Lipopolysaccharide

Lipopolysaccharides (LPS) are large
Lipopolysaccharides can have substantial impacts on human health, primarily through interactions with the immune system. LPS is a potent activator of the immune system and
Discovery
The toxic activity of LPS was first discovered and termed endotoxin by
Functions in bacteria
LPS is a major component of the outer membrane of
Composition
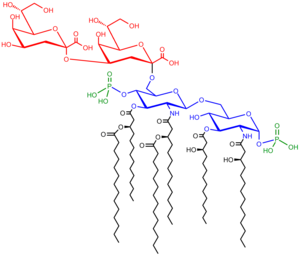
Lipopolysaccharides are composed of three parts: the O antigen (or O polysaccharide), the core oligosaccharide, and Lipid A.
O-antigen
The repetitive
Core
The core domain always contains an oligosaccharide component that attaches directly to lipid A and commonly contains sugars such as heptose and 3-Deoxy-D-manno-oct-2-ulosonic acid (also known as KDO, keto-deoxyoctulosonate).[17] The LPS cores of many bacteria also contain non-carbohydrate components, such as phosphate, amino acids, and ethanolamine substituents.
Lipid A
Lipid A is, in normal circumstances, a
Lipooligosaccharides
The "rough form" of LPS has a lower molecular weight due to the absence of the O polysaccharide. In its place is a short oligosaccharide: this form is known as Lipooligosaccharide (LOS), and is a glycolipid found in the outer membrane of some types of
LPS detoxification
A highly conserved host enzyme called acyloxyacyl hydrolase (AOAH) may detoxify LPS when it enters, or is produced in, animal tissues. It may also convert LPS in the intestine into an LPS inhibitor. Neutrophils, macrophages and dendritic cells produce this lipase, which inactivates LPS by removing the two secondary acyl chains from lipid A to produce tetraacyl LPS. If mice are given LPS parenterally, those that lack AOAH develop high titers of non-specific antibodies, develop prolonged hepatomegaly, and experience prolonged endotoxin tolerance. LPS inactivation may be required for animals to restore homeostasis after parenteral LPS exposure.[21] Although mice have many other mechanisms for inhibiting LPS signaling, none is able to prevent these changes in animals that lack AOAH.
Dephosphorylation of LPS by
Biosynthesis and transport
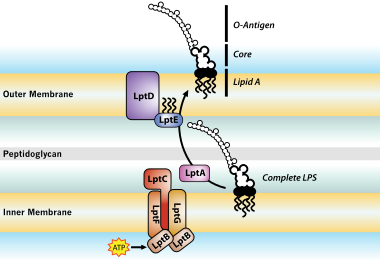
The entire process of making LPS starts with a molecule called lipid A-Kdo2, which is first created on the surface of the bacterial cell's inner membrane. Then, additional sugars are added to this molecule on the inner membrane before it's moved to the space between the inner and outer membranes (
Ultimately, LPS is transported to the outer membrane by a membrane-to-membrane bridge of lipolysaccharide transport (Lpt) proteins.[27][29] This transporter is a potential antibiotic target.[30][31]
Biological effects on hosts infected with Gram-negative bacteria
Immune response
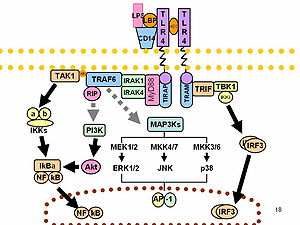
LPS acts as the prototypical endotoxin because it binds the
As part of the cellular
LPS function has been under experimental research for several years due to its role in activating many transcription factors. LPS also produces many types of mediators involved in septic shock. Humans are much more sensitive to LPS than other animals (e.g., mice). A dose of 1 µg/kg induces shock in humans, but mice will tolerate a dose up to a thousand times higher.[36] This may relate to differences in the level of circulating natural antibodies between the two species.[37][38] Said et al. showed that LPS causes an
Endotoxins are in large part responsible for the dramatic clinical manifestations of infections with pathogenic Gram-negative bacteria, such as
Portions of the LPS from several bacterial strains have been shown to be chemically similar to human host cell surface molecules; the ability of some bacteria to present molecules on their surface which are chemically identical or similar to the surface molecules of some types of host cells is termed molecular
LPS can be sensed directly by hematopoietic stem cells (HSCs) through the bonding with TLR4, causing them to proliferate in reaction to a systemic infection. This response activate the TLR4-TRIF-ROS-p38 signaling within the HSCs and through a sustained TLR4 activation can cause a proliferative stress, leading to impair their competitive repopulating ability.[41] Infection in mice using S. typhimurium showed similar results, validating the experimental model also in vivo.
Effect of variability on immune response
O-antigens (the outer carbohydrates) are the most variable portion of the LPS molecule, imparting antigenic specificity. In contrast, lipid A is the most conserved part. However, lipid A composition also may vary (e.g., in number and nature of
It has been speculated that conical lipid A (e.g., from
Normal human blood serum contains anti-LOS antibodies that are bactericidal and patients that have infections caused by serotypically distinct strains possess anti-LOS antibodies that differ in their specificity compared with normal serum.[47] These differences in humoral immune response to different LOS types can be attributed to the structure of the LOS molecule, primarily within the structure of the oligosaccharide portion of the LOS molecule.[47]
In
Non-canonical pathways of LPS recognition
Recently, it was shown that in addition to
Testing
Lipopolysaccharide is a significant factor that makes bacteria harmful, and it helps categorize them into different groups based on their structure and function. This makes LPS a useful marker for telling apart various Gram-negative bacteria. Swiftly identifying and understanding the types of pathogens involved is crucial for promptly managing and treating infections. Since LPS is the main trigger for the immune response in our cells, it acts as an early signal of an acute infection. Therefore, LPS testing is more specific and meaningful than many other serological tests.[54]
The current methods for testing LPS are quite sensitive, but many of them struggle to differentiate between different LPS groups. Additionally, the nature of LPS, which has both water-attracting and water-repelling properties (amphiphilic), makes it challenging to develop sensitive and user-friendly tests.[54]
The typical detection methods rely on identifying the lipid A part of LPS. However, this method has limitations because Lipid A is very similar among different bacterial species and serotypes. LPS testing techniques fall into six categories, and they often overlap: in vivo tests, in vitro tests, modified immunoassays, biological assays, and chemical assays.[54]
Pathophysiology
LPS is a powerful toxin that, when in the body, triggers inflammation by binding to cell receptors. Excessive LPS in the blood can lead to endotoxemia, potentially causing a harmful condition called septic shock. This condition includes symptoms like rapid heart rate, quick breathing, temperature changes, and blood clotting issues, resulting in blood vessels widening and reduced blood volume, leading to cellular dysfunction.[54]
Recent research indicates that even small LPS exposure is associated with autoimmune diseases and allergies. High levels of LPS in the blood can lead to metabolic syndrome, increasing the risk of conditions like diabetes, heart disease, and liver problems.[54]
LPS also plays a crucial role in symptoms caused by infections from harmful bacteria, including severe conditions like Waterhouse-Friderichsen syndrome, meningococcemia, and meningitis. Certain bacteria can adapt their LPS to cause long-lasting infections in the respiratory and digestive systems.[54]
Recent studies have shown that LPS disrupts cell membrane lipids, affecting cholesterol and metabolism, potentially leading to high cholesterol, abnormal blood lipid levels, and non-alcoholic fatty liver disease. In some cases, LPS can interfere with toxin clearance, which may be linked to neurological issues.[54]
Health effects
In general the health effects of LPS are due to its abilities as a potent activator and modulator of the immune system, especially its inducement of inflammation.
Endotoxemia
The presence of endotoxins in the blood is called endotoxemia. High level of endotoxemia can lead to septic shock,[55] while lower concentration of endotoxins in the bloodstream is called metabolic endotoxemia.[56] Endotoxemia is associated with obesity, diet,[57] cardiovascular diseases,[57] and diabetes,[56] while also host genetics might have an effect.[58]
Moreover, endotoxemia of intestinal origin, especially, at the
It has also been reported that gonococcal LOS can cause damage to human fallopian tubes.[47]
Auto-immune disease
The molecular mimicry of some LOS molecules is thought to cause autoimmune-based host responses, such as flareups of multiple sclerosis.[6][40] Other examples of bacterial mimicry of host structures via LOS are found with the bacteria Helicobacter pylori and Campylobacter jejuni, organisms which cause gastrointestinal disease in humans, and Haemophilus ducreyi which causes chancroid. Certain C. jejuni LPS serotypes (attributed to certain tetra- and pentasaccharide moieties of the core oligosaccharide) have also been implicated with Guillain–Barré syndrome and a variant of Guillain–Barré called Miller-Fisher syndrome.[6]
Link to obesity
Epidemiological studies have shown that increased endotoxin load, which can be a result of increased populations of endotoxin-producing bacteria in the intestinal tract, is associated with certain obesity-related patient groups.
Depression
There is experimental and observational evidence that LPS might play a role in depression. Administration of LPS in mice can lead to depressive symptoms, and there seem to be elevated levels of LPS in some people with depression. Inflammation may sometimes play a role in the development of depression, and LPS is pro-inflammatory.[8]
Cellular senescence
Inflammation induced by LPS can induce
Role as contaminant in biotechnology and research
Lipopolysaccharides are frequent contaminants in plasmid DNA prepared from bacteria or proteins expressed from bacteria, and must be removed from the DNA or protein to avoid contaminating experiments and to avoid toxicity of products manufactured using industrial fermentation.[66]
Ovalbumin is frequently contaminated with endotoxins. Ovalbumin is one of the extensively studied proteins in animal models and also an established model allergen for airway hyper-responsiveness (AHR). Commercially available ovalbumin that is contaminated with LPS can falsify research results, as it does not accurately reflect the effect of the protein antigen on animal physiology.[67]
In pharmaceutical production, it is necessary to remove all traces of endotoxin from drug product containers, as even small amounts of endotoxin will cause illness in humans. A depyrogenation oven is used for this purpose. Temperatures in excess of 300 °C are required to fully break down LPS.[68]
The standard
- Testing
- Lipopolysaccharide, is a significant factor that makes bacteria harmful, and it helps categorize them into different groups based on their structure and function. This makes LPS a useful marker for telling apart various Gram-negative bacteria. Swiftly identifying and understanding the types of pathogens involved is crucial for promptly managing and treating infections. Since LPS is the main trigger for the immune response in our cells, it acts as an early signal of an acute infection. Therefore, LPS testing is more specific and meaningful than many other serological tests. The current methods for testing LPS are quite sensitive, but many of them struggle to differentiate between different LPS groups. Additionally, the nature of LPS, which has both water-attracting and water-repelling properties (amphiphilic), makes it challenging to develop sensitive and user-friendly tests. The typical detection methods rely on identifying the lipid A part of LPS. However, this method has limitations because Lipid A is very similar among different bacterial species and serotypes. LPS testing techniques fall into six categories, and they often overlap: in vivo tests, in vitro tests, modified immunoassays, biological assays, and chemical assays. [54]
- Pathophysiology
- LPS is a powerful toxin that, when in the body, triggers inflammation by binding to cell receptors. Excessive LPS in the blood can lead to endotoxemia, potentially causing a harmful condition called septic shock. This condition includes symptoms like rapid heart rate, quick breathing, temperature changes, and blood clotting issues, resulting in blood vessels widening and reduced blood volume, leading to cellular dysfunction. Recent research indicates that even small LPS exposure is associated with autoimmune diseases and allergies. High levels of LPS in the blood can lead to metabolic syndrome, increasing the risk of conditions like diabetes, heart disease, and liver problems. LPS also plays a crucial role in symptoms caused by infections from harmful bacteria, including severe conditions like Waterhouse-Friderichsen syndrome, meningococcemia, and meningitis. Certain bacteria can adapt their LPS to cause long-lasting infections in the respiratory and digestive systems. Recent studies have shown that LPS disrupts cell membrane lipids, affecting cholesterol and metabolism, potentially leading to high cholesterol, abnormal blood lipid levels, and non-alcoholic fatty liver disease. In some cases, LPS can interfere with toxin clearance, which may be linked to neurological issues.[54]
See also
- Bioaerosol
- Depyrogenation
- Host-pathogen interface
- Mucopolysaccharide
- Nesfatin-1
- Schwartzman reaction
- AOAH
References
- ^ PMID 33746909.
- S2CID 28156137.
- PMID 3023091.
- ^ PMID 25428854.
- S2CID 34855187.
- ^ PMID 8988391.
- ^ S2CID 218665128.
- ^ S2CID 51924586.
- ISBN 978-8131221631.
- PMID 20825345.
- PMID 24148302.
- PMID 15293449.
- PMID 12045108.
- PMID 12960272.
- S2CID 2742306.
- PMID 4296687.
- PMID 11948150.
- S2CID 49481716.
- ^ PMID 23165926.
- PMID 19755182.
- PMID 28316376.
- PMID 18078689.
- PMID 23598380.
- PMID 24506153.
- PMID 19815028.
- ^ PMID 19633680.
- PMID 36543281.
- PMID 29449493.
- PMID 38172635.
- PMID 38172634.
- ISBN 978-1-4160-2974-8.
- PMID 9851930.
- ^ "The 2011 Nobel Prize in Physiology or Medicine - Press Release". www.nobelprize.org. Archived from the original on 23 March 2018. Retrieved 28 April 2018.
- PMID 31212769.
- PMID 20001600.
- PMID 9218618.
- PMID 9858525.
- PMID 20208540.
- ^ PMID 22168423.
- PMID 28736216.
- S2CID 36784237.
- PMID 11864841.
- PMID 10806403.
- PMID 12014174. 264.
- PMID 15473911.
- ^ PMID 7982947.
- PMID 11101573.
- PMID 30103489.
- PMID 24445575.
- PMID 27296646.
- S2CID 13681499.
- PMID 29057902.
- ^ PMID 35155966.
- PMID 20519895.
- ^ PMID 28183445.
- ^ S2CID 24020127.
- PMID 34668383.
- PMID 17016141.
- PMID 10845660.
- ^ S2CID 16951072.
- S2CID 12136094.
- S2CID 44494003.
- ^ PMID 17456850.
- PMID 23235292.
- PMID 7779915.
- PMID 12909619.
- ^ Komski L (16 December 2014). "The Detection of Endotoxins Via the LAL Test, the Chromogenic Method". Wako Chemicals USA, Inc. Archived from the original on 29 March 2015. Retrieved 14 March 2015.
- PMID 24019589.
- PMID 11451451. Archived from the original(PDF) on 2 January 2014. Retrieved 2 January 2014.
External links
- Lipopolysaccharides at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
