Diphenoxylate
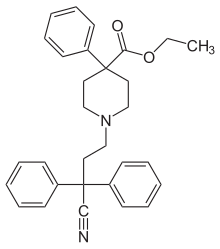 | |
 | |
| Clinical data | |
|---|---|
| Other names | R-1132, NIH-756 |
| AHFS/Drugs.com | Monograph |
| Routes of administration | Oral |
| Drug class | Opioid Antidiarrheal |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 74–95% |
| Elimination half-life | 12–14 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Diphenoxylate is a centrally active
Medical use
Diphenoxylate is used to treat diarrhea in adults; it is only available as a combination drug with a subtherapeutic dose of atropine to prevent abuse.[2]
It should not be used in children due to the risk of
It should not be taken with other central depressants like alcohol, as they can increase its risks.[2]
It should not be used for people with diarrhea caused by an infection, for example with
Adverse effects
The drug label (in some jurisdictions) has warnings with regard to the risk of
Other adverse effects include numbness in the hands and feet, euphoria, depression, lethargy, confusion, drowsiness, dizziness, restlessness, headache, hallucinations, edema, hives, swollen gums, itchiness, vomiting, nausea, loss of appetite, and stomach pain.[2]
Pharmacology
Diphenoxylate is rapidly metabolized to difenoxin; it is eliminated mostly in feces but also in urine.[2]
Like other
History and chemistry
Diphenoxylate was first synthesized by
Diphenoxylate is made by combining a precursor of normethadone with norpethidine. Loperamide (Imodium) and bezitramide are analogs. [5] Like loperamide, it has a methadone-like structure and a piperidine moiety.[6]
Society and culture
Pricing
In 2017 Hikma Pharmaceuticals raised the price of its liquid formulation of generic diphenoxylate-atropine in the US by 430%, from $16 to $84.00.[7]
Regulation
In the United States, drugs containing diphenoxylate combined with atropine salts are classified as Schedule V controlled substances.[8][2] (Diphenoxlate by itself is a Schedule II controlled substance.)
It is on Schedule III of the Single Convention on Narcotic Drugs, only in forms that contain, according to the Yellow List: "not more than 2.5 milligrams of diphenoxylate calculated as base and a quantity of atropine sulfate equivalent to at least 1 per cent of the dose of diphenoxylate".[9]
Research
Diphenoxylate and atropine have been studied in small trials as a treatment for fecal incontinence; it appears to be less efficacious and have more adverse effects when compared with loperamide or codeine.[10]
References
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d e f g h i "US label: Diphenoxylate hydrochloride and atropine sulfate tablets" (PDF). FDA. 12 February 2018. For label updates see FDA index page for NDA 012462
- PMID 14702928.
- ISBN 9780080861142.
- ISBN 9781489905857.
- ISBN 9780199697397.
- ^ Crow D (20 August 2017). "Hikma hikes price of US medicines by up to 430%". Financial Times. Archived from the original on 11 December 2022. Retrieved 10 May 2018.
- ^ "Diphenoxylate". MedlinePlus. 15 April 2018. Retrieved 10 May 2018.
- ^ "Yellow List: List of Narcotic Drugs Under International Control, 50th Edition" (PDF). International Narcotics Control Board. 2011. p. 8. Retrieved 10 May 2018.
- PMID 23757096.

