Electromagnetic radiation

| Articles about |
| Electromagnetism |
|---|
 |
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy.[1] Types of EMR include radio waves, microwaves, infrared, (visible) light, ultraviolet, X-rays, and gamma rays, all of which are part of the electromagnetic spectrum.[2]
Classically, electromagnetic radiation consists of electromagnetic waves, which are synchronized oscillations of electric and magnetic fields. In a vacuum, electromagnetic waves travel at the speed of light, commonly denoted c. There, depending on the frequency of oscillation, different wavelengths of electromagnetic spectrum are produced. In homogeneous, isotropic media, the oscillations of the two fields are on average perpendicular to each other and perpendicular to the direction of energy and wave propagation, forming a transverse wave. The position of an electromagnetic wave within the electromagnetic spectrum can be characterized by either its frequency of oscillation or its wavelength. Electromagnetic waves of different frequency are called by different names since they have different sources and effects on matter. In order of increasing frequency and decreasing wavelength these are: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.[3]
Electromagnetic waves are emitted by electrically charged particles undergoing acceleration,[4][5] and these waves can subsequently interact with other charged particles, exerting force on them. EM waves carry energy, momentum, and angular momentum away from their source particle and can impart those quantities to matter with which they interact. Electromagnetic radiation is associated with those EM waves that are free to propagate themselves ("radiate") without the continuing influence of the moving charges that produced them, because they have achieved sufficient distance from those charges. Thus, EMR is sometimes referred to as the far field. In this language, the near field refers to EM fields near the charges and current that directly produced them, specifically electromagnetic induction and electrostatic induction phenomena.
In
The effects of EMR upon chemical compounds and biological organisms depend both upon the radiation's power and its frequency. EMR of lower energy ultraviolet or lower frequencies (i.e., Near ultraviolet, visible light, infrared, microwaves, and radio waves) is non-ionizing because its photons do not individually have enough energy to ionize atoms or molecules or to break chemical bonds. The effect of non-ionizing radiation on chemical systems and living tissue is primarily simply heating, through the combined energy transfer of many photons. In contrast, high frequency ultraviolet, X-rays and gamma rays are ionizing – individual photons of such high frequency have enough energy to ionize molecules or break chemical bonds. Ionizing radiation can cause chemical reactions and damage living cells beyond simply heating, and can be a health hazard and dangerous.
Physics
Theory
Maxwell's equations
Near and far fields

Maxwell's equations established that some charges and currents (sources) produce local
By contrast, the far field is composed of radiation that is free of the transmitter, in the sense that the transmitter requires the same power to send changes in the field out regardless of whether anything absorbs the signal, e.g. a radio station does not need to increase its power when more receivers use the signal. This far part of the electromagnetic field is electromagnetic radiation. The far fields propagate (radiate) without allowing the transmitter to affect them. This causes them to be independent in the sense that their existence and their energy, after they have left the transmitter, is completely independent of both transmitter and receiver. Due to conservation of energy, the amount of power passing through any spherical surface drawn around the source is the same. Because such a surface has an area proportional to the square of its distance from the source, the power density of EM radiation from an isotropic source decreases with the inverse square of the distance from the source; this is called the inverse-square law. This is in contrast to dipole parts of the EM field, the near field, which varies in intensity according to an inverse cube power law, and thus does not transport a conserved amount of energy over distances but instead fades with distance, with its energy (as noted) rapidly returning to the transmitter or absorbed by a nearby receiver (such as a transformer secondary coil).
In the Liénard–Wiechert potential formulation of the electric and magnetic fields due to motion of a single particle (according to Maxwell's equations), the terms associated with acceleration of the particle are those that are responsible for the part of the field that is regarded as electromagnetic radiation. By contrast, the term associated with the changing static electric field of the particle and the magnetic term that results from the particle's uniform velocity are both associated with the near field, and do not comprise electromagnetic radiation.[citation needed]
Properties

Electric and magnetic fields obey the properties of
The electromagnetic fields of light are not affected by traveling through static electric or magnetic fields in a linear medium such as a vacuum. However, in nonlinear media, such as some crystals, interactions can occur between light and static electric and magnetic fields—these interactions include the Faraday effect and the Kerr effect.[15][16]
In refraction, a wave crossing from one medium to another of different density alters its speed and direction upon entering the new medium. The ratio of the refractive indices of the media determines the degree of refraction, and is summarized by Snell's law. Light of composite wavelengths (natural sunlight) disperses into a visible spectrum passing through a prism, because of the wavelength-dependent refractive index of the prism material (dispersion); that is, each component wave within the composite light is bent a different amount.[17]
EM radiation exhibits both wave properties and
Some experiments display both the wave and particle natures of electromagnetic waves, such as the self-interference of a single
A quantum theory of the interaction between electromagnetic radiation and matter such as electrons is described by the theory of quantum electrodynamics.
Electromagnetic waves can be polarized, reflected, refracted, or diffracted, and can interfere with each other.[20][21][22]
Wave model

In homogeneous, isotropic media, electromagnetic radiation is a transverse wave,[23] meaning that its oscillations are perpendicular to the direction of energy transfer and travel. It comes from the following equations:
The electric and magnetic parts of the field in an electromagnetic wave stand in a fixed ratio of strengths to satisfy the two Maxwell equations that specify how one is produced from the other. In dissipation-less (lossless) media, these E and B fields are also in phase, with both reaching maxima and minima at the same points in space (see illustrations). In the far-field EM radiation which is described by the two source-free Maxwell curl operator equations, a time-change in one type of field is proportional to the curl of the other. These derivatives require that the E and B fields in EMR are in-phase (see mathematics section below).[citation needed] An important aspect of light's nature is its
A monochromatic wave (a wave of a single frequency) consists of successive troughs and crests, and the distance between two adjacent crests or troughs is called the wavelength. Waves of the electromagnetic spectrum vary in size, from very long radio waves longer than a continent to very short gamma rays smaller than atom nuclei. Frequency is inversely proportional to wavelength, according to the equation:[24]
where v is the speed of the wave (c in a vacuum or less in other media), f is the frequency and λ is the wavelength. As waves cross boundaries between different media, their speeds change but their frequencies remain constant.
Electromagnetic waves in free space must be solutions of Maxwell's
Interference is the superposition of two or more waves resulting in a new wave pattern. If the fields have components in the same direction, they constructively interfere, while opposite directions cause destructive interference. An example of interference caused by EMR is
The energy in electromagnetic waves is sometimes called radiant energy.[26][27][28]
Particle model and quantum theory
An anomaly arose in the late 19th century involving a contradiction between the wave theory of light and measurements of the electromagnetic spectra that were being emitted by thermal radiators known as
where h is the Planck constant, is the wavelength and c is the
Likewise, the momentum p of a photon is also proportional to its frequency and inversely proportional to its wavelength:
The source of Einstein's proposal that light was composed of particles (or could act as particles in some circumstances) was an experimental anomaly not explained by the wave theory: the
As a photon is absorbed by an atom, it excites the atom, elevating an electron to a higher energy level (one that is on average farther from the nucleus). When an electron in an excited molecule or atom descends to a lower energy level, it emits a photon of light at a frequency corresponding to the energy difference. Since the energy levels of electrons in atoms are discrete, each element and each molecule emits and absorbs its own characteristic frequencies. Immediate photon emission is called fluorescence, a type of photoluminescence. An example is visible light emitted from fluorescent paints, in response to ultraviolet (blacklight). Many other fluorescent emissions are known in spectral bands other than visible light. Delayed emission is called phosphorescence.[33][34]
Wave–particle duality
The modern theory that explains the nature of light includes the notion of wave–particle duality.
Wave and particle effects of electromagnetic radiation
Together, wave and particle effects fully explain the emission and absorption spectra of EM radiation. The matter-composition of the medium through which the light travels determines the nature of the absorption and emission spectrum. These bands correspond to the allowed energy levels in the atoms. Dark bands in the
These phenomena can aid various chemical determinations for the composition of gases lit from behind (absorption spectra) and for glowing gases (emission spectra). Spectroscopy (for example) determines what
Propagation speed
When any wire (or other conducting object such as an
As a wave, light is characterized by a velocity (the speed of light), wavelength, and frequency. As particles, light is a stream of photons. Each has an energy related to the frequency of the wave given by Planck's relation E = hf, where E is the energy of the photon, h is the Planck constant, 6.626 × 10−34 J·s, and f is the frequency of the wave.[37]
In a medium (other than vacuum),
History of discovery
Electromagnetic radiation of wavelengths other than those of visible light were discovered in the early 19th century. The discovery of
In 1801, German physicist Johann Wilhelm Ritter discovered ultraviolet in an experiment similar to Herschel's, using sunlight and a glass prism. Ritter noted that invisible rays near the violet edge of a solar spectrum dispersed by a triangular prism darkened silver chloride preparations more quickly than did the nearby violet light. Ritter's experiments were an early precursor to what would become photography. Ritter noted that the ultraviolet rays (which at first were called "chemical rays") were capable of causing chemical reactions.[40][41]

(1831–1879)
In 1862–64 James Clerk Maxwell developed equations for the electromagnetic field which suggested that waves in the field would travel with a speed that was very close to the known speed of light. Maxwell therefore suggested that visible light (as well as invisible infrared and ultraviolet rays by inference) all consisted of propagating disturbances (or radiation) in the electromagnetic field. Radio waves were first produced deliberately by Heinrich Hertz in 1887, using electrical circuits calculated to produce oscillations at a much lower frequency than that of visible light, following recipes for producing oscillating charges and currents suggested by Maxwell's equations. Hertz also developed ways to detect these waves, and produced and characterized what were later termed radio waves and microwaves.[42]: 286, 7
The last portion of the EM spectrum to be discovered was associated with
Electromagnetic spectrum

ELF = Extremely low frequency
EM radiation (the designation 'radiation' excludes static electric and magnetic and
For certain classes of EM waves, the waveform is most usefully treated as random, and then spectral analysis must be done by slightly different mathematical techniques appropriate to random or
The behavior of EM radiation and its interaction with matter depends on its frequency, and changes qualitatively as the frequency changes. Lower frequencies have longer wavelengths, and higher frequencies have shorter wavelengths, and are associated with photons of higher energy. There is no fundamental limit known to these wavelengths or energies, at either end of the spectrum, although photons with energies near the
Radio and microwave
When radio waves impinge upon a
Electromagnetic radiation phenomena with wavelengths ranging from as long as one meter to as short as one millimeter are called microwaves; with frequencies between 300 MHz (0.3 GHz) and 300 GHz.
At radio and microwave frequencies, EMR interacts with matter largely as a bulk collection of charges which are spread out over large numbers of affected atoms. In electrical conductors, such induced bulk movement of charges (electric currents) results in absorption of the EMR, or else separations of charges that cause generation of new EMR (effective reflection of the EMR). An example is absorption or emission of radio waves by antennas, or absorption of microwaves by water or other molecules with an electric dipole moment, as for example inside a microwave oven. These interactions produce either electric currents or heat, or both.
Infrared
Like radio and microwave, infrared (IR) also is reflected by metals (and also most EMR, well into the ultraviolet range). However, unlike lower-frequency radio and microwave radiation, Infrared EMR commonly interacts with dipoles present in single molecules, which change as atoms vibrate at the ends of a single chemical bond. It is consequently absorbed by a wide range of substances, causing them to increase in temperature as the vibrations dissipate as heat. The same process, run in reverse, causes bulk substances to radiate in the infrared spontaneously (see thermal radiation section below).
Infrared radiation is divided into spectral subregions. While different subdivision schemes exist,[43][44] the spectrum is commonly divided as near-infrared (0.75–1.4 μm), short-wavelength infrared (1.4–3 μm), mid-wavelength infrared (3–8 μm), long-wavelength infrared (8–15 μm) and far infrared (15–1000 μm).[45]
Visible light
Natural sources produce EM radiation across the spectrum. EM radiation with a wavelength between approximately 400 nm and 700 nm is directly detected by the human eye and perceived as visible light. Other wavelengths, especially nearby infrared (longer than 700 nm) and ultraviolet (shorter than 400 nm) are also sometimes referred to as light.
As frequency increases into the visible range, photons have enough energy to change the bond structure of some individual molecules. It is not a coincidence that this happens in the visible range, as the mechanism of vision involves the change in bonding of a single molecule, retinal, which absorbs a single photon. The change in retinal causes a change in the shape of the rhodopsin protein it is contained in, which starts the biochemical process that causes the retina of the human eye to sense the light.
Animals that detect infrared make use of small packets of water that change temperature, in an essentially thermal process that involves many photons.
Infrared, microwaves and radio waves are known to damage molecules and biological tissue only by bulk heating, not excitation from single photons of the radiation.
Visible light is able to affect only a tiny percentage of all molecules. Usually not in a permanent or damaging way, rather the photon excites an electron which then emits another photon when returning to its original position. This is the source of color produced by most dyes. Retinal is an exception. When a photon is absorbed, the retinal permanently changes structure from cis to trans, and requires a protein to convert it back, i.e. reset it to be able to function as a light detector again.
Limited evidence indicate that some
Ultraviolet
As frequency increases into the ultraviolet, photons now carry enough energy (about three
At the higher end of the ultraviolet range, the energy of photons becomes large enough to impart enough energy to electrons to cause them to be liberated from the atom, in a process called
X-rays and gamma rays
Electromagnetic radiation composed of photons that carry minimum-ionization energy, or more, (which includes the entire spectrum with shorter wavelengths), is therefore termed
Atmosphere and magnetosphere
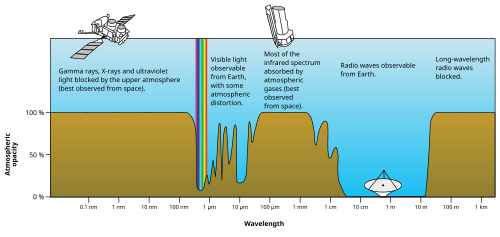
Most UV and X-rays are blocked by absorption first from molecular
Visible light is well transmitted in air, as it is not energetic enough to excite nitrogen, oxygen, or ozone, but too energetic to excite molecular vibrational frequencies of water vapor.[citation needed]
Absorption bands in the infrared are due to modes of vibrational excitation in water vapor. However, at energies too low to excite water vapor, the atmosphere becomes transparent again, allowing free transmission of most microwave and radio waves. [47]
Finally, at radio wavelengths longer than 10 m or so (about 30 MHz), the air in the lower atmosphere remains transparent to radio, but plasma in certain layers of the ionosphere begins to interact with radio waves (see skywave). This property allows some longer wavelengths (100 m or 3 MHz) to be reflected and results in shortwave radio beyond line-of-sight. However, certain ionospheric effects begin to block incoming radiowaves from space, when their frequency is less than about 10 MHz (wavelength longer than about 30 m).[48]
Thermal and electromagnetic radiation as a form of heat
The basic structure of
Ionizing radiation creates high-speed electrons in a material and breaks chemical bonds, but after these electrons collide many times with other atoms eventually most of the energy becomes thermal energy all in a tiny fraction of a second. This process makes ionizing radiation far more dangerous per unit of energy than non-ionizing radiation. This caveat also applies to UV, even though almost all of it is not ionizing, because UV can damage molecules due to electronic excitation, which is far greater per unit energy than heating effects.[49][citation needed]
Infrared radiation in the spectral distribution of a black body is usually considered a form of heat, since it has an equivalent temperature and is associated with an entropy change per unit of thermal energy. However, "heat" is a technical term in physics and thermodynamics and is often confused with thermal energy. Any type of electromagnetic energy can be transformed into thermal energy in interaction with matter. Thus, any electromagnetic radiation can "heat" (in the sense of increase the thermal energy temperature of) a material, when it is absorbed.[50]
The inverse or time-reversed process of absorption is thermal radiation. Much of the thermal energy in matter consists of random motion of charged particles, and this energy can be radiated away from the matter. The resulting radiation may subsequently be absorbed by another piece of matter, with the deposited energy heating the material.[51]
The electromagnetic radiation in an opaque cavity at thermal equilibrium is effectively a form of thermal energy, having maximum radiation entropy.[52]
Biological effects
Bioelectromagnetics is the study of the interactions and effects of EM radiation on living organisms. The effects of electromagnetic radiation upon living cells, including those in humans, depends upon the radiation's power and frequency. For low-frequency radiation (radio waves to near ultraviolet) the best-understood effects are those due to radiation power alone, acting through heating when radiation is absorbed. For these thermal effects, frequency is important as it affects the intensity of the radiation and penetration into the organism (for example, microwaves penetrate better than infrared). It is widely accepted that low frequency fields that are too weak to cause significant heating could not possibly have any biological effect.[53]
Some research suggests that weaker non-thermal electromagnetic fields (including weak ELF magnetic fields, although the latter does not strictly qualify as EM radiation[53][54][55]) and modulated RF and microwave fields can have biological effects, though the significance of this is unclear.[56][57]
The
This group contains possible carcinogens such as lead, DDT, and styrene.At higher frequencies (some of visible and beyond), the effects of individual photons begin to become important, as these now have enough energy individually to directly or indirectly damage biological molecules.[60] All UV frequencies have been classed as Group 1 carcinogens by the World Health Organization. Ultraviolet radiation from sun exposure is the primary cause of skin cancer.[61][62]
Thus, at UV frequencies and higher, electromagnetic radiation does more damage to biological systems than simple heating predicts. This is most obvious in the "far" (or "extreme") ultraviolet. UV, with X-ray and gamma radiation, are referred to as
Use as a weapon
The heat ray is an application of EMR that makes use of microwave frequencies to create an unpleasant heating effect in the upper layer of the skin. A publicly known heat ray weapon called the
Derivation from electromagnetic theory
Electromagnetic waves are predicted by the classical laws of electricity and magnetism, known as Maxwell's equations. There are nontrivial solutions of the homogeneous Maxwell's equations (without charges or currents), describing waves of changing electric and magnetic fields. Beginning with Maxwell's equations in free space:
|
|
(1)
|
|
|
(2)
|
|
|
(3)
|
|
|
(4)
|
where
- and are the /m2), respectively;
- yields the divergence and the curl of a vector field
- and are partial derivatives(rate of change in time, with location fixed) of the magnetic and electric field;
- is the permeability of a vacuum (4π × 10−7 H/m), and is the permittivity of a vacuum (8.85 × 10−12 F/m);
Besides the trivial solution
Taking the curl of the second Maxwell equation (2) yields:
|
|
(5)
|
Evaluating the left hand side of (5) with the above identity and simplifying using (1), yields:
|
|
(6)
|
Evaluating the right hand side of (5) by exchanging the sequence of derivatives and inserting the fourth Maxwell equation (4), yields:
|
|
(7)
|
Combining (6) and (7) again, gives a vector-valued differential equation for the electric field, solving the homogeneous Maxwell equations:
Taking the curl of the fourth Maxwell equation (4) results in a similar differential equation for a magnetic field solving the homogeneous Maxwell equations:
Both differential equations have the form of the general wave equation for waves propagating with speed where is a function of time and location, which gives the amplitude of the wave at some time at a certain location:
Comparing the terms for the speed of propagation, yields in the case of the electric and magnetic fields:
This is the speed of light in vacuum. Thus Maxwell's equations connect the vacuum permittivity , the vacuum permeability , and the speed of light, c0, via the above equation. This relationship had been discovered by Wilhelm Eduard Weber and Rudolf Kohlrausch prior to the development of Maxwell's electrodynamics, however Maxwell was the first to produce a field theory consistent with waves traveling at the speed of light.
These are only two equations versus the original four, so more information pertains to these waves hidden within Maxwell's equations. A generic vector wave for the electric field has the form
Here, is a constant vector, is any second differentiable function, is a unit vector in the direction of propagation, and is a position vector. is a generic solution to the wave equation. In other words,
From the first of Maxwell's equations, we get
Thus,
Thus,
The remaining equations will be satisfied by this choice of .
The electric and magnetic field waves in the far-field travel at the speed of light. They have a special restricted orientation and proportional magnitudes, , which can be seen immediately from the Poynting vector. The electric field, magnetic field, and direction of wave propagation are all orthogonal, and the wave propagates in the same direction as . Also, E and B far-fields in free space, which as wave solutions depend primarily on these two Maxwell equations, are in-phase with each other. This is guaranteed since the generic wave solution is first order in both space and time, and the
From the viewpoint of an electromagnetic wave traveling forward, the electric field might be oscillating up and down, while the magnetic field oscillates right and left. This picture can be rotated with the electric field oscillating right and left and the magnetic field oscillating down and up. This is a different solution that is traveling in the same direction. This arbitrariness in the orientation with respect to propagation direction is known as polarization. On a quantum level, it is described as photon polarization. The direction of the polarization is defined as the direction of the electric field.
More general forms of the second-order wave equations given above are available, allowing for both non-vacuum propagation media and sources. Many competing derivations exist, all with varying levels of approximation and intended applications. One very general example is a form of the electric field equation,[65] which was factorized into a pair of explicitly directional wave equations, and then efficiently reduced into a single uni-directional wave equation by means of a simple slow-evolution approximation.
See also
- Antenna measurement
- Bioelectromagnetics
- Bolometer
- CONELRAD
- Electromagnetic pulse
- Electromagnetic radiation and health
- Evanescent wave coupling
- Finite-difference time-domain method
- Gravitational wave
- Helicon
- Impedance of free space
- Radiation reaction
- Health effects of sunlight exposure
- Sinusoidal plane-wave solutions of the electromagnetic wave equation
References
- ISBN 978-1-107-01402-2. p 430: "These waves... require no medium to support their propagation. Traveling electromagnetic waves carry energy, and... the Poynting vector describes the energy flow...;" p 440: ... the electromagnetic wave must have the following properties: 1) The field pattern travels with speed c (speed of light); 2) At every point within the wave... the electric field strength E equals "c" times the magnetic field strength B; 3) The electric field and the magnetic field are perpendicular to one another and to the direction of travel, or propagation."
- ISBN 978-0-07-161399-6.; p319: "For historical reasons, different portions of the EM spectrum are given different names, although they are all the same kind of thing. Visible light constitutes a narrow range of the spectrum, from wavelengths of about 400-800 nm.... ;p 320 "An electromagnetic wave carries forward momentum... If the radiation is absorbed by a surface, the momentum drops to zero and a force is exerted on the surface... Thus the radiation pressure of an electromagnetic wave is (formula)."
- S2CID 186207827.
- ISBN 978-0-387-91501-2.
- ISBN 978-3-319-48329-0.
- ^ "The Dual Nature of Light as Reflected in the Nobel Archives". nobelprize.org. Archived from the original on 15 July 2017. Retrieved 4 September 2017.
- ^ "Electromagnetic Spectrum facts, information, pictures | Encyclopedia.com articles about Electromagnetic Spectrum". encyclopedia.com. Archived from the original on 13 June 2017. Retrieved 4 September 2017.
- ISBN 978-1-57259-491-3.
- ^ Elert, Glenn. "Electromagnetic Waves". The Physics Hypertextbook. Retrieved 4 June 2018.
- ^ "The Impact of James Clerk Maxwell's Work". clerkmaxwellfoundation.org. Archived from the original on 17 September 2017. Retrieved 4 September 2017.
- ^ "Maxwell's equations and the secrets of nature". plus.maths.org. 18 December 2015. Retrieved 2 May 2021.
- ^ "Electromagnetic radiation | Spectrum, Examples, & Types | Britannica". www.britannica.com. 15 September 2023. Retrieved 16 October 2023.
- ^ Purcell, p442: "Any number of electromagnetic waves can propagate through the same region without affecting one another. The field E at a space time point is the vector sum of the electric fields of the individual waves, and the same goes for B".
- ^ "PV Performance Modeling Collaborative | Plane of Array (POA) Irradiance". Retrieved 14 January 2022.
- S2CID 16080209.
- ^ Crowther, James Arnold (1920). The life and discoveries of Michael Faraday. Society for promoting Christian knowledge. pp. 54–57. Retrieved 15 June 2014.
- ^ "Prisms". Spectroscopy. Spectroscopy-09-01-2008. 23 (9). September 2008. Retrieved 17 January 2021.
- ^ Carmichael, H. J. "Einstein and the Photoelectric Effect" (PDF). Quantum Optics Theory Group, University of Auckland. Archived from the original (PDF) on 27 June 2007. Retrieved 22 December 2009.
- (PDF) from the original on 1 February 2016.
- ^ "DATE". galileo.phys.virginia.edu. Archived from the original on 12 May 2015. Retrieved 4 September 2017.
- ^ "Physics – Waves". www-jcsu.jesus.cam.ac.uk. Archived from the original on 4 September 2017. Retrieved 4 September 2017.
- ^ "Wave Behaviors | Science Mission Directorate". science.nasa.gov. Archived from the original on 14 May 2017. Retrieved 4 September 2017.
- ISBN 978-0-470-13153-4.
- ^ "Electromagnetic Radiation | COSMOS". astronomy.swin.edu.au. Retrieved 29 March 2020.
- PMID 27184813.
- ^ "What Is Electromagnetic Radiation?". Live Science. Archived from the original on 4 September 2017. Retrieved 4 September 2017.
- ISBN 978-1-4668-1443-1.
- ^ The Michigan Technic. UM Libraries. 1960.
- ISBN 978-0-471-49180-4.
- ISBN 978-0-521-55001-7.
- ISBN 978-1-107-06399-0.
- ISBN 978-1-947172-22-7.
- ^ Haneef, Deena T. Kochunni, Jazir. "7 Differences between Fluorescence and Phosphorescence". Archived from the original on 4 September 2017. Retrieved 4 September 2017.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ISBN 978-1-4832-8435-4.
- ^ Browne, p 376: "Radiation is emitted or absorbed only when the electron jumps from one orbit to the other, and the frequency of radiation depends only upon on the energies of the electron in the initial and final orbits.
- ^ "Spectroscopy". National Redshift Project. Archived from the original on 1 February 2017. Retrieved 19 January 2017.
- ISBN 978-0-367-38811-9.
- JSTOR 107057.
- PMID 20463675.
- ^ "Ultraviolet | COSMOS". astronomy.swin.edu.au. Archived from the original on 1 March 2021. Retrieved 29 September 2021.
- S2CID 135584871.
- ^ a b c Jeans, James (1947) The Growth of Physical Science. Cambridge University Press
- ^ Henderson, Roy. "Wavelength considerations". Instituts für Umform- und Hochleistungs. Archived from the original on 28 October 2007. Retrieved 18 October 2007.
- ^ "Near, Mid and Far-Infrared". NASA IPAC. Archived from the original on 29 May 2012. Retrieved 4 April 2007.
- ISBN 978-1-4020-9252-7.
- PMID 22318388.
- ^ Chaplin, Martin (15 May 2013). "Infared Spectroscopy" (PDF): water.lsbu.ac.uk. Retrieved 19 April 2022.
{{cite journal}}: Cite journal requires|journal=(help) - S2CID 121347063.
- ^ a b "CANDU textbook". nuceng.ca. Archived from the original on 20 April 2017. Retrieved 24 March 2017.
- ^ "Blackbody Radiation". docs.kde.org. Archived from the original on 8 August 2017. Retrieved 24 March 2017.
- ^ "Thermodynamics Part 1: Work, Heat, Internal Energy and Enthalpy". www2.southeastern.edu. Archived from the original on 24 March 2017. Retrieved 24 March 2017.
- ^ "Planck's law" (PDF). astro.lu.se. Archived from the original (PDF) on 30 November 2016. Retrieved 24 March 2017.
- ^ OCLC 49700531.
- PMID 7107514.
- PMID 9383244.
- PMID 16495939.
- PMID 2346507.
- ^ IARC classifies Radiofrequency Electromagnetic Fields as possibly carcinogenic to humans Archived 1 June 2011 at the Wayback Machine. World Health Organization. 31 May 2011
- ^ "Trouble with cell phone radiation standard". CBS News. Archived from the original on 9 May 2013.
- pyrimidine dimers.
- S2CID 22224492.
- PMID 15753968.
- ^ "DVIDS – News – New Marine Corps non-lethal weapon heats things up". DVIDS. Retrieved 1 November 2014.
- ^ "Effects on the human body: Extremely low frequency RF | Radio Frequency | Radio Spectrum". Scribd. Retrieved 8 March 2021.
- ^ Kinsler, P. (2010). "Optical pulse propagation with minimal approximations". Phys. Rev. A. 81 (1): 013819. .
- "Light: Electromagnetic waves, the electromagnetic spectrum and photons (article)". Khan Academy. Retrieved 2 May 2021.
Further reading
- Hecht, Eugene (2001). Optics (4th ed.). Pearson Education. ISBN 978-0-8053-8566-3.
- Serway, Raymond A.; Jewett, John W. (2004). Physics for Scientists and Engineers (6th ed.). Brooks Cole. ISBN 978-0-534-40842-8.
- Tipler, Paul (2004). Physics for Scientists and Engineers: Electricity, Magnetism, Light, and Elementary Modern Physics (5th ed.). W. H. Freeman. ISBN 978-0-7167-0810-0.
- Reitz, John; Milford, Frederick; Christy, Robert (1992). Foundations of Electromagnetic Theory (4th ed.). Addison Wesley. ISBN 978-0-201-52624-0.
- ISBN 978-0-471-30932-1.
- ISBN 978-1-58053-832-9.
External links
- The Feynman Lectures on Physics Vol. I Ch. 28: Electromagnetic Radiation
 Media related to Electromagnetic radiation at Wikimedia Commons
Media related to Electromagnetic radiation at Wikimedia Commons- Electromagnetic Waves from Maxwell's Equations on Project PHYSNET.
- "Electromagnetic radiation" in the Encyclopædia Britannica















































