Early-onset Alzheimer's disease
This article needs more primary sources. (December 2021) |  |
| Early-onset Alzheimer's disease | |
|---|---|
| Other names |
|
| Specialty | Neurology |
Early-onset Alzheimer's disease (EOAD), also called younger-onset Alzheimer's disease (YOAD),
Nonfamilial early-onset AD can develop in people who are in their 30s or 40s, but this is extremely rare,[3] and mostly people in their 50s or early 60s are affected.
Familial and nonfamilial Alzheimer's disease
Alzheimer's disease (AD) is a
Signs and symptoms
Early-onset Alzheimer's disease strikes earlier in life, defined as before the age of 65 (usually between 30 and 60 years of age).[
Causes
Familial AD is inherited in an
Genetics
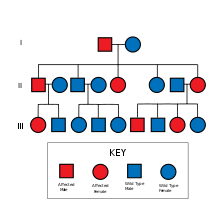

Familial Alzheimer disease is caused by a mutation in one of at least three genes, which code for
PSEN1 – Presenilin 1
The presenilin 1 gene (
The gene contains 14 exons, and the coding portion is estimated at 60 kb, as reported by Rogaev (1997)[9] and Del-Favero (1999).[10] The protein the gene codes for (PS1) is an integral membrane protein. As stated by Ikeuchi (2002)[11] it cleaves the protein Notch1 so is thought by Koizumi (2001)[12] to have a role in somitogenesis in the embryo. It also has an action on an amyloid precursor protein, which gives its probable role in the pathogenesis of FAD. Homologs of PS1 have been found in plants, invertebrates and other vertebrates.
Some of the mutations in the gene, of which over 90 are known, include: His163Arg, Ala246Glu, Leu286Val and Cys410Tyr. Most display complete penetrance, but a common mutation is Glu318Gly and this predisposes individuals to familial AD, with a study by Taddei (2002)[13] finding an incidence of 8.7% in patients with familial AD.
PSEN2 – Presenilin 2
The presenilin 2 gene (
The mutations have not been studied as much as PSEN1, but distinct allelic variants have been identified. These include Asn141Ile, which was identified first by Rudolph Tanzi and Jerry Schellenberg in Volga German families with familial Alzheimer disease (Levy-Lahad et al. Nature, 1995). One of these studies by Nochlin (1998) found severe amyloid angiopathy in the affected individuals in a family. This phenotype may be explained by a study by Tomita (1997)[17] suggesting that the Asn141Ile mutation alters APP metabolism causing an increased rate of protein deposition into plaques.
Similarly, miR-212-3p, another molecule implicated in Alzheimer's disease, has recently been shown to control inflammation in the brain, which could potentially influence plaque formation and Alzheimer's progression.[18]
Other allelic variants are Met239Val which was identified in an Italian pedigree by Rogaev (1995)[19] who also suggested early on that the gene may be similar to PSEN1, and an Asp439Ala mutation in exon 12 of the gene which is suggested by Lleo (2001)[20] to change the endoproteolytic processing of the PS2.
APP – amyloid beta (A4) precursor protein
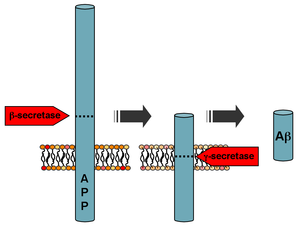
Mutations to the
[22] This type accounts for no more than 10–15% of EOFAD.[medical citation needed]
As of 2023, the count of known pathogenic APP mutations stands at just over 20.[23] The most prevalent among these mutations - APP V717I, known as the London Mutation - was first identified in 1991 within the family of Carol Jennings by a research team led by John Hardy.[24][25][26] Other notable APP mutations include the Swedish[27] and Arctic mutations.[28] Functional analyses of these mutations have significantly increased the understanding of the disease pathogenesis. Whereas the Swedish mutation, located at the cleavage site for β-secretase, results in an overall higher production of Aβ peptides by increasing the β-secretory cleavage,[29] the London mutation, as well as other mutations in the APP at codon 717, shifts the ratio of toxic Aβ species to the more aggregate-prone 42 amino-acid length peptide,[30] while the Arctic mutation leads to a conformation change of the Aβ peptide and increased formation of toxic Aβ protofibrils.[31]
Non-genetic risk factors
Non-genetic risk factors for early onset sporadic Alzheimer's disease and other forms of early onset dementia are understudied. However, recent research suggests that there are multiple modifiable and nonmodifiable risk factors for young onset dementia.[32]
Mechanism
The underlying neurobiology of this disease is just recently starting to be understood. Researchers have been working on mapping the inflammation pathways associated with the development, progression, and degenerative properties of AD. The major molecules involved in these pathways include glial cells (specifically astrocytes and microglia), beta-amyloid, and proinflammatory compounds. As neurons are injured and die throughout the brain, connections between networks of neurons may break down, and many brain regions begin to shrink. By the final stages of Alzheimer's, this process – called brain atrophy – is widespread, causing significant loss of brain volume. This loss of brain volume affects ones ability to live and function properly, ultimately being fatal.[33]
Beta-amyloid is a small piece of a larger protein called
Following cleavage by
Genetic testing
Genetic testing is available for symptomatic individuals and asymptomatic relatives.[6] Among families with EOFAD, 40–80% will have a detectable mutation in the APP, PSEN1, or PSEN2 gene. Therefore, some families with EOFAD will not have an identifiable mutation by testing.[medical citation needed]
Prognosis
The atypical lifecourse timing of early-onset Alzheimer's means that it presents distinctive impacts upon experience. For example, the disease can have devastating effects on the careers, caretakers and family members of patients.[36][37]
Those who are working lose their ability to perform their jobs competently, and are forced into early retirement. When this can be predicted, employees must discuss their future with their employers and the loss of skills they expect to face.[38] Those who are forced to retire early may not have access to the full range of benefits available to those who retire at the minimum age set by the government.[38] With some jobs, a mistake may have devastating consequences on a large number of people, and cases have been reported in which a person with early-onset Alzheimer's who is unaware of their condition has caused distress.[39]
Younger people with Alzheimer's may also lose their ability to take care of their own needs, such as money management.[40]
It has been suggested that conceptualizations of Alzheimer's and ageing should resist the notion that there are two distinct conditions.[41] A binary model, which focuses in particular on the needs of younger people, could lead to the challenges experienced by older people being understated.[42]
History
The symptoms of Alzheimer's disease as a distinct nosologic entity were first identified by Emil Kraepelin, who worked in Alzheimer's laboratory, and the characteristic neuropathology was first observed by Alois Alzheimer in 1906. Because of the overwhelming importance Kraepelin attached to finding the neuropathological basis of psychiatric disorders, Kraepelin made the decision that the disease would bear Alzheimer's name.[43]
Research directions
While early-onset familial AD is estimated to account for only 1% of total Alzheimer's disease,[3] it has presented a useful model in studying various aspects of the disorder. Currently, the early-onset familial AD gene mutations guide the vast majority of animal model-based therapeutic discovery and development for AD.[44]
See also
- Still Alice (novel) and the movie Still Alice, whose main protagonist has EOAD
- Spirit Unforgettable, a documentary film about the farewell tour of musician John Mann and his band Spirit of the West following his diagnosis with early-onset Alzheimer's
- Thanmathra (film), an award-winning Indian film detailing the effects of early-onset Alzheimer's disease on a father and his relationship with his son.
References
- ^ "Younger/ Early-onset Alzheimer's". Alzheimer's Association. Retrieved 9 July 2020.
- ^ "What Are the Signs of Alzheimer's Disease?".
- ^ PMID 12933919.
- PMID 23276979.
- S2CID 5946769.
- ^ PMID 19276785.
- ^ PMID 17659183.
- S2CID 4308372.
- PMID 9073509.
- PMID 10095119.
- S2CID 21552663.
- PMID 11262239.
- PMID 12192622.
- S2CID 27296868.
- S2CID 25596140.
- PMID 8661049.
- PMID 9050898.
- PMID 35150479.
- S2CID 4259326.
- S2CID 19309825.
- PMID 35150479.
- ISBN 978-0071481274.[page needed]
- ^ "APP". Alzheimer Research Forum. Retrieved 13 September 2023.
- ^ "APP V717I (London)". Alzheimer Research Forum. Retrieved 13 September 2023.
- S2CID 4336069.
- PMID 32164763.
- S2CID 20046036.
- S2CID 13516479.
- S2CID 43080346.
- PMID 11487570.
- PMID 16817891.
- PMID 38147328.
- ^ "What Happens to the Brain in Alzheimer's Disease?". National Institute on Aging. Retrieved 2020-05-07.
- PMID 35978311.
- PMID 20232515.
- ^ Mayo Clinic staff, Early-onset Alzheimer's: When symptoms begin before 65, Mayo Clinic
- ^ Mary Brophy Marcus, Family shares journey after early Alzheimer's diagnosis, USA Today (September 2, 2008).
- ^ a b Living With Early-Onset Alzheimer's Disease Archived 2007-10-19 at the Wayback Machine, Cleveland Clinic Health System
- ^ Early Onset Alzheimer's On The Rise, CBS News (March 8, 2008). Archived February 10, 2009, at the Wayback Machine
- ^ Fackelmann, Kathleen (June 11, 2007). "Who thinks of Alzheimer's in someone so young?". USA Today. Archived from the original on August 19, 2009.
- ^ Rahman, S. (2016). Young Onset Dementia: a label too far?, Dementia Society, July 27, 2016
- .
- PMID 9447568.
- PMID 31996736.
