Discovery and development of beta-blockers

Development

History
The β-blockers are an immensely important class of drugs due to their high
In the early 1960s,
In April 1963, toxicity tests for pronethalol showed results of thymic tumours in mice. Nevertheless, it was launched under the trade name Alderlin, as the first clinically useful β-blocker. The launch took place in November 1963 when many small-scale
The evolution of non-selective and selective β-blockers

By the time propranolol was launched, ICI was beginning to experience competition from other companies. This potential threat led to ongoing refinements in the pharmacologic structure of β-blockers and subsequent advances in drug delivery. ICI studied analogues further and in 1970 launched practolol (figure 4) under the trade name Eraldin. It was withdrawn from the market a few years later because of the severe side effects it caused, nevertheless it played a large role in the fundamental study of β-blockade and β-receptors.[4] The withdrawal of Eraldin gave ICI the nudge to launch another β-blocker,
The progress in β-blocker development led to the introduction of drugs with variety of properties. β-blockers were developed having a relative
Mechanism of action
Pharmacokinetics
The β-adrenergic receptor antagonists all have similar
Binding to β adrenergic receptors

Three different types of β-adrenergic receptors have been identified by molecular pharmacology.
β-blockers are
Selectivity
β-blockers can be
Synthesis


Synthesis for a standard β-blocker begins with the mono-alkylation of catechol to give an ether (see figure 4).[15]
The fundamental step, and usually the last, in the synthesis of β-blockers consists of adding a propanolamine side chain. This can be done following two paths which both involve
(S)-propranolol

Propranolol exist in two different enantiomers, (S)-(−)- and (R)-(+)-enantiomers. The (S)-
Structure-activity relationship (SAR)
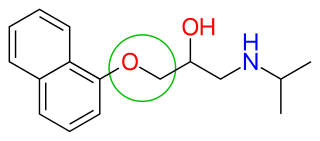
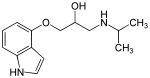
β-blockers' binding site to the receptor is the same as for
After the release of propranolol, relative lipophilicity of β-blockers as a significant factor in their varied and complex pharmacology, became an important factor. It was suspected that propranolol's centrally induced side effects could be due to its high lipophilicity. Thus, it was focused on synthesizing analogues with hydrophilic moieties, favourably placed to see if the side effects would decrease. Selecting para-acylamino groups as the hydrophilic moiety, scientists synthesized a group of para-acylphenoxyethanol and propanolamines, and selected practolol for clinical trials. Practolol had one property not previously seen with β-blockers, it exhibited cardioselectivity (β1 selectivity). Studies from practolol showed that moving the acylamino group to meta or ortho positions, on the benzene ring, caused a loss of selectivity but not loss of the β-blockade itself. This illustrated the significance of para-substitution for β1 selectivity of β-blockers.[5]

Figure 8 shows the
- The X part of the side chain can either be directly linked to the aromatic ring or linked through a —OCH2— group
- When X is —CH2CH2—, —CH=CH—, —SCH2— or —NCH2—, there is little or no activity
- The R1 group can only be a secondary substitution and branched is the optimal choice
- Alkyl (—CH3) substituents on the α, β or γ carbon (if X = —OCH2—) lower beta blockade, especially at the α carbon
The general rule for aromatic substitution is: ortho > meta > para. This gives non-selective β-blockers. Large para-substituents usually decrease activity but large ortho-groups retain some activity. Polysubstitution on carbon 2 and 6 makes the compound inactive but when the substitution is on carbon 3 and 5 there's some activity. For the highest cardioselectivity, the substituents should be as following: para > meta > ortho. All the β-blockade is in one isomer, (S)-aryloxypropylamine and (R)-ethanolamine.[5]
Clinical use
Cardiovascular indications
For decades β-blockers have been used in
Other indications
There are few diseases, other than cardiovascular diseases, that β-blockers have a clinical effect on. These diseases are mentioned in the following sub-chapters. In addition, there are diseases which β-blockers have a clinical effect but are not the first choice of treatment. They won't be mentioned in the sub-chapters.
Essential tremor
When symptoms of the essential tremors are considerably high, non-selective β-blockers are an important treatment option and usually the first choice. Studies have shown that propranolol did reduce symptoms the most in that category. The β-blockers can be used alone or in a combination.[28]
Glaucoma
Teratogenicity
Hypertension is reported to complicate one out of ten pregnancies, which makes it the most common medical disorder in pregnancy. It is important to have a correct diagnosis of hypertension during pregnancy, with the emphasis on differentiating pre-existing hypertension from pregnancy induced hypertension (gestational and the syndrome of pre-eclampsia). During pregnancy, the challenge is to determine when to use antihypertensive medications and which level of blood pressure to target.[30] A balance has to be found between the potential risk to the health of the baby related to drug-exposure and the risk to the mother and baby due to an untreated medical condition (severe hypertension).[31]
Antihypertensive drug use during pregnancy is relatively common and increasing.[32] Only a small proportion of available antihypertensive drugs have been tested in pregnant women, and many are contraindicated.[citation needed] It is important to make the exposure of medications to the baby as small as possible.[30] It is not clear if treating women who have mild or moderate hypertension during pregnancy with anti-hypertensive medication is beneficial.[33][31]
The most common
| Non-selective | FDA category | β1-selective | FDA category |
|---|---|---|---|
| Labetalol | C | Atenolol | D |
| Pindolol | B | Acebutolol | B |
| Propranolol | C | Bisoprolol | C |
| Timolol | C | Nebivolol | C |
| Sotalol | B | Metoprolol | C |
See also
References
- ISBN 0071124322.
- ^ ISBN 978-0-7817-6879-5.
- ^ a b Frishman, W.H. (December 2008). "Fifty years of beta-blockers: a revolution in CV pharmacotherapy". Cardiologytoday. Retrieved 5 November 2013.
- ^ PMID 16502872.
- ^ ISBN 0-471-18545-0.
- ^ ISBN 1-84376-050-9.
- ^ S2CID 19506044.
- ^ PMID 11466176.
- PMID 26471965.
- ^ PMID 22279967.
- PMID 23614528.
- ISBN 978-0-7817-6879-5.
- PMID 22595449.
- S2CID 24667000.
- ^ ISBN 0-471-19657-6.
- ^ .
- .
- .
- PMID 21125979.
- ^ PMID 10349897.
- PMID 24167581.
- PMID 10381708.
- ^ Bui, Quynh (2010-06-01). "First-Line Treatment for Hypertension". American Family Physician. 81 (11): 1333–1335.
- PMID 8810786.
- PMID 24072434.
- PMID 23122672.
- S2CID 35802069.
- S2CID 58374.
- S2CID 20300467.
- ^ PMID 18259046.
- ^ PMID 12917933.
- PMID 22966012.
- PMID 24504933.
- PMID 22815467.
- PMID 23753416.
- ^ "FDA Pregnancy Categories" (PDF). University of Washington. Archived from the original (PDF) on 14 December 2010. Retrieved 11 November 2013.