Endometrial cancer
| Endometrial cancer | |
|---|---|
| Other names | Uterine cancer |
| Prognosis | Five-year survival rate ~80% (US)[5] |
| Frequency | 3.8 million (total affected in 2015)[6] |
| Deaths | 89,900 (2015)[7] |
Endometrial cancer is a
Approximately 40% of cases are related to
The leading treatment option for endometrial cancer is
In 2012, endometrial cancers newly occurred in 320,000 women and caused 76,000 deaths.
Signs and symptoms
Vaginal bleeding or spotting in women after menopause occurs in 90% of endometrial cancer.[2][13][14] Bleeding is especially common with adenocarcinoma, occurring in two-thirds of all cases.[2][10] Abnormal menstrual cycles or extremely long, heavy, or frequent episodes of bleeding in women before menopause may also be a sign of endometrial cancer.[10]
Symptoms other than bleeding are not common. Other symptoms include thin white or clear
Risk factors
Risk factors for endometrial cancer include
Hormones
Most of the risk factors for endometrial cancer involve high levels of estrogens. An estimated 40% of cases are thought to be related to obesity.
Genetics

Women with a family history of endometrial cancer are at higher risk.[9] Two genes most commonly associated with some other women's cancers, BRCA1 and BRCA2, do not cause endometrial cancer. There is an apparent link with these genes but it is attributable to the use of tamoxifen, a drug that itself can cause endometrial cancer, in breast and ovarian cancers.[17] The inherited genetic condition Cowden syndrome can also cause endometrial cancer. Women with this disorder have a 5–10% lifetime risk of developing endometrial cancer,[3] compared to the 2–3% risk for unaffected women.[18]
Common genetic variation has also been found to affect endometrial cancer risk in large-scale genome-wide association studies.[28][29] Sixteen genomic regions have been associated with endometrial cancer and the common variants explain up to 7% of the familial relative risk.[29]
Other health problems
Some therapies for other forms of cancer increase the lifetime risk of endometrial cancer, which is a baseline 2–3%.
Low immune function has also been implicated in endometrial cancer.[15] High blood pressure is also a risk factor,[22] but this may be because of its association with obesity.[27] Sitting regularly for prolonged periods is associated with higher mortality from endometrial cancer. The risk is not negated by regular exercise, though it is lowered.[33]
Protective factors
Pathophysiology
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Endometrial cancer forms when there are errors in normal endometrial
In 10–20% of endometrial cancers, mostly Grade 3 (the highest

PTEN and
Type I and Type II cancers (explained below) tend to have different mutations involved. ARID1A, which often carries a
Development of an endometrial hyperplasia (overgrowth of endometrial cells) is a significant risk factor because hyperplasias can and often do develop into adenocarcinoma, though cancer can develop without the presence of a hyperplasia.[20] Within ten years, 8–30% of atypical endometrial hyperplasias develop into cancer, whereas 1–3% of non-atypical hyperplasias do so.[40] An atypical hyperplasia is one with visible abnormalities in the nuclei. Pre-cancerous endometrial hyperplasias are also referred to as endometrial intraepithelial neoplasia.[41] Mutations in the KRAS gene can cause endometrial hyperplasia and therefore Type I endometrial cancer.[37] Endometrial hyperplasia typically occurs after the age of 40.[9] Endometrial glandular dysplasia occurs with an overexpression of p53, and develops into a serous carcinoma.[15]
Diagnosis
Diagnosis of endometrial cancer is made first by a physical examination, endometrial biopsy, or dilation and curettage (removal of endometrial tissue; D&C). This tissue is then examined histologically for characteristics of cancer. If cancer is found, medical imaging may be done to see whether the cancer has spread or invaded tissues like the myometrium (the muscular wall of the womb) or the uterine cervix. A study from 2024 indicates that transvaginal ultrasound provides diagnostic performance comparable to magnetic resonance imaging regarding the myometrial infiltration assessment. However, magnetic resonance imaging showed significantly better specificity in low-grade endometrial cancer. [42]
Examination

Routine screening of asymptomatic people is not indicated since the disease is highly curable in its early, symptomatic stages. Instead, women, particularly menopausal women, should be aware of the symptoms and risk factors of endometrial cancer. A
Women with
Dilation and curettage or an endometrial biopsy are used to obtain a tissue sample for histological examination. Endometrial biopsy is the less invasive option, but it may not give conclusive results every time. Hysteroscopy only shows the gross anatomy of the endometrium, which is often not indicative of cancer, and is therefore not used, unless in conjunction with a biopsy.[44] Hysteroscopy can be used to confirm a diagnosis of cancer. New evidence shows that D&C has a higher false negative rate than endometrial biopsy.[22]
Before treatment is begun, several other investigations are recommended. These include a chest x-ray,
Classification
Endometrial cancers may be tumors derived from epithelial cells (carcinomas), mixed epithelial and mesenchymal tumors (carcinosarcomas), or mesenchymal tumors.[45]

Traditional classification of endometrial carcinomas is based either on clinical and endocrine features (Type I and Type II), or histopathological characteristics (endometrioid, serous, and clear-cell). Some tumors are difficult to classify and have features overlapping more than one category. High grade endometrioid tumors, in particular, tend to have both type I and type II features.[45]
Carcinoma
The vast majority of endometrial cancers are carcinomas (usually adenocarcinomas), meaning that they originate from the single layer of epithelial cells that line the endometrium and form the endometrial glands. There are many microscopic subtypes of endometrial carcinoma, but they are broadly organized into two categories, Type I and Type II, based on clinical features and pathogenesis. The two subtypes are genetically distinct.[10]
Type I endometrial carcinomas occur most commonly before and around the time of menopause. In the United States, they are more common in white women, particularly those with a history of endometrial hyperplasia. Type I endometrial cancers are often low-grade, minimally invasive into the underlying uterine wall (myometrium), estrogen-dependent, and have a good outcome with treatment.[10] Type I carcinomas represent 75–90% of endometrial cancer.[15][47]
Type II endometrial carcinomas usually occur in older, post-menopausal people, in the United States are more common in
Endometrioid adenocarcinoma
In
The genetic mutations most commonly associated with endometrioid adenocarcinoma are in the genes PTEN, a tumor suppressor; PIK3CA, a kinase; KRAS, a GTPase that functions in signal transduction; and CTNNB1, involved in adhesion and cell signaling. The CTNNB1 (beta-catenin) gene is most commonly mutated in the squamous subtype of endometrioid adenocarcinoma.[49]
Serous carcinoma
Serous carcinoma is a Type II endometrial tumor that makes up 5–10% of diagnosed endometrial cancer and is common in postmenopausal women with atrophied endometrium and black women. Serous endometrial carcinoma is aggressive and often invades the myometrium and metastasizes within the peritoneum (seen as
The genetic mutations seen in serous carcinoma are
Clear cell carcinoma
Clear cell carcinoma is a Type II endometrial tumor that makes up less than 5% of diagnosed endometrial cancer. Like serous cell carcinoma, it is usually aggressive and carries a poor prognosis. Histologically, it is characterized by the features common to all clear cells: the eponymous clear cytoplasm when H&E stained and visible, distinct cell membranes.[47] The p53 cell signaling system is not active in endometrial clear cell carcinoma.[15] This form of endometrial cancer is more common in postmenopausal women.[20]
Mucinous carcinoma
Mixed or undifferentiated carcinoma
Mixed carcinomas are those that have both Type I and Type II cells, with one making up at least 10% of the tumor.[48] These include the malignant mixed Müllerian tumor, which derives from endometrial epithelium and has a poor prognosis.[50]
Undifferentiated endometrial carcinomas make up less than 1–2% of diagnosed endometrial cancers. They have a worse prognosis than grade III tumors. Histologically, these tumors show sheets of identical epithelial cells with no identifiable pattern.[48]
Other carcinomas
Non-metastatic
Sarcoma

In contrast to endometrial carcinomas, the uncommon endometrial stromal
Metastasis
Endometrial cancer frequently metastasizes to the ovaries and Fallopian tubes
Histopathology

There is a three-tiered system for histologically classifying endometrial cancers, ranging from cancers with well-differentiated cells (grade I), to very poorly-differentiated cells (grade III).
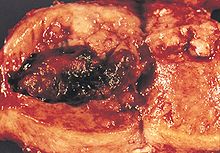
The histopathology of endometrial cancers is highly diverse. The most common finding is a well-differentiated endometrioid adenocarcinoma,[50] which is composed of numerous, small, crowded glands with varying degrees of nuclear atypia, mitotic activity, and stratification. This often appears on a background of endometrial hyperplasia. Frank adenocarcinoma may be distinguished from atypical hyperplasia by the finding of clear stromal invasion, or "back-to-back" glands which represent nondestructive replacement of the endometrial stroma by the cancer. With progression of the disease, the myometrium is infiltrated.[61]
-
A stage III endometrioid adenocarcinoma that has invaded the myometrium
-
Metastatic endometrial cancer seen in a removed lung
-
Grade 1: ≤5% solid non-glandular, non-squamous growth.[62]
-
Grade 2: >5% and ≤50% solid non-glandular, non-squamous growth.[62]
-
Grade 3: >50% solid non-glandular, non-squamous growth.[62]
-
Nuclear grade 1: Oval, mildly enlarged nucleus with evenly distributed chromatin.[63]
-
Nuclear grade 2: Intermediate features.[63]
-
Nuclear grade 3: Markedly enlarged and pleomorphic nuclei, with coarse chromatin and distinct nucleoli.[63]
-
Squamous growth seen as necrotic “ghost cells” of keratinocytes at right in image, leaving pink keratin.
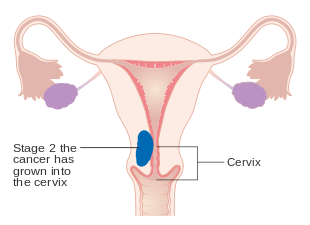
Staging
Endometrial carcinoma is surgically staged using the
| Stage | Description |
|---|---|
| IA | Tumor is confined to the uterus with less than half myometrial invasion |
| IB | Tumor is confined to the uterus with more than half myometrial invasion |
| II | Tumor involves the uterus and the cervical stroma
|
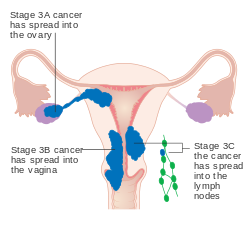
| IIIA | Tumor invades adnexa
|
| IIIB | Vaginal and/or parametrial involvement |
| IIIC1 | Pelvic lymph node involvement |
| IIIC2 | Para-aortic lymph node involvement, with or without pelvic node involvement |
| IVA | Tumor invades bladder mucosa and/or bowel mucosa |
| IVB | Distant metastases including abdominal metastases and/or inguinal lymph nodes
|
- The right panel shows high magnification on a positive area, confirming adenocarcinoma, as it shows tumor cells with large nuclei and prominent nucleoli.
Myometrial invasion and involvement of the pelvic and para-aortic lymph nodes are the most commonly seen patterns of spread.[2] A Stage 0 is sometimes included, in this case it is referred to as "carcinoma in situ".[9] In 26% of presumably early-stage cancers, intraoperative staging revealed pelvic and distant metastases, making comprehensive surgical staging necessary.[27]
-
Stage IA and IB endometrial cancer
-
Stage II endometrial cancer
-
Stage III endometrial cancer
-
Stage IV endometrial cancer
Management
Surgery

The initial treatment for endometrial cancer is surgery; 90% of women with endometrial cancer are treated with some form of surgery.
The few contraindications to surgery include inoperable tumor, massive obesity, a particularly high-risk operation, or a desire to preserve fertility.[68] These contraindications happen in about 5–10% of cases.[22] Women who wish to preserve their fertility and have low-grade stage I cancer can be treated with progestins, with or without concurrent tamoxifen therapy. This therapy can be continued until the cancer does not respond to treatment or until childbearing is done.[69] Uterine perforation may occur during a D&C or an endometrial biopsy.[70] Side effects of surgery to remove endometrial cancer can specifically include sexual dysfunction, temporary incontinence, and lymphedema, along with more common side effects of any surgery, including constipation.[9]
Add-on therapy
There are a number of possible additional therapies. Surgery can be followed by radiation therapy and/or chemotherapy in cases of high-risk or high-grade cancers. This is called adjuvant therapy.[16]
Chemotherapy
In cases where surgery is not indicated,
Low certainty evidence suggests that in women with recurrent endometrial cancer who have had chemotherapy, receiving drugs that inhibit the mTOR pathway may reduce the risk of disease worsening compared to having more chemotherapy or hormonal therapy. Though, mTOR inhibitors may increase the chance of experiencing digestive tract ulcers.[73]
Radiotherapy
Adjuvant radiotherapy is commonly used in early-stage (stage I or II) endometrial cancer. It can be delivered through vaginal brachytherapy (VBT), which is becoming the preferred route due to its reduced toxicity, or external beam radiotherapy (EBRT). Brachytherapy involves placing a radiation source in the organ affected; in the case of endometrial cancer a radiation source is placed directly in the vagina. External beam radiotherapy involves a beam of radiation aimed at the affected area from outside the body. VBT is used to treat any remaining cancer solely in the vagina, whereas EBRT can be used to treat remaining cancer elsewhere in the pelvis following surgery. However, the benefits of adjuvant radiotherapy are controversial. Though EBRT significantly reduces the rate of relapse in the pelvis, overall survival and metastasis rates are not improved.[2] VBT provides a better quality of life than EBRT.[22]
Radiotherapy can also be used before surgery in certain cases. When pre-operative imaging or clinical evaluation shows tumor invading the cervix, radiation can be given before a
Hormonal therapy
Hormonal therapy is only beneficial in certain types of endometrial cancer. It was once thought to be beneficial in most cases.
In 2010 hormonal therapy is of unclear effect in those with advanced or recurrent endometrial cancer.[76] There is insufficient evidence to inform women considering hormone replacement therapy after treatment for endometrial cancer.[77]
Targeted therapy
Dostarlimab has been approved by the FDA for therapy of endometrial cancer with specific biomarker[78]
Monitoring
The tumor marker CA-125 is frequently elevated in endometrial cancer and can be used to monitor response to treatment, particularly in serous cell cancer or advanced disease.[32][44][79] Periodic MRIs or CT scans may be recommended in advanced disease and women with a history of endometrial cancer should receive more frequent pelvic examinations for the five years following treatment.[79] Examinations conducted every three to four months are recommended for the first two years following treatment, and every six months for the next three years.[22]
Women with endometrial cancer should not have routine surveillance imaging to monitor the cancer unless new symptoms appear or
Prognosis
Survival rates
| Stage | 5-year survival rate |
|---|---|
| I-A | 88% |
| I-B | 75% |
| II | 69% |
| III-A | 58% |
| III-B | 50% |
| III-C | 47% |
| IV-A | 17% |
| IV-B | 15% |
The five-year survival rate for endometrial adenocarcinoma following appropriate treatment is 80%.
Recurrence rates
Recurrence of early stage endometrial cancer ranges from 3 to 17%, depending on primary and adjuvant treatment.[82] Most recurrences (75–80%) occur outside of the pelvis, and most occur within two to three years of treatment—64% within two years and 87% within three years.[58]
Higher-staged cancers are more likely to recur, as are those that have invaded the myometrium or cervix, or that have metastasized into the lymphatic system. Papillary serous carcinoma, clear cell carcinoma, and endometrioid carcinoma are the subtypes at the highest risk of recurrence.[23] High-grade histological subtypes are also at elevated risk for recurrence.[15]
The most common site of recurrence is in the vagina;[2] vaginal relapses of endometrial cancer have the best prognosis. If relapse occurs from a cancer that has not been treated with radiation, EBRT is the first-line treatment and is often successful. If a cancer treated with radiation recurs, pelvic exenteration is the only option for curative treatment. Palliative chemotherapy, cytoreductive surgery, and radiation are also performed.[88] Radiation therapy (VBT and EBRT) for a local vaginal recurrence has a 50% five-year survival rate. Pelvic recurrences are treated with surgery and radiation, and abdominal recurrences are treated with radiation and, if possible, chemotherapy.[22] Other common recurrence sites are the pelvic lymph nodes, para-aortic lymph nodes, peritoneum (28% of recurrences), and lungs, though recurrences can also occur in the brain (<1%), liver (7%), adrenal glands (1%), bones (4–7%; typically the axial skeleton), lymph nodes outside the abdomen (0.4–1%), spleen, and muscle/soft tissue (2–6%).[58]
Epidemiology
As of 2014[update], approximately 320,000 women are diagnosed with endometrial cancer worldwide each year and 76,000 die, making it the sixth most common cancer in women.[3] It is more common in developed countries, where the lifetime risk of endometrial cancer in women is 1.6%, compared to 0.6% in developing countries.[16] It occurs in 12.9 out of 100,000 women annually in developed countries.[23]
In the United States, endometrial cancer is the most frequently diagnosed gynecologic cancer and, in women, the fourth most common cancer overall,[11][20] representing 6% of all cancer cases in women.[89] In that country, as of 2014[update] it was estimated that 52,630 women were diagnosed yearly and 8,590 would die from the disease.[27] Northern Europe, Eastern Europe, and North America have the highest rates of endometrial cancer, whereas Africa and West Asia have the lowest rates. Asia saw 41% of the world's endometrial cancer diagnoses in 2012, whereas Northern Europe, Eastern Europe, and North America together comprised 48% of diagnoses.[3] Unlike most cancers, the number of new cases has risen in recent years, including an increase of over 40% in the United Kingdom between 1993 and 2013.[16] Some of this rise may be due to the increase in obesity rates in developed countries,[23] increasing life expectancies, and lower birth rates.[11] The average lifetime risk for endometrial cancer is approximately 2–3% in people with uteruses.[18] In the UK, approximately 7,400 cases are diagnosed annually, and in the EU, approximately 88,000.[22]
Endometrial cancer appears most frequently during
Research
There are several experimental therapies for endometrial cancer under research, including immunologic, hormonal, and chemotherapeutic treatments. Trastuzumab (Herceptin), an antibody against the Her2 protein, has been used in cancers known to be positive for the Her2/neu oncogene, but research is still underway. Immunologic therapies are also under investigation, particularly in uterine papillary serous carcinoma.[37]
Cancers can be analyzed using genetic techniques (including DNA sequencing and immunohistochemistry) to determine if certain therapies specific to mutated genes can be used to treat it. PARP inhibitors are used to treat endometrial cancer with PTEN mutations,[3] specifically, mutations that lower the expression of PTEN. The PARP inhibitor shown to be active against endometrial cancer is olaparib. Research is ongoing in this area as of the 2010s.[26][90][91]
Research is ongoing on the use of metformin, a diabetes medication, in obese women with endometrial cancer before surgery. Early research has shown it to be effective in slowing the rate of cancer cell proliferation.[21][36] Preliminary research has shown that preoperative metformin administration can reduce expression of tumor markers. Long-term use of metformin has not been shown to have a preventative effect against developing cancer, but may improve overall survival.[21]
Temsirolimus, an mTOR inhibitor, is under investigation as a potential treatment.[22] Research shows that mTOR inhibitors may be particularly effective for cancers with mutations in PTEN.[3] Ridaforolimus (deforolimus) is also being researched as a treatment for people who have previously had chemotherapy. Preliminary research has been promising, and a stage II trial for ridaforolimus was completed by 2013.[22] There has also been research on combined ridaforolimus/progestin treatments for recurrent endometrial cancer.[92] Bevacizumab and tyrosine kinase inhibitors, which inhibit angiogenesis, are being researched as potential treatments for endometrial cancers with high levels of vascular endothelial growth factor.[3] Ixabepilone is being researched as a possible chemotherapy for advanced or recurrent endometrial cancer.[92] Treatments for rare high-grade undifferentiated endometrial sarcoma are being researched, as there is no established standard of care yet for this disease. Chemotherapies being researched include doxorubicin and ifosfamide.[55]
There is also research in progress on more genes and biomarkers that may be linked to endometrial cancer. The protective effect of combined oral contraceptives and the IUD is being investigated. Preliminary research has shown that the levonorgestrel IUD placed for a year, combined with six monthly injections of gonadotropin-releasing hormone, can stop or reverse the progress of endometrial cancer in young women; specifically complex atypical hyperplasia however the results have been inconclusive.[93][94] An experimental drug that combines a hormone with doxorubicin is also under investigation for greater efficacy in cancers with hormone receptors. Hormone therapy that is effective in treating breast cancer, including use of aromatase inhibitors, is also being investigated for use in endometrial cancer. One such drug is anastrozole, which is currently being researched in hormone-positive recurrences after chemotherapy.[92] Research into hormonal treatments for endometrial stromal sarcomas is ongoing as well. It includes trials of drugs like mifepristone, a progestin antagonist, and aminoglutethimide and letrozole, two aromatase inhibitors.[54]
Research continues into the best imaging method for detecting and staging endometrial cancer. As current diagnostic methods are invasive and inaccurate, researchers are looking into new ways to catch endometrial cancer, especially in its early stages. A study found that using a technique involving
There is not enough evidence to determine if people with endometrial cancer benefit from additional behavioural and life style interventions that are aimed at losing excess weight.[97]
History and culture
Endometrial cancer is not widely known by the general populace, despite its frequency. There is low awareness of the symptoms, which can lead to later diagnosis and worse survival.[98]
References
- ^ a b c d e f g h i "General Information About Endometrial Cancer". National Cancer Institute. 22 April 2014. Archived from the original on 3 September 2014. Retrieved 3 September 2014.
- ^ PMID 22513918.
- ^ ISBN 978-92-832-0429-9.
- ^ a b c d e f g "Endometrial Cancer Treatment (PDQ®)". National Cancer Institute. 23 April 2014. Archived from the original on 3 September 2014. Retrieved 3 September 2014.
- ^ a b "SEER Stat Fact Sheets: Endometrial Cancer". National Cancer Institute. Archived from the original on 6 July 2014. Retrieved 18 June 2014.
- PMID 27733282.
- PMID 27733281.
- ^ "Defining Cancer". National Cancer Institute. 17 September 2007. Archived from the original on 25 June 2014. Retrieved 10 June 2014.
- ^ a b c d e f g h i "What You Need To Know: Endometrial Cancer". NCI. National Cancer Institute. Archived from the original on 8 August 2014. Retrieved 6 August 2014.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ "Uterine Cancer - Cancer Stat Facts". SEER. Retrieved 3 February 2019.
- ^ ISBN 978-0-07-163515-8.
- PMID 30083701.
- ^ S2CID 206893378.
- ^ PMID 24832785.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ PMID 25032011.
- PMID 35276057.
- ^ ISBN 978-0-323-06986-1.
- ^ PMID 25013215.
- ^ PMID 24078661.
- ^ PMID 22895938.
- from the original on 16 December 2014.
- PMID 37168552.
- ^ PMID 24098868.
- ^ PMID 24905773.
- PMID 27135401.
- ^ PMID 30093612.
- PMID 23076938.
There is evidence from other reports that tamoxifen increases the risk of endometrial cancer although the data presented in this review describes only 10 events occurring in 1798 participants (0.5%) after seven years of follow-up.
- ^ a b c d "Endometrial Cancer Prevention". PDQ. NIH. 28 February 2014. Archived from the original on 5 January 2015.
- ^ ISBN 978-0-323-06986-1.
- S2CID 7256176.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- PMID 35219087.
- ^ PMID 25045437.
- ^ ISBN 978-0-323-06986-1.
- license.
- PMID 25058385.
A recent exome-sequencing study revealed that 8% of serious endometrial cancers and 9% of clear cell endometrial cancers have SPOP mutations
- PMID 30521671.
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ Madár, I.; Szabó, A.; Vleskó, G.; Hegyi, P.; Ács, N.; Fehérvári, P.; Kói, T.; Kálovics, E.; Szabó, G. Diagnostic Accuracy of Transvaginal Ultrasound and Magnetic Resonance Imaging for the Detection of Myometrial Infiltration in Endometrial Cancer: A Systematic Review and Meta-Analysis. Cancers 2024, 16, 907. https://doi.org/10.3390/cancers16050907
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ PMID 24872110.
- PMID 19078929.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ PMID 21908501.
- ^ PMID 21975736.
- S2CID 205746422.
- PMID 18620731.
- PMID 16990716.
- ^ S2CID 26057966.
- ^ PMID 24451763.
- PMID 19853898.
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ PMID 23545091.
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- PMID 22496699.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ISBN 978-0-7216-7253-3.
- ^ PMID 30550484.
- ^ PMID 22496699.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ "Stage Information for Endometrial Cancer". National Cancer Institute. January 1980. Archived from the original on 6 April 2014. Retrieved 23 April 2014.
- .
- ^ PMID 28968482.
- ^ PMID 30379327.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ISBN 978-0-323-06986-1.
- ^ ISBN 978-0-323-06986-1.
- PMID 25017099.
- PMID 31588998.
- PMID 21678331.
- PMID 25406186.
- PMID 21154390.
- ISSN 1361-259X.
- FDA. 22 April 2021.
- ^ ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ "Five Things Physicians and Patients Should Question". Choosing Wisely. Society of Gynecologic Oncology. 31 October 2013. Archived from the original on 1 December 2013. Retrieved 27 July 2014.
- ^ "Survival by stage of endometrial cancer". American Cancer Society. 2 March 2014. Archived from the original on 31 July 2014. Retrieved 10 June 2014.
- ^ PMID 23435365.
- PMID 24497188.
- ISBN 978-0-323-06986-1.
- PMID 22507532.
- PMID 19406460.
- PMID 26011914.
- ISBN 978-0-07-171672-7. Archivedfrom the original on 4 January 2014.
- ^ "General Information about Endometrial Cancer". Endometrial Cancer Treatment (PDQ). NIH. 23 April 2014. Archived from the original on 20 June 2014.
- PMID 24225019.
- S2CID 3079985.
- ^ a b c d "Womb cancer research". CancerHelp UK. Cancer Research UK. Archived from the original on 22 August 2014. Retrieved 31 August 2014.
- PMID 20876910.
- PMID 29215513.
- S2CID 241679195.
- PMID 32429365.
- PMID 36971688.
- ^ Carlisle D (21 September 2014). "Womb cancer: the most common diagnosis you've never heard of". The Guardian. Archived from the original on 22 September 2014. Retrieved 29 September 2014.



![Grade 1: ≤5% solid non-glandular, non-squamous growth.[62]](http://upload.wikimedia.org/wikipedia/commons/thumb/5/52/Histopathology_of_FIGO_%28architectural%29_grade_1_endometrial_adenocarcinoma.png/120px-Histopathology_of_FIGO_%28architectural%29_grade_1_endometrial_adenocarcinoma.png)
![Grade 2: >5% and ≤50% solid non-glandular, non-squamous growth.[62]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/28/FIGO_grade_2_endometrial_adenocarcinoma.jpg/120px-FIGO_grade_2_endometrial_adenocarcinoma.jpg)
![Grade 3: >50% solid non-glandular, non-squamous growth.[62]](http://upload.wikimedia.org/wikipedia/commons/thumb/4/45/FIGO_grade_3_endometrial_adenocarcinoma.jpg/120px-FIGO_grade_3_endometrial_adenocarcinoma.jpg)
![Nuclear grade 1: Oval, mildly enlarged nucleus with evenly distributed chromatin.[63]](http://upload.wikimedia.org/wikipedia/commons/thumb/f/fa/Histopathology_of_endometrioid_cancer%2C_grade_1%2C_nuclear_grade_1.jpg/120px-Histopathology_of_endometrioid_cancer%2C_grade_1%2C_nuclear_grade_1.jpg)
![Nuclear grade 2: Intermediate features.[63]](http://upload.wikimedia.org/wikipedia/commons/thumb/1/11/Histopathology_of_endometrioid_cancer%2C_grade_1%2C_nuclear_grade_2.jpg/120px-Histopathology_of_endometrioid_cancer%2C_grade_1%2C_nuclear_grade_2.jpg)
![Nuclear grade 3: Markedly enlarged and pleomorphic nuclei, with coarse chromatin and distinct nucleoli.[63]](http://upload.wikimedia.org/wikipedia/commons/thumb/c/cf/Endometrial_endometrioid_adenocarcinoma%2C_nuclear_and_architectural_grade_3.jpg/120px-Endometrial_endometrioid_adenocarcinoma%2C_nuclear_and_architectural_grade_3.jpg)