Electron capture

Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral
-
p
+
e−
→
n
+
ν
e - or when written as a nuclear reaction equation, ν
Since this single emitted neutrino carries the entire decay energy, it has this single characteristic energy. Similarly, the momentum of the neutrino emission causes the daughter atom to recoil with a single characteristic momentum.
The resulting
Following capture of an inner electron from the atom, an outer electron replaces the electron that was captured and one or more characteristic X-ray photons is emitted in this process. Electron capture sometimes also results in the Auger effect, where an electron is ejected from the atom's electron shell due to interactions between the atom's electrons in the process of seeking a lower energy electron state.
Following electron capture, the atomic number is reduced by one, the neutron number is increased by one, and there is no change in mass number. Simple electron capture by itself results in a neutral atom, since the loss of the electron in the electron shell is balanced by a loss of positive nuclear charge. However, a positive atomic ion may result from further Auger electron emission.
Electron capture is an example of weak interaction, one of the four fundamental forces.
Electron capture is the primary
If the energy difference between the parent atom and the daughter atom is less than 1.022
History
The theory of electron capture was first discussed by
Reaction details
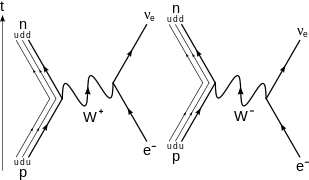
The electron that is captured is one of the atom's own electrons, and not a new, incoming electron, as might be suggested by the way the reactions are written below. A few examples of electron capture are:
Radioactive isotopes that decay by pure electron capture can be inhibited from radioactive decay if they are fully
Chemical bonds can also affect the rate of electron capture to a small degree (in general, less than 1%) depending on the proximity of electrons to the nucleus. For example, in 7Be, a difference of 0.9% has been observed between half-lives in metallic and insulating environments.[9] This relatively large effect is due to the fact that beryllium is a small atom that employs valence electrons that are close to the nucleus, and also in orbitals with no orbital angular momentum. Electrons in s orbitals (regardless of shell or primary quantum number), have a probability antinode at the nucleus, and are thus far more subject to electron capture than p or d electrons, which have a probability node at the nucleus.
Around the elements in the middle of the periodic table, isotopes that are lighter than stable isotopes of the same element tend to decay through electron capture, while isotopes heavier than the stable ones decay by electron emission. Electron capture happens most often in the heavier neutron-deficient elements where the mass change is smallest and positron emission is not always possible. When the loss of mass in a nuclear reaction is greater than zero but less than 2mec2 the process cannot occur by positron emission, but occurs spontaneously for electron capture.
Common examples
Some common radionuclides that decay solely by electron capture include:
|
|
|
For a full list, see the table of nuclides.
See also
References
- ^
Cottingham, W.N.; Greenwood, D.A. (1986). An introduction to nuclear physics. ISBN 978-0-521-31960-7.
- ^ "The Reines-Cowan experiments: Detecting the poltergeist" (PDF). Los Alamos National Laboratory. 25: 3. 1997.
- ^ ISBN 978-0-226-81304-2– via archive.org.
- ^ "Luis Alvarez, biography". Nobel Prize. The Nobel Prize in Physics 1968. Retrieved 7 October 2009.
- .
- .
- .
- S2CID 250860726. Archived from the original(PDF) on 2013-12-26.
- S2CID 121883028.
External links
- "The LIVEChart of Nuclides". IAEA Nuclear Data Section. Vienna, Austria: International Atomic Energy Agency. Retrieved 16 August 2020. with filter on electron capture


