Drug discovery
In the fields of medicine,
Historically, drugs were discovered by identifying the active ingredient from traditional remedies or by
Modern drug discovery involves the identification of screening hits,
Modern drug discovery is thus usually a
Discovering drugs that may be a commercial success, or a public health success, involves a complex interaction between investors, industry, academia,
History
The idea that the effect of a drug in the human body is mediated by specific interactions of the drug molecule with biological macromolecules, (proteins or nucleic acids in most cases) led scientists to the conclusion that individual chemicals are required for the biological activity of the drug. This made for the beginning of the modern era in pharmacology, as pure chemicals, instead of crude extracts of medicinal plants, became the standard drugs. Examples of drug compounds isolated from crude preparations are morphine, the active agent in opium, and digoxin, a heart stimulant originating from Digitalis lanata. Organic chemistry also led to the synthesis of many of the natural products isolated from biological sources.
Historically, substances, whether crude extracts or purified chemicals, were screened for biological activity without knowledge of the biological target. Only after an active substance was identified was an effort made to identify the target. This approach is known as classical pharmacology, forward pharmacology,[10] or phenotypic drug discovery.[11]
Later, small molecules were synthesized to specifically target a known physiological/pathological pathway, avoiding the mass screening of banks of stored compounds. This led to great success, such as the work of
Gertrude Elion, working mostly with a group of fewer than 50 people on purine analogues, contributed to the discovery of the first anti-viral; the first immunosuppressant (azathioprine) that allowed human organ transplantation; the first drug to induce remission of childhood leukemia; pivotal anti-cancer treatments; an anti-malarial; an anti-bacterial; and a treatment for gout.
Cloning of human proteins made possible the screening of large libraries of compounds against specific targets thought to be linked to specific diseases. This approach is known as reverse pharmacology and is the most frequently used approach today.[17]
In the 2020s, qubit and quantum computing started to be used to reduce the time needed to drug discovery.[18]
Targets
A "target" is produced within the pharmaceutical industry.[8] Generally, the "target" is the naturally existing cellular or molecular structure involved in the pathology of interest where the drug-in-development is meant to act.[8] However, the distinction between a "new" and "established" target can be made without a full understanding of just what a "target" is. This distinction is typically made by pharmaceutical companies engaged in the discovery and development of therapeutics.[8] In an estimate from 2011, 435 human genome products were identified as therapeutic drug targets of FDA-approved drugs.[19]
"Established targets" are those for which there is a good scientific understanding, supported by a lengthy publication history, of both how the target functions in normal physiology and how it is involved in human pathology.
Screening and design
The process of finding a new drug against a chosen target for a particular disease usually involves
Another function of HTS is to show how selective the compounds are for the chosen target, as one wants to find a molecule which will interfere with only the chosen target, but not other, related targets.[21] To this end, other screening runs will be made to see whether the "hits" against the chosen target will interfere with other related targets – this is the process of cross-screening.[21] Cross-screening is useful because the more unrelated targets a compound hits, the more likely that off-target toxicity will occur with that compound once it reaches the clinic.[21]
It is unlikely that a perfect drug candidate will emerge from these early screening runs. One of the first steps is to screen for compounds that are unlikely to be developed into drugs; for example compounds that are hits in almost every assay, classified by medicinal chemists as "
- increase activity against the chosen target
- reduce activity against unrelated targets
- improve the druglikeness or ADME properties of the molecule.
This process will require several iterative screening runs, during which, it is hoped, the properties of the new molecular entities will improve, and allow the favoured compounds to go forward to in vitro and in vivo testing for activity in the disease model of choice.
Amongst the
A range of parameters can be used to assess the quality of a compound, or a series of compounds, as proposed in the
While HTS is a commonly used method for novel drug discovery, it is not the only method. It is often possible to start from a molecule which already has some of the desired properties. Such a molecule might be extracted from a natural product or even be a drug on the market which could be improved upon (so-called "me too" drugs). Other methods, such as virtual high throughput screening, where screening is done using computer-generated models and attempting to "dock" virtual libraries to a target, are also often used.[21]
Another method for drug discovery is de novo
There is also a paradigm shift in the drug discovery community to shift away from HTS, which is expensive and may only cover limited
Phenotypic screens have also provided new chemical starting points in drug discovery.[46][47] A variety of models have been used including yeast, zebrafish, worms, immortalized cell lines, primary cell lines, patient-derived cell lines and whole animal models. These screens are designed to find compounds which reverse a disease phenotype such as death, protein aggregation, mutant protein expression, or cell proliferation as examples in a more holistic cell model or organism. Smaller screening sets are often used for these screens, especially when the models are expensive or time-consuming to run.[48] In many cases, the exact mechanism of action of hits from these screens is unknown and may require extensive target deconvolution experiments to ascertain. The growth of the field of chemoproteomics has provided numerous strategies to identify drug targets in these cases.[49]
Once a lead compound series has been established with sufficient target potency and selectivity and favourable drug-like properties, one or two compounds will then be proposed for drug development. The best of these is generally called the lead compound, while the other will be designated as the "backup". These decisions are generally supported by computational modelling innovations.[50][51][52]

Nature as source
Traditionally, many drugs and other chemicals with biological activity have been discovered by studying chemicals that organisms create to affect the activity of other organisms for survival.[54]
Despite the rise of combinatorial chemistry as an integral part of lead discovery process, natural products still play a major role as starting material for drug discovery.[55] A 2007 report[56] found that of the 974 small molecule new chemical entities developed between 1981 and 2006, 63% were natural derived or semisynthetic derivatives of natural products. For certain therapy areas, such as antimicrobials, antineoplastics, antihypertensive and anti-inflammatory drugs, the numbers were higher.[citation needed]
Natural products may be useful as a source of novel chemical structures for modern techniques of development of antibacterial therapies.[57]
Plant-derived
Many secondary metabolites produced by plants have potential therapeutic medicinal properties. These secondary metabolites contain, bind to, and modify the function of proteins (receptors, enzymes, etc.). Consequently, plant derived natural products have often been used as the starting point for drug discovery.[58][59][60][61][3]
History
Until the
Jasmonates
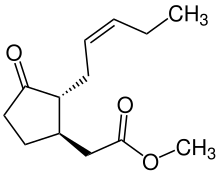
Jasmonates are important in responses to injury and intracellular signals. They induce apoptosis[66][67] and protein cascade via proteinase inhibitor,[66] have defense functions,[68] and regulate plant responses to different biotic and abiotic stresses.[68][69] Jasmonates also have the ability to directly act on mitochondrial membranes by inducing membrane depolarization via release of metabolites.[70]
Jasmonate derivatives (JAD) are also important in wound response and tissue
Salicylates

Salicylic acid (SA), a
Animal-derived
Some drugs used in modern medicine have been discovered in animals or are based on compounds found in animals. For example, the anticoagulant drugs, hirudin and its synthetic congener, bivalirudin, are based on saliva chemistry of the leech, Hirudo medicinalis.[75] Used to treat type 2 diabetes, exenatide was developed from saliva compounds of the Gila monster, a venomous lizard.[76]
Microbial metabolites
Microbes compete for living space and nutrients. To survive in these conditions, many microbes have developed abilities to prevent competing species from proliferating. Microbes are the main source of antimicrobial drugs. Streptomyces isolates have been such a valuable source of antibiotics, that they have been called medicinal molds. The classic example of an antibiotic discovered as a defense mechanism against another microbe is penicillin in bacterial cultures contaminated by Penicillium fungi in 1928.[citation needed]
Marine invertebrates
Marine environments are potential sources for new bioactive agents.
Chemical diversity
As above mentioned, combinatorial chemistry was a key technology enabling the efficient generation of large screening libraries for the needs of high-throughput screening. However, now, after two decades of combinatorial chemistry, it has been pointed out that despite the increased efficiency in chemical synthesis, no increase in lead or drug candidates has been reached.
Screening
Two main approaches exist for the finding of new bioactive chemical entities from natural sources.
The first is sometimes referred to as random collection and screening of material, but the collection is far from random. Biological (often botanical) knowledge is often used to identify families that show promise. This approach is effective because only a small part of the earth's biodiversity has ever been tested for pharmaceutical activity. Also, organisms living in a species-rich environment need to evolve defensive and competitive mechanisms to survive. Those mechanisms might be exploited in the development of beneficial drugs.
A collection of plant, animal and microbial samples from rich ecosystems can potentially give rise to novel biological activities worth exploiting in the drug development process. One example of successful use of this strategy is the screening for antitumor agents by the
The second main approach involves
Artemisinin, an antimalarial agent from sweet wormtree Artemisia annua, used in Chinese medicine since 200BC is one drug used as part of combination therapy for multiresistant Plasmodium falciparum.
Additionally, since machine learning has become more advanced, virtual screening is now an option for drug developers. AI algorithms are being used to perform virtual screening of chemical compounds, which involves predicting the activity of a compound against a specific target. By using machine learning algorithms to analyse large amounts of chemical data, researchers can identify potential new drug candidates that are more likely to be effective against a specific disease. Algorithms, such as Nearest-Neighbour classifiers, RF, extreme learning machines, SVMs, and deep neural networks (DNNs), are used for VS based on synthesis feasibility and can also predict in vivo activity and toxicity.[78]
Structural elucidation
The elucidation of the chemical structure is critical to avoid the re-discovery of a chemical agent that is already known for its structure and chemical activity. Mass spectrometry is a method in which individual compounds are identified based on their mass/charge ratio, after ionization. Chemical compounds exist in nature as mixtures, so the combination of liquid chromatography and mass spectrometry (LC-MS) is often used to separate the individual chemicals. Databases of mass spectra for known compounds are available and can be used to assign a structure to an unknown mass spectrum. Nuclear magnetic resonance spectroscopy is the primary technique for determining chemical structures of natural products. NMR yields information about individual hydrogen and carbon atoms in the structure, allowing detailed reconstruction of the molecule's architecture.
New Drug Application
When a drug is developed with evidence throughout its history of research to show it is safe and effective for the intended use in the United States, the company can file an application – the New Drug Application (NDA) – to have the drug commercialized and available for clinical application.[79] NDA status enables the FDA to examine all submitted data on the drug to reach a decision on whether to approve or not approve the drug candidate based on its safety, specificity of effect, and efficacy of doses.[79]
See also
- Antitarget
- Bioinformatics
- Biomedical informatics
- Cheminformatics
- Drug discovery hit to lead
- Drug metabolism
- Fragment-based drug discovery
- High content screening
- Pharmacogenetics
- Pharmacognosy
- Physiologically-based pharmacokinetic modelling
- Pre-clinical development
- Protein-directed dynamic combinatorial chemistry
- Discovery and development of proton pump inhibitors
- Discovery and development of melatonin receptor agonists
- Discovery and development of nucleoside and nucleotide reverse transcriptase inhibitors
- Discovery and development of Bcr-Abl tyrosine kinase inhibitors
- Discovery and development of antiandrogens
- Discovery and development of cephalosporins
- Retrometabolic drug design
References
- ^ "The drug development process". US Food and Drug Administration. 4 January 2018. Retrieved 18 December 2019.
- ^ a b c "The drug development process: Step 1: Discover". US Food and Drug Administration. 4 January 2018. Retrieved 18 December 2019.
- ^ a b Helleboid S, Haug C, Lamottke K, et al. The Identification of Naturally Occurring Neoruscogenin as a Bioavailable, Potent, and High-Affinity Agonist of the Nuclear Receptor RORα (NR1F1). Journal of Biomolecular Screening. 2014;19(3):399–406. https://doi.org/10.1177/1087057113497095.
- ^ Herrmann, A., Roesner, M., Werner, T. et al. Potent inhibition of HIV replication in primary human cells by novel synthetic polyketides inspired by Aureothin. Sci Rep 10, 1326 (2020). https://doi.org/10.1038/s41598-020-57843-9.
- ^ "The drug development process: Step 3: Clinical research". US Food and Drug Administration. 4 January 2018. Retrieved 18 December 2019.
- OCLC 77706455. Archived from the originalon 21 September 2012. Retrieved 25 July 2009.
- S2CID 1299234.
- ^ a b c d Current Model for Financing Drug Development: From Concept Through Approval. Institute of Medicine (US), Forum on Drug Discovery, Development, and Translation, National Academies Press, Washington (DC). 2009.
- PMID 21395642.
- PMID 11589663.
- PMID 22409666.
- PMID 8494337.
- ^ Elion GB. "The purine path to chemotherapy. Nobel Lecture 1988".
- ^ Black J. "Drugs from emasculated hormones: the principles of synoptic antagonism. Nobel Lecture 1988". Retrieved 28 February 2014.
- ^ Endo A. "The discovery of the statins and their development". Retrieved 28 February 2014.
- S2CID 54305535.
- S2CID 19171881.
- ^ "Qubit Pharmaceuticals Accelerates Drug Discovery with Hybrid Quantum Computing". HPC Wire. 30 November 2022.
- S2CID 3328752.
- PMID 26265138.
- ^ PMID 32725781.
- PMID 28079090.
- PMID 25163000.
- PMID 20131845.
- PMID 15109945.
- PMID 19500981.
- S2CID 205476574.
- PMID 18600572.
- PMID 18084917.
- PMID 19128219.
- PMID 22035460.
- PMID 22183577.
- PMID 29733895.
- PMID 15214773.
- ISBN 978-3-527-31291-7.
- )
- ^ Edward Z, Michael S (2008). Fragment-based drug discovery a practical approach. Wiley.
- ^ Greaney MF, Bhat VT (2010). "Chapter 2: Protein-directed dynamic combinatorial chemistry". In Miller BL (ed.). Dynamic combinatorial chemistry: in drug discovery, bioinorganic chemistry, and materials sciences. New Jersey: John Wiley & Sons. pp. 43–82.
- PMID 27438816.
- PMID 25706945.
- PMID 24296754.
- ISBN 9783527312917.
- PMID 23795969.
- PMID 23202926.
- PMID 17206823.
- PMID 23850704.
- S2CID 19171881.
- S2CID 195798523.
- PMID 22284350.
- PMID 27069774.
- PMID 30411538.
- .
- ISBN 978-1-394-16628-2, retrieved 27 January 2023
- ISBN 978-1-4020-4279-9
- ^ PMID 12546556.
- ^ PMID 17309302.
- PMID 16881035.
The handling of natural products is cumbersome, requiring nonstandardized workflows and extended timelines. Revisiting natural products with modern chemistry and target-finding tools from biology (reversed genomics) is one option for their revival.
- S2CID 207777087.
With the current framework of HTS in major pharmaceutical industries and increasing government restrictions on drug approvals, it is possible that the number of new natural product–derived drugs could go to zero. However, this is likely to be temporary, as the potential for new discoveries in the longer term is enormous.
- S2CID 12369182.
Here, we review strategies for natural product screening that harness the recent technical advances that have reduced [technical barriers to screening natural products in high-throughput assays]. The growing appreciation of functional assays and phenotypic screens may further contribute to a revival of interest in natural products for drug discovery.
- PMID 26852623.
... the utilization of natural products and/or their novel structures, in order to discover and develop the final drug entity, is still alive and well. For example, in the area of cancer, over the time frame from around the 1940s to the end of 2014, of the 175 small molecules approved, 131, or 75%, are other than "S" (synthetic), with 85, or 49%, actually being either natural products or directly derived therefrom.
- PMID 28264468.
The outputs from 2016 indicate the so-called small molecules are losing ground against biologics, biomolecules, and other molecules inspired [by] natural products
- ISBN 978-0-500-25139-3.
- ^ PMID 27998396.
- PMID 28930211.
- PMID 29154803.
- ^ PMID 11960340.
- PMID 26208889.
- ^ PMID 12045275.
- PMID 27379115.
- PMID 15753398.
- ^ PMID 22509841.
- PMID 28602514.
- ^ PMID 27303403.
- ISBN 978-0-12-809447-1.
- PMID 33174088.
- PMID 21975753.
- PMID 15039835.
- ^ krishgen2023 (10 March 2023). "Artificial Intelligence in Drug Discovery Research: A Brief Overview". Krishgen Biosystems. Retrieved 16 September 2023.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ^ a b "The drug development process. Step 4: FDA drug review". US Food and Drug Administration. 4 January 2018. Retrieved 18 December 2019.
Further reading
- Gad SC (2005). Drug Discovery Handbook. Hoboken, N.J: Wiley-Interscience/J. Wiley. ISBN 978-0-471-21384-0.
- Madsen U, Krogsgaard-Larsen P, Liljefors T (2002). Textbook of Drug Design and Discovery. Washington, DC: Taylor & Francis. ISBN 978-0-415-28288-8.
- ISBN 978-1-42141-340-2.
External links
- Drug Discovery at Curlie

