Enfuvirtide
 | ||
| Clinical data | ||
|---|---|---|
| Trade names | Fuzeon | |
| AHFS/Drugs.com | Monograph | |
| MedlinePlus | a603023 | |
| License data |
| |
| Pregnancy category |
| |
Subcutaneous | ||
| ATC code | ||
| Legal status | ||
| Legal status | ||
JSmol) | ||
| ||
| ||
| | ||
Enfuvirtide (
Medical uses
Enfuvirtide is
Adverse effects
Common
Pharmacology
Mechanism of action
Enfuvirtide works by disrupting the
HIV binds to the host CD4+ cell receptor via the viral protein gp120; gp41, a viral transmembrane protein, then undergoes a conformational change that assists in the fusion of the viral membrane to the host cell membrane. Enfuvirtide binds to gp41 preventing the creation of an entry pore for the capsid of the virus, keeping it out of the cell.[3]
Enfuvirtide is also an activator of the chemotactic factor receptor, formyl peptide receptor 1, and thereby activates phagocytes and presumably other cells bearing this receptor (see
Microbiology
Enfuvirtide is considered to be active against HIV-1 only. Low activity against HIV-2 isolates has been demonstrated in vitro.[5]
Variable susceptibility to enfuvirtide has been observed in clinical isolates, with
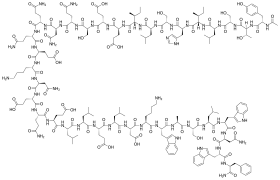
Structural formula
Enfuvirtide is a 36-amino acid peptide with the following sequence:[7][8]
(Ac-YTSLIHSLIEESQNQQEKNEQELLELDKWASLWNWF-NH2)History
Enfuvirtide originated at
References
- ^ a b "Product". guildlink.com.au. Archived from the original on 8 January 2023. Retrieved 8 January 2023.
- ^ ISBN 0-9757919-2-3.
- S2CID 32014873.
- PMID 10339497.
- ^ Roche Products Pty Ltd. Fuzeon (Australian Approved Product Information). Dee Why (NSW): Roche; 2005.
- PMID 15231762.
- PMID 7786578.
- PMID 7543152.
- ^ "Drugs@FDA: FDA Approved Drug Products – Fuzeon (Click on 'Approval Date(s) and History, Letters, Labels, Reviews for NDA 021481')". accessdata.fda.gov. United States Food and Drug Administration. Archived from the original on 28 June 2017. Retrieved 6 January 2019.
