Cereblon E3 ligase modulator
| Cereblon E3 ligase modulator | |
|---|---|
NF-kB, etc. | |
| Clinical data | |
| Drugs.com | Drug Classes |
| Legal status | |
| In Wikidata | |
Cereblon E3 ligase modulators, also known as immunomodulatory imide drugs (IMiDs), are a class of immunomodulatory drugs[1] (drugs that adjust immune responses) containing an imide group. The IMiD class includes thalidomide and its analogues (lenalidomide, pomalidomide, mezigdomide and iberdomide).[1] These drugs may also be referred to as 'Cereblon modulators'. Cereblon (CRBN) is the protein targeted by this class of drugs.
The name "IMiD" alludes to both "IMD" for "immunomodulatory drug" and the forms imide, imido-, imid-, and imid.
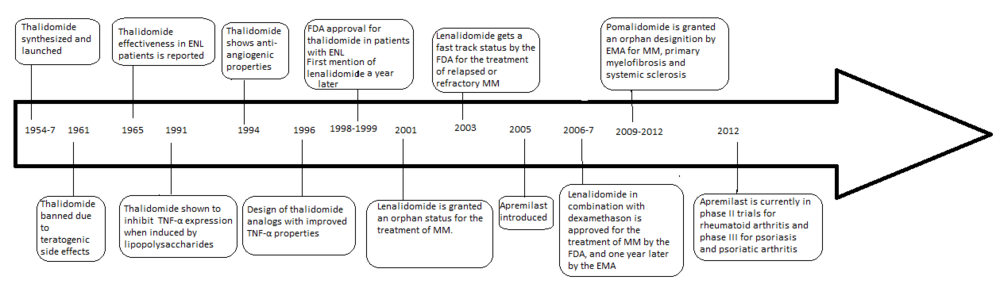
The development of analogs of thalidomide was precipitated by the discovery of the anti-angiogenic and anti-inflammatory properties of the drug yielding a new way of fighting cancer as well as some inflammatory diseases after it had been banned in 1961. The problems with thalidomide included teratogenic side effects, high incidence of other adverse reactions, poor solubility in water and poor absorption from the intestines.
In 1998 thalidomide was approved by the U.S.
History
Thalidomide was originally released in the Federal Republic of Germany (West Germany) under the label of Contergan on October 1, 1957, by Chemie Grünenthal (now
Four years after thalidomide was withdrawn from the market for its ability to induce severe birth defects, its anti-inflammatory properties were discovered when patients with
Lenalidomide is the first analog of thalidomide which is marketed. It is considerably more potent than its parent drug with only two differences at a molecular level, with an added
Pomalidomide (3-aminothalidomide) was the second thalidomide analog to enter the clinic being more potent than both of its predecessors.[12] First reported in 2001, pomalidomide was noted to directly inhibit myeloma cell proliferation and thus inhibiting MM both on the tumor and vascular compartments.[13] This dual activity of pomalidomide makes it more efficacious than thalidomide both in vitro and in vivo.[14] This effect is not related to TNF-α inhibition since potent TNF-α inhibitors such as rolipram and pentoxifylline did not inhibit myeloma cell growth nor angiogenesis.[9] Upregulation of interferon gamma, IL-2 and IL-10 have been reported for pomalidomide and may contribute to its anti-angiogenic and anti-myeloma activities.
Development
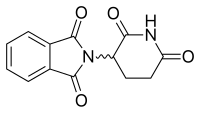
The thalidomide molecule is a synthetic derivative of glutamic acid and consists of a glutarimide ring and a phthaloyl ring (Figure 5).[15][16] Its IUPAC name is 2-(2,6-dioxopiperidin-3-yl)isoindole-1,3-dione and it has one chiral center[15] After thalidomide's selective inhibition of TNF-α had been reported, a renewed effort was put in thalidomide's clinical development. The clinical development led to the discovery of new analogs which strived to have improved activities and decreased side effects.[8][17]
Clinically, thalidomide has always been used as a racemate. Generally the S-isomer is associated with the infamous teratogenic effects of thalidomide and the R-isomer is devoid of the teratogenic properties but conveys the sedative effects,[8] however this view is highly debated and it has been argued that the animal model that these different R- and S-effects were seen in was not sensitive to the thalidomide teratogenic effects. Later reports in rabbits, which is a sensitive species, unveiled teratogenic effects from both isomers.[8][15][16][17] Moreover, thalidomide enantiomers have been shown to be interconversed in vivo due to the acidic chiral hydrogen in the asymmetric center (shown, for the EM-12 analog, in Figure 3),[16][17] so the plan to administer a purified single enantiomer to avoid the teratogenic effects will most likely be in vain.[8][15][16]
Development of lenalidomide and pomalidomide

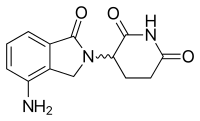
One of the analogs of interest was made by isoindolinone replacement of the phthaloyl ring. It was given the name EM-12 (Figure 3). This replacement was thought to increase the bioavailability of the substance because of increased stability. The molecule had been reported to be an even more potent teratogenic agent than thalidomide in rats, rabbits and monkeys. Additionally, these analogs are more potent inhibitors of angiogenesis than thalidomide.[13] As well, the amino-thalidomide and amino-EM-12 were potent inhibitors of TNF-α.[16] These two analogs later got the name lenalidomide, which is the EM-12 amino analog, and pomalidomide, the thalidomide amino analog.[8]
Medical use
The primary use of IMiDs in medicine is in the treatment of cancers and autoimmune diseases (including one that is a response to the infection leprosy).[18] Indications for these agents that have received regulatory approval include:[19]
- acute myeloid leukaemia
- Erythema nodosum, a complication of leprosy
- Multiple myeloma
Off-label indications for which they seem promising treatments include:[20]
- Hodgkin's lymphoma
- Light chain-associated (AL) amyloidosis
- Primary myelofibrosis(PMF)
- Acute myeloid leukaemia(AML)
- Prostate cancer
- Metastatic renal cell carcinoma (mRCC)
Thalidomide
Thalidomide has been approved by the FDA for ENL and MM in combination with
Lenalidomide
Lenalidomide is approved in nearly 70 countries, in combination with dexamethasone for the treatment of patients with MM who have received at least one prior therapy. Orphan indications include
Pomalidomide
Pomalidomide was submitted for FDA approval on April 26, 2012
Adverse effects
The major toxicities of approved IMiDs are
Teratogenicity
Thalidomide's teratogenicity has been a subject of much debate and over the years numerous hypotheses have been proposed. Two of the best-known have been the anti-angiogenesis hypothesis and oxidative stress model hypothesis, with considerable experimental evidence supporting these two hypotheses regarding thalidomide's teratogenicity.[29]
Recently, new findings have emerged that suggest a novel mechanism of teratogenicity.
Findings also support the hypothesis that an increase in the expression of cereblon is an essential element of the anti-myeloma effect of both lenalidomide and pomalidomide.[29] Cereblon expression was three times higher in responding patients compared to non-responders and higher cereblon expression was also associated with partial or full response while lower expression was associated with stable or progressive disease.[30]
Mechanism of action
Their mechanism of action is not entirely clear, but it is known that they inhibit the production of
Thalidomide and its analogs, lenalidomide and pomalidomide, are believed to act in a similar fashion even though their exact mechanism of action is not yet fully understood. It is believed that they work through different mechanisms in various diseases. The net effect is probably due to different mechanisms combined. Mechanism of action will be explained in light of today's knowledge.
Thalidomide, lenalidomide and pomalidomide
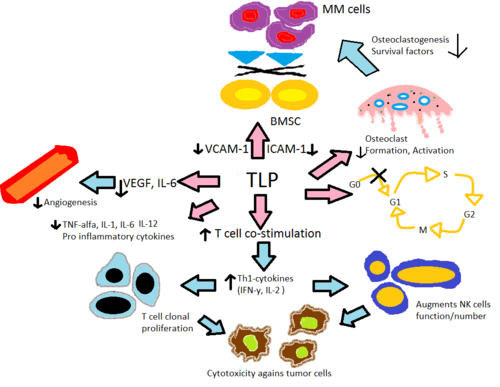
Altering cytokine production
Thalidomide and its immune-modulating analogs alter the production of the inflammatory cytokines TNF-α,
Lenalidomide is believed to be about 1000 times more potent in vitro than thalidomide in anti-inflammatory properties and pomalidomide about 10 times more potent than lenalidomide. It is worth noticing however that, when comparing lenalidomide and pomalidomide, clinical relevance of higher in vitro potency is unclear since
T-cell activation
Thalidomide and its analogs help with the co-stimulation of T-cells through the
Anti-angiogenesis
Angiogenesis or the growth of new blood vessels has been reported to correspond with MM progression where
Anti-tumor activity
In vivo anti-tumor activity of thalidomide is believed to be due to the potent anti-angiogenic effect and also through changes in cytokine expression. In vitro assays on
Bone marrow environment
The role of angiogenesis in the support of myeloma was first discovered by Vacca in 1994.[40] They discovered increased bone marrow angiogenesis correlates with myeloma growth and supporting stromal cells are a significant source for angiogenic molecules in myeloma. This is believed to be a main component of the mechanism in vivo by which thalidomide inhibits multiple myeloma.
Additionally, inflammatory responses within the bone marrow are believed to foster many hematological diseases. The secretion of IL-6 by
Structure-activity relationship

Since the mechanism of action of thalidomide and its analogs is not fully clear and the bioreceptor for these substances has not been identified, the insight into the relationship between the structure and activity of thalidomide and its analogs are mostly derived from molecular modelling and continued research investigation.[17][41] The information on SAR of thalidomide and its analogs is still in process so any trends detailed here are observed during individual studies. Research has mainly focused on improving the TNF-α and PDE4 inhibition of thalidomide,[8][15] as well as the anti-angiogenesis activity.[42][43]
TNF-α inhibitors (not via PDE4)
Research indicated that a substitution at the phthaloyl ring would increase TNF-α inhibition activity (Figure 5). An amino group substitution was tested at various locations on the phthaloyl ring (C4, C5, C6, C7) of thalidomide and EM-12 (previously described). Amino addition at the C4 location on both thalidomide and EM-12 resulted in much more potent inhibition of TNF-α. This also revealed that the amino group needed to be directly opposite the carbonyl group on the isoindolinone ring system for the most potent activity.
PDE4 inhibitors


The common structure for analogs that inhibit TNF-α via inhibition of PDE4 is prepared on the basis of hydrolysing the glutarimide ring of thalidomide. These analogs do not have an acidic chiral hydrogen, unlike thalidomide, and would therefore be expected to be chirally stable.[16]
On the phenyl ring, a 3,4-dialkoxyphenyl moiety (Figure 6) is a known pharmacophore in PDE4 inhibitors such as rolipram. Optimal activity is achieved with a methoxy group at the 4-position (X2) and a bigger group, such as cyclopentoxy at the 3-position carbon (X3). However the thalidomide PDE4 inhibitory analogs do not follow the SAR of rolipram analogs directly. For thalidomide analogs, an ethoxy group at X3 and a methoxy group at X2, with X1 being just a hydrogen, gave the highest PDE4 and TNF-α inhibition.[15] Substitutes larger than diethoxy at the X2–X3 position had decreased activity. The effects of these substitutions seem to be mediated by steric effects.[16]
For the Y-position, a number of groups have been explored. Substituted amides that were larger than methylamide (CONHCH3) decrease PDE4 inhibition activity.[16] Using a carboxylic acid as a starting point, an amide group has similar PDE4 inhibition activity but both groups were shown to be a considerably less potent than a methyl ester group, which had about six-fold increase in PDE4 inhibitory activity. Sulfone group had similar PDE4 inhibition as the methyl ester group. The best PDE4 inhibition was observed when a nitrile group was attached, which has 32 times more PDE4 inhibitory activity than the carboxyl acid.[15] Substituents at Y leading to increasing PDE4 inhibitory activity thus followed the order:
- COOH ≤ CONH2 ≤ COOCH3 ≤ SO2CH3 < CN
Substitutions on the phthaloyl ring have been explored and it was noticed that nitro groups at the C4 or C5 location decreased activity but C4 or C5 amino substitution increased it dramatically.[16] When the substitution at the 4 (Z) location on the phthaloyl ring was examined, hydroxyl and methoxy groups seem to make the analog a less potent PDE4 inhibitor. An increase in activity was observed with amino and dimethylamino to a similar extent but a methyl group improved the activity further than the aforementioned groups. A 4-N-acetylamino group had slightly lower PDE4 inhibitory activity, compared with the methyl group, but increased the compound's TNF-α inhibitory activity to a further extent.[15] Substituents at Z leading to increasing PDE4 inhibitory activity thus followed the order:
- N(CH3)2 ≤ NH2 < NHC(O)CH3 < CH3
Angiogenesis inhibition

For angiogenesis inhibition activity, an intact glutarimide ring seems to be required. Different groups were tested in the R position. The substances that had nitrogen salts as the R group showed good activity. The improved angiogenesis inhibitory activity could be due to increased solubility or that the positively charged nitrogen has added interaction with the active site. Tetrafluorination of the phthaloyl ring seems to increase the angiogenesis inhibition.[42]
Synthesis
This section may be too technical for most readers to understand. (April 2017) |
Described below are schemes for
Thalidomide


Synthesis of thalidomide has usually been performed as seen in scheme 1. This synthesis is a reasonably simplistic three step process. The downside of this process however is that the last step requires a high-temperature melt reaction which demands multiple recrystallizations and is not compliant with standard equipment.
Scheme 2 is the newer synthesis route which was designed to make the reaction more direct and to produce better yields. This route uses L-glutamine rather than L-glutamic acid as a starting material and by letting it react with N-carbethoxyphthalimide gives N-phthaloyl-L-glutamine (4), with 50–70% yield. The substance 4 is then stirred in a mixture with carbonyldiimidazole (CDI) with enough 4-dimethylaminopyridine (DMAP) in tetrahydrofuran (THF) to catalyze the reaction and heated to reflux for 15–18 hours. During the reflux thalidomide crystallizes out of the mixture. The final step gives 85–93% yield of thalidomide, bringing the total yield to 43–63%.[46]
Lenalidomide and pomalidomide

Both of the amino analogs are prepared from the condensation of 3-aminopiperidine-2,6-dione hydrochloride (Compound 3) which is synthesized in a two step reaction from commercially available

Lenalidomide is synthesized in a similar way using compound 3 (3-aminopiperidine-2,6-dione) treated with a nitro-substituted methyl 2-(bromomethyl) benzoate, and hydrogenation of the nitro group.[44]
Pharmacokinetics
Thalidomide
| Thalidomide | ||
|---|---|---|
| Tmax [drug] | 4–6 hours in subjects with MM[47] |
|
| Protein binding | 55–65%[48] | |
| Metabolites | Hydrolized metabolites[48] | |
| Half-life [t1/2] | 5.5–7.6 hours[48] | |
Lenalidomide
| Lenalidomide | ||
|---|---|---|
| Tmax [drug] | 0.6–1.5 hours in healthy subjects[49] 0.5–4 hours in subjects with MM[50] |
|
| Protein binding | ~30%[49] | |
| Metabolites | Has not yet been studied[49] | |
| Half-life [t1/2] | 3 hours in healthy subjects[49] 3.1–4.2 hours in subjects with MM[50] | |
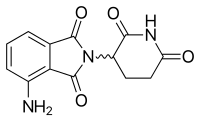
Pomalidomide
| Pomalidomide | ||
|---|---|---|
| Tmax [drug] | 0.5–8 hours[51] |
|
| Protein binding | Unknown | |
| Metabolites | Unknown | |
| Half-life [t1/2] | 6.2–7.9 hours[51] | |
See also
- Cancer
- Multiple myeloma
- Drug design
- Thalidomide
- Lenalidomide
- Pomalidomide
- Apremilast
- Organic chemistry
- Health crisis
- Immunomodulation therapy
- Immunosuppressant
- Immunomodulatory drug
References
- ^ PMID 16085014.
- PMID 17975653.
- ^ Reversal of Fortune: How a Vilified Drug Became a Life-saving Agent in the "War" Against Cancer - Onco'Zine - The International Oncology Network (November 30, 2013) Archived January 3, 2014, at archive.today
- PMID 22770990.
- S2CID 24167431.
- ^ PMID 7513432.
- PMID 10408702.
- ^ S2CID 7293027.
- ^ PMID 11740816.
- .
- S2CID 5336195.
- ^ "Vector has moved".
- ^ PMID 11740816.
- PMID 11956087.
- ^ PMID 19256507.
- ^ PMID 8765505.
- ^ PMID 14505639.
- PMID 22796518.
- PMID 22727252.
- ^ S2CID 7981368.
- ^ "Thalomid (Thalidomide) dosing, indications, interactions, adverse effects, and more". MedScape reference. Retrieved 18 September 2012.
- ^ "Thalidomide Celgene (previously Thalidomide Pharmion)". European Medicines Agency. Retrieved 18 September 2012.
- ^ "Celgene Biopharmaceutical - Investor relations - Press Releases". Archived from the original on 19 January 2013. Retrieved 18 September 2012.
- ^ "Revlimid (lenalidomide) dosing, indications, interactions, adverse effects, and more". Medscape references. Retrieved 18 September 2012.
- ^ "Search of: lenalidomide - List Results". Clinical Trials. Retrieved 18 September 2012.
- ^ "Clinical Trials Register". EU Clinical Trials Register. Retrieved 18 September 2012.
- ^ "Celgene Submits Pomalidomide For FDA Approval". The myeloma beacon.
- ^ "European Medicines Agency - Search results from your query". European Medicines Agency. Archived from the original on 5 March 2016. Retrieved 18 September 2012.
- ^ PMID 22348778.
- ^ PMID 22919394.
- PMID 19907437.
- S2CID 12782141.
- PMID 22727252.
- PMID 22003441.
- ^ .
- ^ PMID 17369076.
- ^ PMID 19907437.
- PMID 18070715.
- ^ PMID 19674465.
- PMID 7527645.
- PMID 16843662.
- ^ PMID 15084120.
- PMID 16183272.
- ^ PMID 10386948.
- PMID 17851074.
- .
- PMID 15355928.
- ^ a b c "Summary of product characteristics: Thalidomid Celgene" (PDF). European Medicines Agency. Retrieved 23 September 2012.
- ^ PMID 18452408.
- ^ PMID 12384400.
- ^ PMID 15249589.
