Endomorphin-1
| |

| |
| Names | |
|---|---|
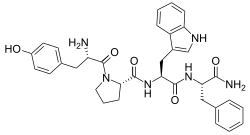
| IUPAC name
L-Tyrosyl-L-prolyl-L-tryptophyl-L-phenylalaninamide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
IUPHAR/BPS |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C34H38N6O5 | |
| Molar mass | 610.703 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Endomorphin-1 (EM-1) (
By combining N-terminal guadino modifications, a new class of endonmorphin-1 was synthesized, the range of their bioactivities were measured by radioligand binding assay in order to conclude its potency as an opioid.[6] Endomorphin-1 has high affinity and specificity for opioid receptors for behavioral, physiological and pharmacological assays, it is also a potent analgesic agent which brings effects on cardiovascular, respiratory and gastrointestinal functions as well as in immune system responses.[citation needed] This endogenous opioid peptide can help with neuropathic pain without having the common side effects many neuropathic drugs caused which produces constipation. To make this drug side effect-free, a modification at the N-terminus by 2-aminodecainoic acid is made which in term showed an improve in the drug's metabolic stability along with improving its membrane permeability, while holding its high receptor binding affinity, helping the drug act as a potent agonist[7]
See also
References
- ^ ISBN 978-0-8018-6827-6.
- ISBN 978-0-7923-8757-2.
- ^ ISBN 978-3-7643-5875-4.
- ^ ISBN 978-3-540-38916-3.
- PMID 17964607.
- ^ "BrowZine". browzine.com. Retrieved 2021-11-02.
- PMID 22912681.
