Diborane(4)
| |
| Names | |
|---|---|
| IUPAC name
Diborane(4)
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| 24760 | |
PubChem CID
|
|
| |
| |
| Properties | |
| B2H4 | |
| Molar mass | 25.65 g·mol−1 |
| Related compounds | |
Related compounds
|
Tetrahydroxydiborane
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
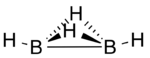
Diborane(4) is a transient inorganic compound with the chemical formula B
2H
4. Stable derivatives are known.
Diborane(4) has been produced by abstraction of two hydrogen atoms from diborane(6) using atomic fluorine and detected by photoionization mass spectrometry.[1] Computational studies predict a structure in which are two hydrogen atoms bridging the two boron atoms via three-centre two-electron bonds in addition to the 2-centre, 2-electron bond between the two boron atoms and one terminal hydrogen atom bonded to each boron atom.[2]
Several stable derivatives of diborane(4) have been reported.[3][4][5]
References
- doi:10.1063/1.456745.
- PMID 21698334.
- PMID 22187045.
- PMID 22526934.
- PMID 29345706.
