Dihedral angle

| Types of angles |
|---|
| 2D angles |
| Spherical |
| 2D angle pairs |
|
Adjacent Vertical Complementary Supplementary Transversal |
| 3D angles |
| Solid |
A dihedral angle is the
Mathematical background
When the two intersecting planes are described in terms of
the dihedral angle, between them is given by:
and satisfies It can easily be observed that the angle is independent of and .
Alternatively, if nA and nB are
where nA · nB is the dot product of the vectors and |nA| |nB| is the product of their lengths.[1]
The absolute value is required in above formulas, as the planes are not changed when changing all coefficient signs in one equation, or replacing one normal vector by its opposite.
However the
- ,
and satisfies In this case, switching the two half-planes gives the same result, and so does replacing with In chemistry (see below), we define a dihedral angle such that replacing with changes the sign of the angle, which can be between −π and π.
In polymer physics
In some scientific areas such as polymer physics, one may consider a chain of points and links between consecutive points. If the points are sequentially numbered and located at positions r1, r2, r3, etc. then bond vectors are defined by u1=r2−r1, u2=r3−r2, and ui=ri+1−ri, more generally.[2] This is the case for kinematic chains or amino acids in a protein structure. In these cases, one is often interested in the half-planes defined by three consecutive points, and the dihedral angle between two consecutive such half-planes. If u1, u2 and u3 are three consecutive bond vectors, the intersection of the half-planes is oriented, which allows defining a dihedral angle that belongs to the interval (−π, π]. This dihedral angle is defined by[3]
or, using the function atan2,
This dihedral angle does not depend on the orientation of the chain (order in which the point are considered) — reversing this ordering consists of replacing each vector by its opposite vector, and exchanging the indices 1 and 3. Both operations do not change the cosine, but change the sign of the sine. Thus, together, they do not change the angle.
A simpler formula for the same dihedral angle is the following (the proof is given below)
or equivalently,
This can be deduced from previous formulas by using the
Given the definition of the cross product, this means that is the angle in the clockwise direction of the fourth atom compared to the first atom, while looking down the axis from the second atom to the third. Special cases (one may say the usual cases) are , and , which are called the trans, gauche+, and gauche− conformations.
In stereochemistry
|

|

|
| Configuration names according to dihedral angle |
syn n-Butane in the gauche− conformation (−60°) Newman projection |
syn n-Butane sawhorse projection |

In
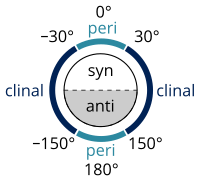
The two types of terms can be combined so as to define four ranges of angle; 0° to ±30° synperiplanar (sp); 30° to 90° and −30° to −90° synclinal (sc); 90° to 150° and −90° to −150° anticlinal (ac); ±150° to 180° antiperiplanar (ap). The synperiplanar conformation is also known as the syn- or cis-conformation; antiperiplanar as anti or trans; and synclinal as gauche or skew.
For example, with n-butane two planes can be specified in terms of the two central carbon atoms and either of the methyl carbon atoms. The syn-conformation shown above, with a dihedral angle of 60° is less stable than the anti-conformation with a dihedral angle of 180°.
For macromolecular usage the symbols T, C, G+, G−, A+ and A− are recommended (ap, sp, +sc, −sc, +ac and −ac respectively).
Proteins

A Ramachandran plot (also known as a Ramachandran diagram or a [φ,ψ] plot), originally developed in 1963 by G. N. Ramachandran, C. Ramakrishnan, and V. Sasisekharan,[7] is a way to visualize energetically allowed regions for backbone dihedral angles ψ against φ of amino acid residues in protein structure. In a protein chain three dihedral angles are defined:
- ω (omega) is the angle in the chain Cα − C' − N − Cα,
- φ (phi) is the angle in the chain C' − N − Cα − C'
- ψ (psi) is the angle in the chain N − Cα − C' − N (called φ′ by Ramachandran)
The figure at right illustrates the location of each of these angles (but it does not show correctly the way they are defined).[8]
The planarity of the
The side chain dihedral angles are designated with χn (chi-n).[10] They tend to cluster near 180°, 60°, and −60°, which are called the trans, gauche−, and gauche+ conformations. The stability of certain sidechain dihedral angles is affected by the values φ and ψ.[11] For instance, there are direct steric interactions between the Cγ of the side chain in the gauche+ rotamer and the backbone nitrogen of the next residue when ψ is near -60°.[12] This is evident from statistical distributions in backbone-dependent rotamer libraries.
Converting from dihedral angles to Cartesian coordinates in chains
It is common to represent polymers backbones, notably proteins, in
Geometry
Every polyhedron has a dihedral angle at every edge describing the relationship of the two faces that share that edge. This dihedral angle, also called the face angle, is measured as the
Every dihedral angle in an
Law of cosines for dihedral angle
Given 3 faces of a polyhedron which meet at a common vertex P and have edges AP, BP and CP, the cosine of the dihedral angle between the faces containing APC and BPC is:[14]
This can be deduced from Spherical law of cosines
See also
References
- ^ "Angle Between Two Planes". TutorVista.com. Archived from the original on 2020-10-28. Retrieved 2018-07-06.
- ISBN 3540262105.
- .
- ISBN 978-1891389313.
- PMID 13990617.
- PMID 7020376.
- S2CID 52031431.
- ^ "Side Chain Conformation".
- PMID 8464064.
- S2CID 9157373.
- S2CID 2279574
- ^ "dihedral angle calculator polyhedron". www.had2know.com. Archived from the original on 25 November 2015. Retrieved 25 October 2015.
External links
- The Dihedral Angle in Woodworking at Tips.FM
- Analysis of the 5 Regular Polyhedra gives a step-by-step derivation of these exact values.


















![{\displaystyle (\mathbf {u} _{1}\times \mathbf {u} _{2})\times (\mathbf {u} _{2}\times \mathbf {u} _{3})=[(\mathbf {u} _{2}\times \mathbf {u} _{3})\cdot \mathbf {u} _{1}]\mathbf {u} _{2}-[(\mathbf {u} _{2}\times \mathbf {u} _{3})\cdot \mathbf {u} _{2}]\mathbf {u} _{1}=[(\mathbf {u} _{2}\times \mathbf {u} _{3})\cdot \mathbf {u} _{1}]\mathbf {u} _{2}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/78dfd079ef2f25bdd1f87bcbd9d208c2af4ca75b)



