Drospirenone
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | Droe-SPY-re-nown |
| Trade names | Alone: Slynd With estradiol: Angeliq With ethinylestradiol: Yasmin, Yasminelle, Yaz, others With estetrol: Nextstellis |
| Other names | Dihydrospirenone; Dihydrospirorenone; 1,2-Dihydrospirorenone; MSp; SH-470; ZK-30595; LF-111; 17β-Hydroxy-6β,7β:15β,16β-dimethylene-3-oxo-17α-pregn-4-ene-21-carboxylic acid, γ-lactone |
| AHFS/Drugs.com | Professional Drug Facts |
| License data |
|
Antimineralocorticoid; Steroidal antiandrogen | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 66–85%[1][4][5] |
| Protein binding | 95–97% (to albumin)[3][1][4] |
| Metabolism | Liver (mostly CYP450-independent (reduction, sulfation, and cleavage of lactone ring), some CYP3A4 contribution)[4][6][7][8] |
| Metabolites | • Drospirenone acid[3] • 4,5-Dihydrodrospirenone 3-sulfate[3] |
| Elimination half-life | 25–33 hours[3][4][1] |
| Excretion | Urine, feces[3] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Drospirenone is a
Common
Drospirenone was patented in 1976 and introduced for medical use in 2000.[14][15] It is available widely throughout the world.[9] The medication is sometimes referred to as a "fourth-generation" progestin.[16][17] It is available as a generic medication.[18] In 2020, a formulation of drospirenone with ethinylestradiol was the 145th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[19][20]
Medical uses
Drospirenone (DRSP) is used by itself as a
Studies have found that EE/DRSP is superior to
Available forms
Drospirenone is available in the following formulations, brand names, and indications:[35][36]
- Drospirenone 4 mg (Slynd) – progestogen-only birth control pill[3]
- Drospirenone 3 mg and estetrol 14.2 mg (Nextstellis (US)) – combined birth control pill[37][38][39]
- Ethinylestradiol 30 μg and drospirenone 3 mg (Ocella, Syeda, Yasmin, Zarah, Zumandimine) – combined birth control pill[40][41][42][43]
- Ethinylestradiol 20 μg and drospirenone 3 mg (Gianvi, Jasmiel, Loryna, Lo-Zumandimine, Nikki, Vestura, Yaz) – combined birth control pill, acne, PMS, PMDD, dysmenorrhea[21]
- Estradiol 0.5 or 1 mg and drospirenone 0.25 or 0.5 mg (Angeliq) – menopausal hormone therapy (menopausal syndrome, postmenopausal osteoporosis)[23]
Contraindications
Side effects
High potassium levels
Drospirenone is an
Blood clots
Birth control pills containing
Different progestins in ethinylestradiol-containing birth control pills have been associated with different risks of VTE.
Androgenic progestins have been found to antagonize to some degree the effects of ethinylestradiol on coagulation.[59][60][61][62] As a result, more androgenic progestins, like levonorgestrel and norethisterone, may oppose the procoagulatory effects of ethinylestradiol and result in a lower increase in risk of VTE.[11][63] Conversely, this would be the case less or not at all with progestins that are less androgenic, like desogestrel and gestodene, as well as with progestins that are antiandrogenic, like drospirenone and cyproterone acetate.[11][63]
In the early 2010s, the FDA updated the label for birth control pills containing drospirenone and other progestins to include warnings for stopping use prior to and after surgery, and to warn that such birth control pills may have a higher risk of blood clots.[49]
| Type | Route | Medications | Odds ratio (95% CI) |
|---|---|---|---|
Menopausal hormone therapy |
Oral | Estradiol alone ≤1 mg/day >1 mg/day |
1.27 (1.16–1.39)* 1.22 (1.09–1.37)* 1.35 (1.18–1.55)* |
| Conjugated estrogens alone ≤0.625 mg/day >0.625 mg/day |
1.49 (1.39–1.60)* 1.40 (1.28–1.53)* 1.71 (1.51–1.93)* | ||
| Estradiol/medroxyprogesterone acetate | 1.44 (1.09–1.89)* | ||
| Estradiol/dydrogesterone ≤1 mg/day E2 >1 mg/day E2 |
1.18 (0.98–1.42) 1.12 (0.90–1.40) 1.34 (0.94–1.90) | ||
| Estradiol/norethisterone ≤1 mg/day E2 >1 mg/day E2 |
1.68 (1.57–1.80)* 1.38 (1.23–1.56)* 1.84 (1.69–2.00)* | ||
Estradiol/norgestrel or estradiol/drospirenone |
1.42 (1.00–2.03) | ||
| Conjugated estrogens/medroxyprogesterone acetate | 2.10 (1.92–2.31)* | ||
| Conjugated estrogens/norgestrel ≤0.625 mg/day CEEs >0.625 mg/day CEEs |
1.73 (1.57–1.91)* 1.53 (1.36–1.72)* 2.38 (1.99–2.85)* | ||
| Tibolone alone | 1.02 (0.90–1.15) | ||
| Raloxifene alone | 1.49 (1.24–1.79)* | ||
Transdermal |
Estradiol alone ≤50 μg/day >50 μg/day |
0.96 (0.88–1.04) 0.94 (0.85–1.03) 1.05 (0.88–1.24) | |
| Estradiol/progestogen | 0.88 (0.73–1.01) | ||
Vaginal |
Estradiol alone | 0.84 (0.73–0.97) | |
| Conjugated estrogens alone | 1.04 (0.76–1.43) | ||
Combined birth control |
Oral | Ethinylestradiol/norethisterone | 2.56 (2.15–3.06)* |
| Ethinylestradiol/levonorgestrel | 2.38 (2.18–2.59)* | ||
Ethinylestradiol/norgestimate |
2.53 (2.17–2.96)* | ||
| Ethinylestradiol/desogestrel | 4.28 (3.66–5.01)* | ||
| Ethinylestradiol/gestodene | 3.64 (3.00–4.43)* | ||
| Ethinylestradiol/drospirenone | 4.12 (3.43–4.96)* | ||
| Ethinylestradiol/cyproterone acetate | 4.27 (3.57–5.11)* | ||
| Notes: (1) Bioidentical progesterone was not included, but is known to be associated with no additional risk relative to estrogen alone. Footnotes: * = Statistically significant (p < 0.01). Sources: See template.
| |||
Breast cancer
Drospirenone has been found to stimulate the
Data on risk of breast cancer in women with newer progestins like drospirenone are lacking at present.[69] Progestogen-only birth control is not generally associated with a higher risk of breast cancer.[69] Conversely, combined birth control and menopausal hormone therapy with an estrogen and a progestogen are associated with higher risks of breast cancer.[70][69][71]
Overdose
These have been no reports of serious
Interactions
Pharmacology
Pharmacodynamics
Drospirenone binds with high
| Progestogen | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|
| Drospirenone | 19–70 | 1–65 | 0–1 | 1–6 | 100–500 | 0 | 0 |
| Progesterone | 100 | 0–80 | 0–1 | 6–35 | 100–1000 | 0 | 0 |
| Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PR, metribolone for the AR, estradiol for the ER, dexamethasone for the GR, and aldosterone for the MR. Sources: [4][1][5][74][65] | |||||||
Progestogenic activity
Drospirenone is an
Due to its antigonadotropic effects, drospirenone inhibits the secretion of the
Antimineralocorticoid activity
Drospirenone is an antagonist of the MR, the biological target of mineralocorticoids like aldosterone, and hence is an antimineralocorticoid.[72] It has about 100 to 500% of the affinity of aldosterone for the MR and about 50 to 230% of the affinity of progesterone for the MR.[1][4][74][65] Drospirenone is about 5.5 to 11 times more potent as an antimineralocorticoid than spironolactone in animals.[72][78][85] Accordingly, 3 to 4 mg drospirenone is said to be equivalent to about 20 to 25 mg spironolactone in terms of antimineralocorticoid activity.[86][3] It has been said that the pharmacological profile of drospirenone more closely resembles that of progesterone than other progestins due to its antimineralocorticoid activity.[72] Drospirenone is the only clinically used progestogen with prominent antimineralocorticoid activity besides progesterone.[1] For comparison to progesterone, a 200 mg dose of oral progesterone is considered to be approximately equivalent in antimineralocorticoid effect to a 25 to 50 mg dose of spironolactone.[87] Both drospirenone and progesterone are actually weak partial agonists of the MR in the absence of mineralocorticoids.[5][4][65]
Due to its antimineralocorticoid activity, drospirenone increases
Antiandrogenic activity
Drospirenone is an antagonist of the AR, the biological target of androgens like testosterone and dihydrotestosterone (DHT).[1][4] It has about 1 to 65% of the affinity of the synthetic anabolic steroid metribolone for the AR.[1][4][5][65] The medication is more potent as an antiandrogen than spironolactone, but is less potent than cyproterone acetate, with about 30% of its antiandrogenic activity in animals.[1][91][72][78] Progesterone displays antiandrogenic activity in some assays similarly to drospirenone,[4] although this issue is controversial and many researchers regard progesterone as having no significant antiandrogenic activity.[92][1][5]
Drospirenone shows antiandrogenic effects on the
Other activity
Drospirenone stimulates the
Pharmacokinetics
Absorption
The
Distribution
The
Metabolism
The
Elimination
Drospirenone is
The
Chemistry
Chemical structures of spirolactones
|
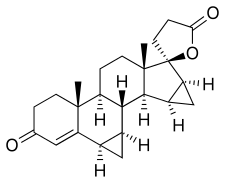
Drospirenone, also known as 1,2-dihydrospirorenone or as 17β-hydroxy-6β,7β:15β,16β-dimethylene-3-oxo-17α-pregn-4-ene-21-carboxylic acid, γ-lactone, is a
Spirolactones like drospirenone and spironolactone are
History
Drospirenone was patented in 1976 and introduced for medical use in 2000.
Society and culture
Generic names
Drospirenone is the
Brand names
Drospirenone is marketed in combination with an estrogen under a variety of brand names throughout the world.[9] Among others, it is marketed in combination with ethinylestradiol under the brand names Yasmin and Yaz, in combination with estetrol under the brand name Nextstellis, and in combination with estradiol under the brand name Angeliq.[9][105]
Availability
Drospirenone is marketed widely throughout the world.[9]
Generation
Drospirenone has been categorized as a "fourth-generation" progestin.[65]
Litigation
Many lawsuits have been filed against
In July 2012, Bayer notified its stockholders that there were more than 12,000 such lawsuits against the company involving Yaz, Yasmin, and other birth control pills with drospirenone.[110] They also noted that the company by then had settled 1,977 cases for US$402.6 million, for an average of US$212,000 per case, while setting aside US$610.5 million to settle the others.[110]
As of 17 July 2015, there have been at least 4,000 lawsuits and claims still pending regarding VTE related to drospirenone.
Research
A combination of
Drospirenone has been suggested for potential use as a progestin in
Drospirenone has been studied in forms for
References
- ^ S2CID 24616324.
- ^ "Health product highlights 2021: Annexes of products approved in 2021". Health Canada. 3 August 2022. Retrieved 25 March 2024.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au Slynd-drospirenone tablet, film coated drug label/data at DailyMed from U.S. National Library of Medicine, National Institutes of Health.
- ^ PMID 11024226.
- ^ PMID 23238854.
- PMID 15033400.
- ^ PMID 19936169.
- ^ PMID 26271371.
- ^ a b c d e f g h i j k drospirenone at Drugs.com: Multum Consumer Information
- PMID 11024226.
- ^ PMID 26598309.
- S2CID 35051390.
- PMID 12659403.
- ^ ISBN 978-3-527-32669-3.
- ^ ISBN 978-3-527-60749-5.
- ^ ISBN 978-1-59708-001-9.
- ^ ISBN 978-0-323-41753-2.
- ^ Generic Yasmin Availability via yasminat Drugs.com
- ^ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ^ "Drospirenone; Ethinyl Estradiol - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ^ a b "Yaz- drospirenone and ethinyl estradiol kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ Cerner Multum, Inc. (11 June 2012). "drospirenone and ethinyl estradiol". Auckland, New Zealand: Drugs.com. Retrieved 24 October 2011.
- ^ a b c "Angeliq- drospirenone and estradiol tablet, film coated". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- PMID 15495039.
- S2CID 25078579.
- ^ S2CID 38095916.
- PMID 28217493.
- PMID 32699777.
- PMID 28477648.
- PMID 37365881.
- S2CID 42803561.
- ^ S2CID 9221524.
- ^ S2CID 1909717.
- S2CID 12572825.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 23 December 2019.
- ^ Center for Drug Evaluation and Research. "Drug Safety and Availability - FDA Drug Safety Communication: Updated information about the risk of blood clots in women taking birth control pills containing drospirenone". www.fda.gov. Retrieved 7 November 2017.
- ^ "NEXTSTELLIS (drospirenone and estetrol tablets)" (PDF). Mayne Pharma. U.S. Food and Drug Administration. April 2021.
- ^ "U.S. FDA Approved NEXTSTELLIS®, New Oral Contraceptive" (PDF). Mayne Pharma. Archived from the original (PDF) on 17 April 2021.
- ^ "Mayne Pharma and Mithra Announce FDA Approval of New Oral Contraceptive Nextstellis®" (PDF).
- ^ "Ocella- drospirenone and ethinyl estradiol kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Syeda- drospirenone and ethinyl estradiol kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Yasmin- drospirenone and ethinyl estradiol kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Zarah- drospirenone and ethinyl estradiol kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Beyaz- drospirenone/ethinyl estradiol/levomefolate calcium and levomefolate calcium kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Tydemy- drospirenone, ethinyl estradiol and levomefolate calcium and levomefolate calcium kit". DailyMed. U.S. National Library of Medicine. Retrieved 17 April 2021.
- ^ "Estetrol monohydrate and Drospirenone Tablets" (PDF). Searchlight Pharma Inc. Archived from the original (PDF) on 13 April 2021. Retrieved 15 January 2022.
- ^ "Mithra and Searchlight Pharma Announce Nextstellis Approval in Canada". Searchlight Pharma (Press release). 8 March 2021. Retrieved 17 April 2021.
- ^ a b c Bayer (25 March 2013). "Summary of Product Characteristics (SPC): Yasmin". London: electronic Medicines Compendium (eMC), Datapharm. Retrieved 24 April 2014.
4.3. Contraindications: • Severe chronic kidney disease or acute kidney failure. • Presence or history of severe hepatic disease as long as liver function values have not returned to normal.
- ^ a b Bayer (10 April 2012). "Yasmin full prescribing information" (PDF). Silver Spring, Md.: Food and Drug Administration (FDA). Retrieved 14 April 2012.
4. Contraindications: • Renal impairment. • Adrenal insufficiency. • Liver disease.
- OCLC 781956734.
- ^ PMID 26780736.
- ^ PMID 27051991.
- ^ a b "FDA Drug Safety Communication: Updated information about the risk of blood clots in women taking birth control pills containing drospirenone". Food and Drug Administration. 27 April 2019. Archived from the original on 27 April 2019.
- PMID 30626577.
- ^ PMID 27854556.
- ^ PMID 27678035.
- PMID 29573722.
- PMID 29388678.
- ^ S2CID 27088428.
- ^ PMID 8794429.
- ^ PMID 23384742.
- ^ S2CID 207481206.
- ^ S2CID 205931204.
- ^ S2CID 4306359.
- ^ S2CID 23630452.
- S2CID 31497860.
- PMID 26357645.
- PMID 31239112.
- ^ PMID 31512725.
- PMID 31474332.
- S2CID 21894200.
- ^ S2CID 36861309.
- PMID 8922878.
- ^ S2CID 12142015.
- S2CID 53246678.
- PMID 22078182.
- ^ a b Kuhl H (2011). "Pharmacology of Progestogens" (PDF). J Reproduktionsmed Endokrinol. 8 (1): 157–177.
- ^ S2CID 41756726.
- PMID 14670641.
- ^ "Drospirenone". pubchem.ncbi.nlm.nih.gov.
- ^ PMID 31446440.
- ^ S2CID 83941717.
- PMID 20933120.
- ISBN 978-1-139-00335-3.
- ^ ISBN 978-0-12-369443-0.
- ISBN 978-0-203-02496-6.
- PMID 8616985.
- S2CID 24872884.
- S2CID 59251387.
- S2CID 2632187.
- S2CID 23112620.
- PMID 23484104.
- ISSN 2197-8735.
- ^ S2CID 29808177.
- ^ ISBN 978-3-527-30247-5.
- ISBN 978-3-319-14385-9.
- S2CID 19701784.
[Spironolactone] was synthesized after the demonstration of the natriuretic effect of progesterone (Landau et al., 1955).
- ISBN 978-1-4557-1126-0.
[Spironolactone] is a potent antimineralocorticoid which was developed as a progestational analog [...]
- ISBN 978-0-12-817783-9.
In addition to spironolactone, which is a derivative of progesterone [...]
- PMID 16101407.
- PMID 2744183.
- S2CID 20150966.
- ^ WO patent 9806738, Mohr, Jörg-Thorsten & Nickisch, Klaus, "PROCESS FOR PRODUCING DROSPIRENONE (6ss,7ss;15ss,16ss-DIMETHYLENE-3-OXO-17 alpha -PREGN-4-EN-21,17-CARBOLACTONE, DRSP), AS WELL AS 7 alpha -(3-HYDROXY-1-PROPYL)-6ss,7ss;15ss,16ss-DIMETHYLENE-5ss-ANDROSTANE-3ss,5,17ss-TRIOL (ZK 92836) AND 6ss,7ss;15ss,16ss-DIMETHYLENE-5ss HYDROXY-5-OXO-17 alpha -ANDROSTANE-21, 17-CARBOLACTONE", issued 1998-02-19, assigned to Shering AG
- ^ US patent 6121465, Mohr, Joerg-Thorston & Nickisch, Klaus, "Process for production drospirenone and intermediate products of the process", issued 2000-09-19, assigned to Scheiring AG and Bayer Schering Pharma
- ^ a b c "Drospirenone/Estetrol - Mithra Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG.
- ^ "Ethinylestradiol/drospirenone". AdisInsight. Springer Nature Switzerland AG.
- ^ "Ethinylestradiol/drospirenone/folic acid". AdisInsight. Springer Nature Switzerland AG.
- ^ "Drospirenone/ethinylestradiol low-dose - Bayer HealthCare Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG.
- ^ "Estradiol/drospirenone - Bayer HealthCare Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG.
- ^ a b Feeley J, Kresge N (31 July 2012). "Bayer's Yasmin lawsuit settlements rise to $402.6 million". Bloomberg News. New York. Retrieved 11 November 2012.
- ^ a b c d "Quarterly Reports of Bayer". Bayer AG.
- ^ a b c "Drospirenone/estradiol/prasterone - ANI Pharmaceuticals/Pantarhei Bioscience". AdisInsight. Springer Nature Switzerland AG.
- PMID 21729745.
- PMID 22940138.
- PMID 23116659.
- S2CID 42932558.
Further reading
- Archer DF (February 2007). "Drospirenone and estradiol: a new option for the postmenopausal woman". Climacteric. 10 (Suppl 1): 3–10. S2CID 9221524.
- Archer DF (February 2007). "Drospirenone-containing hormone therapy for postmenopausal women. Perspective on current data". The Journal of Reproductive Medicine. 52 (2 Suppl): 159–164. PMID 17477110.
- Archer DF (2007). "Drospirenone, a progestin with added value for hypertensive postmenopausal women". Menopause. 14 (3 Pt 1): 352–354. PMID 17414576.
- Batur P, Casey PM (February 2017). "Drospirenone Litigation: Does the Punishment Fit the Crime?". Journal of Women's Health. 26 (2): 99–102. PMID 27854556.
- Bitzer J, Paoletti AM (2009). "Added benefits and user satisfaction with a low-dose oral contraceptive containing drospirenone: results of three multicentre trials". Clinical Drug Investigation. 29 (2): 73–78. S2CID 10356578.
- Carranza-Lira S (2009). "Safety, efficacy and patient acceptability of drospirenone and estradiol in the treatment of menopausal vasomotor symptoms: a review". Clinical Interventions in Aging. 4: 59–62. PMID 19503766.
- Christiansen C (October 2005). "Effects of drospirenone/estrogen combinations on bone metabolism". Climacteric. 8 (Suppl 3): 35–41. S2CID 42803561.
- Dickerson V (November 2002). "Quality of life issues. Potential role for an oral contraceptive containing ethinyl estradiol and drospirenone". The Journal of Reproductive Medicine. 47 (11 Suppl): 985–993. PMID 12497673.
- Fenton C, Wellington K, Moen MD, Robinson DM (2007). "Drospirenone/ethinylestradiol 3mg/20microg (24/4 day regimen): a review of its use in contraception, premenstrual dysphoric disorder and moderate acne vulgaris". Drugs. 67 (12): 1749–1765. S2CID 46976925.
- Foidart JM (October 2005). "Added benefits of drospirenone for compliance". Climacteric. 8 (Suppl 3): 28–34. S2CID 31883491.
- Foidart JM, Faustmann T (December 2007). "Advances in hormone replacement therapy: weight benefits of drospirenone, a 17alpha-spirolactone-derived progestogen". Gynecological Endocrinology. 23 (12): 692–699. S2CID 12572825.
- Genazzani AR, Mannella P, Simoncini T (February 2007). "Drospirenone and its antialdosterone properties". Climacteric. 10 (Suppl 1): 11–18. S2CID 24872884.
- Han L, Jensen JT (October 2014). "Expert opinion on a flexible extended regimen of drospirenone/ethinyl estradiol contraceptive". Expert Opinion on Pharmacotherapy. 15 (14): 2071–2079. S2CID 25338932.
- Heinemann LA, Dinger J (2004). "Safety of a new oral contraceptive containing drospirenone". Drug Safety. 27 (13): 1001–1018. S2CID 1773936.
- Idota N, Kobayashi M, Miyamori D, Kakiuchi Y, Ikegaya H (March 2015). "Drospirenone detected in postmortem blood of a young woman with pulmonary thromboembolism: A case report and review of the literature". Legal Medicine. 17 (2): 109–115. PMID 25454533.
- Keam SJ, Wagstaff AJ (2003). "Ethinylestradiol/drospirenone: a review of its use as an oral contraceptive". Treatments in Endocrinology. 2 (1): 49–70. S2CID 209144694.
- Krattenmacher R (July 2000). "Drospirenone: pharmacology and pharmacokinetics of a unique progestogen". Contraception. 62 (1): 29–38. PMID 11024226.
- Larivée N, Suissa S, Khosrow-Khavar F, Tagalakis V, Filion KB (September 2017). "Drospirenone-containing oral contraceptive pills and the risk of venous thromboembolism: a systematic review of observational studies". BJOG. 124 (10): 1490–1499. PMID 28276140.
- Lete I, Chabbert-Buffet N, Jamin C, Lello S, Lobo P, Nappi RE, et al. (2015). "Haemostatic and metabolic impact of estradiol pills and drospirenone-containing ethinylestradiol pills vs. levonorgestrel-containing ethinylestradiol pills: A literature review". The European Journal of Contraception & Reproductive Health Care. 20 (5): 329–343. S2CID 41601833.
- Li J, Ren J, Sun W (March 2017). "A comparative systematic review of Yasmin (drospirenone pill) versus standard treatment options for symptoms of polycystic ovary syndrome". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 210: 13–21. PMID 27923166.
- Machado RB, Pompei LD, Giribela AG, Giribela CG (January 2011). "Drospirenone/ethinylestradiol: a review on efficacy and noncontraceptive benefits". Women's Health. 7 (1): 19–30. PMID 21175386.
- Mallareddy M, Hanes V, White WB (2007). "Drospirenone, a new progestogen, for postmenopausal women with hypertension". Drugs & Aging. 24 (6): 453–466. S2CID 39236155.
- Motivala A, Pitt B (2007). "Drospirenone for oral contraception and hormone replacement therapy: are its cardiovascular risks and benefits the same as other progestogens?". Drugs. 67 (5): 647–655. S2CID 22985078.
- Oelkers W (December 2002). "Antimineralocorticoid activity of a novel oral contraceptive containing drospirenone, a unique progestogen resembling natural progesterone". The European Journal of Contraception & Reproductive Health Care. 7 (Suppl 3): 19–26, discussion 42–3. PMID 12659403.
- Oelkers W (December 2000). "Drospirenone--a new progestogen with antimineralocorticoid activity, resembling natural progesterone". The European Journal of Contraception & Reproductive Health Care. 5 (Suppl 3): 17–24. S2CID 35051390.
- Oelkers W (March 2004). "Drospirenone, a progestogen with antimineralocorticoid properties: a short review". Molecular and Cellular Endocrinology. 217 (1–2): 255–261. S2CID 19936032.
- Oelkers W (February 2002). "The renin-aldosterone system and drospirenone". Gynecological Endocrinology. 16 (1): 83–87. S2CID 32410408.
- Oelkers WH (October 2005). "Drospirenone in combination with estrogens: for contraception and hormone replacement therapy". Climacteric. 8 (Suppl 3): 19–27. S2CID 42837148.
- Palacios S, Foidart JM, Genazzani AR (November 2006). "Advances in hormone replacement therapy with drospirenone, a unique progestogen with aldosterone receptor antagonism". Maturitas. 55 (4): 297–307. PMID 16949774.
- Pérez-López FR (June 2008). "Clinical experiences with drospirenone: from reproductive to postmenopausal years". Maturitas. 60 (2): 78–91. PMID 18468818.
- Rapkin AJ, Sorger SN, Winer SA (February 2008). "Drospirenone/ethinyl estradiol". Drugs of Today. 44 (2): 133–145. S2CID 32413831.
- Rapkin AJ, Winer SA (May 2007). "Drospirenone: a novel progestin". Expert Opinion on Pharmacotherapy. 8 (7): 989–999. S2CID 6954183.
- Rapkin RB, Creinin MD (October 2011). "The combined oral contraceptive pill containing drospirenone and ethinyl estradiol plus levomefolate calcium". Expert Opinion on Pharmacotherapy. 12 (15): 2403–2410. S2CID 40231903.
- Rübig A (October 2003). "Drospirenone: a new cardiovascular-active progestin with antialdosterone and antiandrogenic properties". Climacteric. 6 (Suppl 3): 49–54. PMID 15018248.
- Scheinfeld NS (2007). "Yaz (3 mg drospirenone/20 microg ethinyl estradiol)". Skinmed. 6 (6): 289. PMID 17975349.
- Sehovic N, Smith KP (May 2010). "Risk of venous thromboembolism with drospirenone in combined oral contraceptive products". The Annals of Pharmacotherapy. 44 (5): 898–903. S2CID 8248469.
- Shulman LP (June 2006). "A review of drospirenone for safety and tolerability and effects on endometrial safety and lipid parameters contrasted with medroxyprogesterone acetate, levonorgestrel, and micronized progesterone". Journal of Women's Health. 15 (5): 584–590. PMID 16796485.
- Simoncini T, Genazzani AR (February 2010). "A review of the cardiovascular and breast actions of drospirenone in preclinical studies". Climacteric. 13 (1): 22–33. S2CID 4306359.
- Sitruk-Ware R (October 2005). "Pharmacology of different progestogens: the special case of drospirenone". Climacteric. 8 (Suppl 3): 4–12. S2CID 24205704.
- Thorneycroft IH (November 2002). "Evolution of progestins. Focus on the novel progestin drospirenone". The Journal of Reproductive Medicine. 47 (11 Suppl): 975–980. PMID 12497671.
- Toni I, Neubert A, Botzenhardt S, Gratzki N, Rascher W (September 2013). "Venous thromboembolism in adolescents associated with drospirenone-containing oral contraceptives - two case reports". Klinische Padiatrie. 225 (5): 266–267. S2CID 19085818.
- White WB (February 2007). "Drospirenone with 17beta-estradiol in the postmenopausal woman with hypertension". Climacteric. 10 (Suppl 1): 25–31. S2CID 9451771.
- Whitehead M (March 2006). "Hormone replacement therapy with estradiol and drospirenone: an overview of the clinical data". The Journal of the British Menopause Society. 12 (Suppl 1): 4–7. S2CID 38095916.
- Wu CQ, Grandi SM, Filion KB, Abenhaim HA, Joseph L, Eisenberg MJ (June 2013). "Drospirenone-containing oral contraceptive pills and the risk of venous and arterial thrombosis: a systematic review". BJOG. 120 (7): 801–810. S2CID 206904730.
- Zhao X, Zhang XF, Zhao Y, Lin X, Li NY, Paudel G, et al. (September 2016). "Effect of combined drospirenone with estradiol for hypertensive postmenopausal women: a systemic review and meta-analysis". Gynecological Endocrinology. 32 (9): 685–689. S2CID 9116138.
- "Drospirenone in HRT?". Drug and Therapeutics Bulletin. 47 (4): 41–44. April 2009. S2CID 1909717.

