. The compound readily reduces carboxylic acids to the corresponding alcohols, whereas ketones react only sluggishly.
History
Diborane was first synthesised in the 19th century by hydrolysis of metal borides, but it was never analysed. From 1912 to 1936, Alfred Stock, the major pioneer in the chemistry of boron hydrides, undertook his research that led to the methods for the synthesis and handling of the highly reactive, volatile, and often toxic boron hydrides. He proposed the first ethane-like structure of diborane.[21] Electron diffraction measurements by S. H. Bauer initially appeared to support his proposed structure.[22][23]
Because of a personal communication with L. Pauling (who supported the ethane-like structure), H. I. Schlessinger and A. B. Burg did not specifically discuss 3-center 2-electron bonding in their then classic review in the early 1940s.[24] The review does, however, discuss the bridged D2h structure in some depth: "It is to be recognized that this formulation easily accounts for many of the chemical properties of diborane..."
In 1943,
Traditionally, diborane has often been described as electron-deficient, because the 12 valence electrons can only form 6 conventional 2-centre 2-electron bonds, which are insufficient to join all 8 atoms.[30][31] However, the more correct description using 3-centre bonds shows that diborane is really electron-precise, since there are just enough valence electrons to fill the 6 bonding molecular orbitals.[32] Nevertheless, some leading textbooks still use the term "electron-deficient".[33]
Other uses
Because of the exothermicity of its reaction with oxygen, diborane has been tested as a rocket propellant.[34] Complete combustion is strongly exothermic. However, combustion is not complete in the rocket engine, as some boron monoxide, B2O, is produced. This conversion mirrors the incomplete combustion of hydrocarbons, to produce carbon monoxide (CO). Diborane also proved difficult to handle.[35][36][37]
Diborane has been investigated as a precursor to metal boride films[38] and for the p-doping of silicon semiconductors.[39]
Safety
Diborane is a pyrophoric gas. Commercially available
References
- ^ a b c Haynes, p. 4.52.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0183". National Institute for Occupational Safety and Health (NIOSH).
- ^ Yerazunis, S., et al. “Solubility of Diborane in the Dimethyl Ether and Diethylene Glycol, in Mixtures of Sodium Borohydride and Dimethyl Ether of Diethylene Glycol, and in Ditertiary Butyl Sulfide.” Journal of Chemical & Engineering Data, vol. 7, no. 3, July 1962, pp. 337–39, doi:10.1021/je60014a004.
- ^ a b c Haynes, p. 5.6.
- ^ a b "Diborane". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "DIBORANE – CAMEO Chemicals - Chemical Datasheet - Database of Hazardous Materials – NOAA". Retrieved 2022-10-26.
- ISBN 9780841203907.)
{{cite book}}: CS1 maint: multiple names: authors list (link - PMID 16838949.
- PMID 10941018.
- S2CID 45856199.
- ISBN 978-0121266011.
- ISBN 9780471238966
- ISBN 9780470132425.
- .
- ^ S2CID 250909492.
- ^ ISBN 978-0-13-175553-6.
- ISBN 0-8014-0412-6.
- ISBN 9780470132463.
- .
- ^ Stock, A. (1933). The Hydrides of Boron and Silicon. New York: Cornell University Press.
- .
- .
- .
- .
- .
- .
- .
- ^ "The Nobel Prize in Chemistry 1976". Nobelprize.org. Retrieved 2012-02-01.
- . Retrieved 15 July 2020.
- ^ Murrell, J. N.; Kettle, S. F. A.; Tedder, J. M. (1965). Valence theory. John Wiley and Sons. p. 243.
- ^ Lipscomb, William N. (11 December 1976). "The Boranes and their relatives (Nobel lecture)" (PDF). nobelprize.org. Nobel Foundation. pp. 224–245. Retrieved 16 July 2020.
One of the simple consequences of these studies was that electron deficient molecules, defined as having more valence orbitals than electrons, are not really electron deficient.
- ISBN 0130-39913-2.
An electron-deficient species possesses fewer valence electrons than are required for a localized bonding scheme.
- ^ Bilstein, Roger. "Stages to Saturn". chapter 5: NASA Public Affairs Office. p. 133. Archived from the original on 25 December 2017. Retrieved 14 November 2015.
{{cite web}}: CS1 maint: location (link) - ^ Gammon, Benson E.; Genco, Russell S.; Gerstein, Melvin (1950). A preliminary experimental and analytical evaluation of diborane as a ram-jet fuel (PDF). National Advisory Committee for Aeronautics.
- ^ Tower, Leonard K.; Breitwieser, Roland; Gammon, Benson E. (1958). Theoretical Combustion Performance of Several High-Energy Fuels for Ramjet Engines (PDF). National Advisory Committee for Aeronautics.
- ^ "LIQUID HYDROGEN AS A PROPULSION FUEL, 1945–1959. Part II: 1950–1957. Chapter 5. NACA Research on High-Energy Propellants". history.nasa.gov.
- ISBN 978-3527306732.
- .
- PMID 8658516.
Cited sources
- Haynes, William M., ed. (2011). ISBN 978-1439855119.
- Yerazunis, S., et al. “Solubility of Diborane in the Dimethyl Ether and Diethylene Glycol, in Mixtures of Sodium Borohydride and Dimethyl Ether of Diethylene Glycol, and in Ditertiary Butyl Sulfide.” Journal of Chemical & Engineering Data, vol. 7, no. 3, July 1962, pp. 337–39, doi:10.1021/je60014a004.

