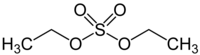
Diethyl sulfate
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Diethyl sulfate | |
| Other names
Sulfuric acid diethyl ester
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.000.536 |
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O4S | |
| Molar mass | 154.18 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.2 g/mL |
| Melting point | −25 °C (−13 °F; 248 K) |
| Boiling point | 209 °C (408 °F; 482 K) (decomposes) |
| decomposes in water | |
| Vapor pressure | 0.29 mm Hg |
| -86.8·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H312, H314, H332, H340, H350 | |
| P201, P202, P260, P261, P264, P270, P271, P280, P281, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P308+P313, P310, P312, P321, P322, P330, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 104 °C (219 °F; 377 K) |
| Related compounds | |
Related compounds
|
Dimethyl sulfate; diethyl sulfite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethyl sulfate (DES) is a highly
Property
Diethyl sulfate is moisture sensitive liquid. Heating can lead to release of toxic gases and vapors.[3] It gets darker over time.[2] It forms ethyl alcohol, ethyl sulfate, and eventually sulfuric acid when exposed to water. This compound is also combustible; when burned, sulfur oxides, ether, and ethylene are produced.[1]
Toxicity
Diethyl sulfate is a strong
Inhalation of this chemical compound has potential to be fatal and can induce nausea or vomiting. Swallowing this substance could also be fatal or lead to nausea, vomiting, or severe abdominal pain. Contact with or absorption through the skin also has potential to be fatal, and can cause severe burns.[8]
Preparation
Diethyl sulfate can be prepared by the reaction of oleum with diethyl ether or ethanol, after which the product is purified by vacuum distillation.[9] Another method involves dissolving ethylene in concentrated sulfuric acid.[9] Diethyl sulfate is produced commercially at scale and is sold by a variety of chemical suppliers.[10]
References
- ^ a b c "DIETHYL SULFATE | CAMEO Chemicals | NOAA". cameochemicals.noaa.gov. Retrieved 2021-03-04.
- ^ a b c "NCI Thesaurus". ncit.nci.nih.gov. Retrieved 2021-04-02.
- ^ "SAFETY DATA SHEET--Diethyl sulfate". ThermoFisher Scientific. Aug 21, 2018. Retrieved June 18, 2020.
- ^ "Agents Classified by the IARC Monographs, Volumes 1–129 – IARC Monographs on the Identification of Carcinogenic Hazards to Humans". monographs.iarc.who.int. Retrieved 2021-04-02.
- ^ IARC (1999). "Diethyl Sulfate". Summaries and Evaluations. 71. International Agency for Research on Cancer (IARC): 1405.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "NCI Thesaurus". ncit.nci.nih.gov. Retrieved 2021-02-18.
- ^ "Diethyl Sulfate" (PDF). United States Environmental Protection Agency. Archived (PDF) from the original on 2016-10-14.
- ^ "DIETHYL SULFATE | CAMEO Chemicals | NOAA". cameochemicals.noaa.gov. Retrieved 2021-04-02.
- ^ a b Dow (June 24, 2006). "Diethyl Sulfate" (PDF). Product Safety Assessment. Dow Chemical Company. Retrieved 2016-03-05.
{{cite journal}}: Cite journal requires|journal=(help) - ^ PubChem. "Hazardous Substances Data Bank (HSDB) : 1636". pubchem.ncbi.nlm.nih.gov. Retrieved 2021-03-18.
Further reading
- Buck, J. R.; Park, M.; Wang, Z.; Prudhomme, D. R.; Rizzo, C. J. (2000). "9-Ethyl-3,6-Dimethylcarbazole (DMECZ)". Organic Syntheses. 77: 153; Collected Volumes, vol. 10, p. 396.
- Theodore, S.; Sai, P. S. T. (2001). "Esterification of Ethanol with Sulfuric Acid: A Kinetic Study". Canadian Journal of Chemical Engineering. 79 (1): 54–64. .
External links
- "Diethyl sulfate". Webbook. NIST.
- "DIETHYL SULFATE -- ICSC: 0570". Inchem.
- "Diethyl sulfate" (PDF). IARC Monographs. 71. IARC. 1992.
{{cite journal}}: Cite journal requires|journal=(help)

