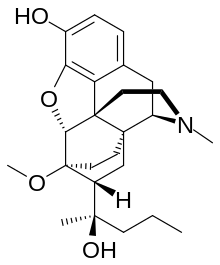
Dihydroetorphine
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Dihydroetorphine was developed by K. W. Bentley at McFarlan-Smith in the 1960s[1] and is a potent opioid analgesic[2] used mainly in China. It is a derivative of the better-known opioid etorphine, a very potent veterinary painkiller and anesthetic medication used primarily for the sedation of large animals such as elephants, giraffes, and rhinos.
Dihydroetorphine is a semi-
Dihydroetorphine is considered to be somewhat less addictive than many other opioids, and it is also sometimes used in China as a substitute maintenance drug for opioid addicts,[5] in a similar fashion to how the related drug buprenorphine is used in western nations.[6][7] It is presumably controlled as an "ester, ether, [or] salt" of etorphine in the United States under the Controlled Substances Act 1970, and/or its pieces of the morphine carbon skeleton put it under the "morphine rule" thereof and/or the 1986 analogues act; it does not have its own ACSCN.[8] Regulation elsewhere may vary but would likely be similar to that for other strong opioid agonists.[citation needed]
Dihydroetorphine is illegal in Italy, as are its parent compounds etorphine and acetorphine.[9]
See also
References
- ^ PMID 6042764.
- PMID 9788770.
- PMID 15032698.
- PMID 12481194.
- S2CID 201878290. Archived from the originalon 2013-05-25. Retrieved 2017-09-09.
- S2CID 23944183.
- PMID 12570846.
- ^ "DEA Diversion Control Division". Archived from the original on 2016-03-02. Retrieved 2016-02-27.
- ^ Salute, Ministero della. "Tabelle delle sostanze stupefacenti e psicotrope". www.salute.gov.it (in Italian). Retrieved 2022-04-09.
External links
- "List of drugs in Schedule II of the Controlled Substances Act". DEA. Archived from the original on 2015-08-04. Retrieved 2007-08-24.
