Dihalomethane
The dihalomethanes are organic compounds in which two hydrogen atoms in
halomethanes
, and contains ten members.
There are four members with only one kind of halogen atom: difluoromethane, dichloromethane, dibromomethane and diiodomethane.
There are six members with two kinds of halogen atoms:
- Bromochloromethane
- Bromofluoromethane
- Bromoiodomethane
- Chlorofluoromethane
- Chloroiodomethane
- Fluoroiodomethane
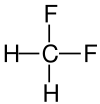
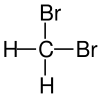
Structural Formula
|
|

|
|

|
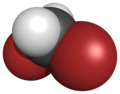
| Name | Difluoromethane | Dichloromethane | Dibromoethane | Diiodomethane |
| Melting point | −136 °C[1]
|
−97 °C[2] | −52 °C[3] | 6 °C[4] |
| Boiling point | −51,7 °C[1] | 40 °C[2] | 97 °C[3] | Zersetzung[4] |
| Space-filling model | 
|

|
|

|
Reference
- ^ a b Record of Difluormethan in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2020-02-29.
- ^ a b Record of Dichlormethan in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2020-02-29.
- ^ a b Record of Dibrommethan in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2020-02-29.
- ^ a b Record of Methyleniodid in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2020-02-29.
See also
Wikimedia Commons has media related to Dihalomethanes.
