Dipeptidyl peptidase-4 inhibitor
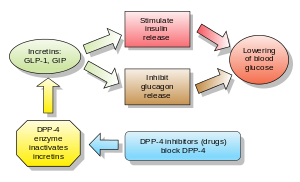
Inhibitors of dipeptidyl peptidase 4 (DPP-4 inhibitors or gliptins) are a class of
The first agent of the class –
A 2018 meta-analysis found no favorable effect of DPP-4 inhibitors on all-cause mortality, cardiovascular mortality, myocardial infarction or stroke in patients with type 2 diabetes.[5]
Examples
Drugs belonging to this class are:
- Januvia)
- Vildagliptin[7] (EU approved 2007, marketed in the EU by Novartis as Galvus)
- Onglyza)
- Linagliptin (FDA approved in 2011, marketed as Tradjenta by Eli Lilly and Company and Boehringer Ingelheim)[8]
- Gemigliptin (approved in Korea in 2012, marketed by LG Life Sciences)[9] Marketed as Zemiglo
- Anagliptin (approved in Japan as Suiny in 2012, marketed by Sanwa Kagaku Kenkyusho Co., Ltd. and Kowa Company, Ltd.)[10]
- Teneligliptin (approved in Japan as Tenelia in 2012[11])
- Alogliptin (FDA approved 2013 as Nesina/ Vipidia, marketed by Takeda Pharmaceutical Company)
- Trelagliptin (approved for use in Japan as Zafatek/ Wedica in 2015)
- Omarigliptin (MK-3102) (approved as Marizev in Japan in 2015,[12] developed by Merck & Co.; research showed that omarigliptin can be used as once-weekly treatment and generally well tolerated throughout the base and extension studies[13])
- Evogliptin (approved as Suganon/ Evodine for use in South Korea[14])
- Gosogliptin (approved as Saterex for use in Russia[15])
- Dutogliptin (PHX- 1149 free base, being developed by Phenomix Corporation), Phase III[16]
- Neogliptin[17]
- Retagliptin (SP-2086), approved in China.
- Denagliptin
- Cofrogliptin (HSK- 7653, compound 2)
- Fotagliptin
- Prusogliptin
Other chemicals which may inhibit DPP-4 include:
- Berberine, an alkaloid found in plants of the genus Berberis, inhibits dipeptidyl peptidase-4 which may at least partly explains its antihyperglycemic activity.[18]
Adverse effects
In those already taking
Adverse effects include nasopharyngitis, headache, nausea, heart failure, hypersensitivity and skin reactions.
The U.S. Food and Drug Administration (FDA) is warning that the type 2 diabetes medicines like sitagliptin, saxagliptin, linagliptin, and alogliptin may cause joint pain that can be severe and disabling. FDA has added a new Warning and Precaution about this risk to the labels of all medicines in this drug class, called dipeptidyl peptidase-4 (DPP-4) inhibitors.[20] However, studies assessing risk of rheumatoid arthritis among DPP-4 inhibitor users have been inconclusive.[21]
A 2014 review found increased risk of heart failure with saxagliptin and alogliptin, prompting the FDA in 2016 to add warnings to the relevant drug labels.[22]
A 2018 meta analysis showed that use of DPP-4 inhibitors was associated with a 58% increased risk of developing acute pancreatitis compared with placebo or no treatment.[23]
A 2018 observational study suggested an elevated risk of developing inflammatory bowel disease (specifically, ulcerative colitis), reaching a peak after three to four years of use and decreasing after more than four years of use.[24]
A 2020 Cochrane systematic review did not find enough evidence of reduction of all-cause mortality, serious adverse events, cardiovascular mortality, non-fatal myocardial infarction, non-fatal stroke or end-stage renal disease when comparing metformin monotherapy to dipeptidyl peptidase-4 inhibitors for the treatment of type 2 diabetes.[25]
Cancer
In response to a report of precancerous changes in the pancreases of rats and organ donors treated with the DPP-4 inhibitor sitagliptin,[26][27] the United States FDA and the European Medicines Agency each undertook independent reviews of all clinical and preclinical data related to the possible association of DPP-4 inhibitors with pancreatic cancer. In a joint letter to the New England Journal of Medicine, the agencies stated that they had not yet reached a final conclusion regarding a possible causative relationship.[28]
A 2014 meta-analysis found no evidence for increased pancreatic cancer risk in people treated with DPP-4 inhibitors, but owing to the modest amount of data available, was not able to completely exclude possible risk.[29]
Combination drugs
Some DPP-4 inhibitor drugs have received approval from the FDA to be used with
See also
- Development of dipeptidyl peptidase-4 inhibitors
References
- ^ "FDA Approves New Treatment for Diabetes" (Press release). U.S. Food and Drug Administration. October 17, 2006. Retrieved 2006-10-17.
- S2CID 9151210.
- PMID 12697069.
- PMID 7789625.
- PMID 29677303.
- ^ Banting and Best Diabetes Centre at UT sitagliptin
- ^ Banting and Best Diabetes Centre at UT vildagliptin
- ^ "FDA approves new treatment for Type 2 diabetes". Fda.gov. 2011-05-02. Retrieved 2013-04-15.
- ^ "LG Life Science". Lgls.com. Archived from the original on 2013-09-06. Retrieved 2013-04-15.
- ^ "New Drugs Approved in FY 2012" (PDF). Archived from the original (PDF) on 2013-07-18. Retrieved 2013-08-07.
- )
- ^ "Merck MARIZEV Once-Weekly DPP-4 Inhibitor For Type2 Diabetes Approved In Japan". NASDAQ. 28 September 2015. Retrieved 29 September 2015.
- PMID 26310692.
- ^ "Dong-A ST's DPP4 inhibitor, SUGANON, got approved for type 2 diabetes in Korea". pipelinereview.com. October 2, 2015.
- ^ "SatRx LLC Announces First Registration in Russia of SatRx (gosogliptin), an Innovative Drug for Treatment of Type 2 Diabetes" (Press release). SatRx LLC.
- ^ "Forest Splits With Phenomix", San Diego Business Journal, Tuesday, April 20, 2010 http://www.sdbj.com/news/2010/apr/20/forest-splits-phenomix/
- PMID 35337071.
- S2CID 25517996.
- PMID 27142267.
- ^ "DPP-4 Inhibitors for Type 2 Diabetes: Drug Safety Communication - May Cause Severe Joint Pain". FDA. 2015-08-28. Retrieved 1 September 2015.
- PMID 29236221.
- ^ "Diabetes Meds Containing Saxagliptin and Alogliptin Linked to Increased HF". Pharmacy Practice News. April 2016.
- PMID 29677303.
- PMID 29563098.
- PMID 32501595.
- PMID 19403868.
- PMID 23524641.
- PMID 24571751.
- S2CID 7642027.
