Environmental radioactivity
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
| Part of a series on |
| Pollution |
|---|
 |
Environmental
Background level in soils
Radioactivity is
Sea and river silt
A recent report on the Sava river in Serbia suggests that many of the river silts contain about 100 Bq kg−1 of natural radioisotopes (226Ra, 232Th, and 238U).[2] According to the United Nations the normal concentration of uranium in soil ranges between 300 μg kg−1 and 11.7 mg kg−1.[3] It is well known that some plants, called hyperaccumulators, are able to absorb and concentrate metals within their tissues; iodine was first isolated from seaweed in France, which suggests that seaweed is an iodine hyperaccumulator.
Synthetic radioisotopes also can be detected in silt. Busby[citation needed] quotes a report on the plutonium activity in Welsh intertidal sediments by Garland et al. (1989), which suggests that the closer a site is to Sellafield, the higher is the concentration of plutonium in the silt. Some relationship between distance and activity can be seen in their data, when fitted to an exponential curve, but the scatter of the points is large (R2 = 0.3683).
Man-made
The additional radioactivity in the biosphere caused by human activity due to the releases of man-made radioactivity and of Naturally Occurring Radioactive Materials (NORM) can be divided into several classes.
- Normal licensed releases which occur during the regular operation of a plant or process handling man-made radioactive materials.
- For instance the release of 99Tc from a nuclear medicine department of a hospital which occurs when a person given a Tc imaging agent expels the agent.
- Releases of man-made radioactive materials which occur during an industrial or research accident.
- For instance the Chernobyl accident.
- For instance the
- Releases which occur as a result of military activity.
- For example, a nuclear weapons test.
- Releases which occur as a result of a crime.
- For example, the Goiânia accident where thieves, unaware of its radioactive content, stole some medical equipment and as a result a number of people were exposed to radiation.
- Releases of naturally occurring radioactive materials (NORM) as a result of mining etc.
- For example, the release of the trace quantities of uranium and thorium in coal, when it is burned in power stations.
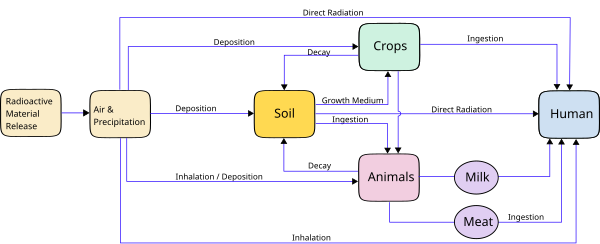
Farming and the transfer to humans of deposited radioactivity
Just because a radioisotope lands on the surface of the soil, does not mean it will enter the human food chain. After release into the environment, radioactive materials can reach humans in a range of different routes, and the chemistry of the element usually dictates the most likely route.
Cows
Jiří Hála claims in his textbook "Radioactivity, Ionizing Radiation and Nuclear Energy" [4] that cattle only pass a minority of the strontium, caesium, plutonium and americium they ingest to the humans who consume milk and meat. Using milk as an example, if the cow has a daily intake of 1000 Bq of the preceding isotopes then the milk will have the following activities.
- 90Sr, 2 Bq/L
- 137Cs, 5 Bq/L
- 239Pu, 0.001 Bq/L
- 241Am, 0.001 Bq/L
Soil
Jiří Hála's
The Trinity test

One dramatic source of man-made radioactivity is a
The 152Eu (half life 13.54 year) and 154Eu (half life 8.59 year) were mainly formed by the neutron activation of the europium in the soil, it is clear that the level of radioactivity for these isotopes is highest where the neutron dose to the soil was larger. Some of the 60Co (half life 5.27 year) is generated by activation of the cobalt in the soil, but some was also generated by the activation of the cobalt in the steel (100 foot) tower. This 60Co from the tower would have been scattered over the site reducing the difference in the soil levels.
The
The 137Cs level is higher in the sample that was further away from the ground zero point – this is thought to be because the precursors to the 137Cs (137I and 137Xe) and, to a lesser degree, the caesium itself are volatile. The natural radioisotopes in the glass are about the same in both locations.

Activation products
The action of
Nuclear bomb tests have increased the specific activity of carbon, whereas the use of fossil fuels has decreased it. See the article on radiocarbon dating for further details.
Fission products
Discharges from nuclear plants within the nuclear fuel cycle introduce fission products to the environment. The releases from nuclear reprocessing plants tend to be medium to long-lived radioisotopes; this is because the nuclear fuel is allowed to cool for several years before being dissolved in the nitric acid. The releases from nuclear reactor accidents and bomb detonations will contain a greater amount of the short-lived radioisotopes (when the amounts are expressed in activity Bq)).
Short lived

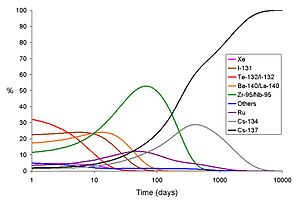
An example of a short-lived fission product is iodine-131, this can also be formed as an activation product by the neutron activation of tellurium.
In both bomb fallout and a release from a power reactor accident, the short-lived isotopes cause the dose rate on day one to be much higher than that which will be experienced at the same site many days later. This holds true even if no attempts at decontamination are made. In the graphs below, the total gamma dose rate and the share of the dose due to each main isotope released by the Chernobyl accident are shown.
Medium lived
An example of a medium lived is 137Cs, which has a half-life of 30 years. Caesium is released in bomb fallout and from the nuclear fuel cycle. A paper has been written on the radioactivity in oysters found in the Irish Sea, these were found by gamma spectroscopy to contain 141Ce, 144Ce, 103Ru, 106Ru, 137Cs, 95Zr and 95Nb.[citation needed] In addition, a zinc activation product (65Zn) was found, this is thought to be due to the corrosion of magnox fuel cladding in cooling ponds.[6] The concentration of all these isotopes in the Irish Sea attributable to nuclear facilities such as Sellafield has significantly decreased in recent decades.
An important part of the Chernobyl release was the caesium-137, this isotope is responsible for much of the long term (at least one year after the fire) external exposure which has occurred at the site. The caesium isotopes in the fallout have had an effect on farming. [2]
A large amount of caesium was released during the Goiânia accident where a radioactive source (made for medical use) was stolen and then smashed open during an attempt to convert it into scrap metal. The accident could have been stopped at several stages; first, the last legal owners of the source failed to make arrangements for the source to be stored in a safe and secure place; and second, the scrap metal workers who took it did not recognise the markings which indicated that it was a radioactive object.
Soudek et al. reported in 2006 details of the uptake of 90Sr and 137Cs into
Caesium binds tightly to
In
Long lived
Examples of long-lived isotopes include iodine-129 and Tc-99, which have nuclear half-lives of 15 million and 200,000 years, respectively.
Plutonium and the other actinides
In popular culture, plutonium is credited with being the ultimate threat to life and limb which is wrong; while ingesting plutonium is not likely to be good for one's health, other radioisotopes such as radium are more toxic to humans. Regardless, the introduction of the transuranium elements such as plutonium into the environment should be avoided wherever possible. Currently, the activities of the nuclear reprocessing industry have been subject to great debate as one of the fears of those opposed to the industry is that large amounts of plutonium will be either mismanaged or released into the environment.
In the past, one of the largest releases of plutonium into the environment has been
- Those tests in the air scattered some plutonium over the entire globe; this great dilution of the plutonium has resulted in the threat to each exposed person being very small as each person is only exposed to a very small amount.
- The underground tests tend to form molten rock, which rapidly cools and seals the actinides into the rock, so rendering them unable to move; again the threat to humans is small unless the site of the test is dug up.
- The safety trials where bombs were subject to simulated accidents pose the greatest threat to people; some areas of land used for such experiments (conducted in the open air) have not been fully released for general use despite in one case an extensive decontamination.
Natural
Activation products from cosmic rays
Cosmogenic isotopes (or
Production modes
Here is a list of radioisotopes formed by the action of cosmic rays on the atmosphere; the list also contains the production mode of the isotope. These data were obtained from the SCOPE50 report, see table 1.9 of chapter 1.
| Isotope | Mode of formation |
|---|---|
| ³H (tritium) | 14N (n, 12C)³H |
| 7Be | Spallation (N and O) |
| 10Be | Spallation (N and O) |
| 11C | Spallation (N and O) |
| 14C | 14N (n, p) 14C |
| 18F | 18O (p, n)18F and Spallation (Ar) |
| 22Na | Spallation (Ar) |
| 24Na | Spallation (Ar) |
| 28Mg | Spallation (Ar) |
| 31Si | Spallation (Ar) |
| 32Si | Spallation (Ar) |
| 32P | Spallation (Ar) |
| 34mCl | Spallation (Ar) |
| 35S | Spallation (Ar) |
| 36Cl | 35Cl (n, )36Cl |
| 37Ar | 37Cl (p, n)37Ar |
| 38Cl | Spallation (Ar) |
| 39Ar | 38Ar (n, )39Ar |
| 39Cl | 40Ar (n, np)39Cl & spallation (Ar) |
| 41Ar | 40Ar (n, )41Ar |
| 81Kr | 80Kr (n, ) 81Kr |
Transfer to ground
The level of
Applications in geology listed by isotope
| element | mass | half-life (years) | typical application |
|---|---|---|---|
| helium | 3 | - stable - | exposure dating of olivine-bearing rocks |
| beryllium | 10 | 1.36 million | exposure dating of quartz-bearing rocks, sediment, dating of ice cores, measurement of erosion rates |
| carbon | 14 | 5,730 | dating of organic matter, water |
| neon | 21 | - stable - | dating of very stable, long-exposed surfaces, including meteorites |
aluminum |
26 | 720,000 | exposure dating of rocks, sediment |
| chlorine | 36 | 308,000 | exposure dating of rocks, groundwater tracer |
| calcium | 41 | 103,000 | exposure dating of carbonate rocks |
| iodine | 129 | 15.7 million | groundwater tracer |
Applications of dating
Because cosmogenic isotopes have long half-lives (anywhere from thousands to millions of years), scientists find them useful for geologic dating. Cosmogenic isotopes are produced at or near the surface of the Earth, and thus are commonly applied to problems of measuring ages and rates of geomorphic and sedimentary events and processes.
Specific applications of cosmogenic isotopes include:
- exposure dating of earth surfaces, including debris
- burial dating of sediment, bedrock, ice
- measurement of steady-state erosion rates
- absolute dating of organic matter (radiocarbon dating)
- absolute dating of water masses, measurement of groundwater transport rates
- absolute dating of meteorites, lunar surfaces
Methods of measurement for the long-lived isotopes
To measure cosmogenic isotopes produced within solid earth materials, such as rock, samples are generally first put through a process of mechanical separation. The sample is crushed and desirable material, such as a particular mineral (quartz in the case of Be-10), is separated from non-desirable material by using a density separation in a heavy liquid medium such as lithium sodium tungstate (LST). The sample is then dissolved, a common isotope carrier added (Be-9 carrier in the case of Be-10), and the aqueous solution is purified down to an oxide or other pure solid.
Finally, the ratio of the rare cosmogenic isotope to the common isotope is measured using accelerator mass spectrometry. The original concentration of cosmogenic isotope in the sample is then calculated using the measured isotopic ratio, the mass of the sample, and the mass of carrier added to the sample.
Radium and radon from the decay of long-lived actinides

Radium and radon are in the environment because they are decay products of uranium and thorium.
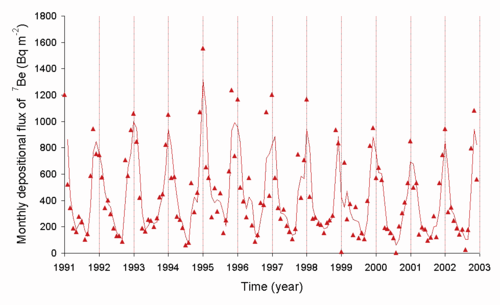
The radon (222Rn) released into the air decays to 210Pb and other radioisotopes, and the levels of 210Pb can be measured. The rate of deposition of this radioisotope is dependent on the weather. Below is a graph of the deposition rate observed in Japan.[8]
Uranium-lead dating
Uranium-lead dating is usually performed on the mineral zircon (ZrSiO4), though other materials can be used. Zircon incorporates uranium atoms into its crystalline structure as substitutes for zirconium, but strongly rejects lead. It has a high blocking temperature, is resistant to mechanical weathering and is chemically inert. Zircon also forms multiple crystal layers during metamorphic events, which each may record an isotopic age of the event. These can be dated by a SHRIMP ion microprobe.
One of the advantages of this method is that any sample provides two clocks, one based on uranium-235's decay to lead-207 with a half-life of about 703 million years, and one based on uranium-238's decay to lead-206 with a half-life of about 4.5 billion years, providing a built-in crosscheck that allows accurate determination of the age of the sample even if some of the lead has been lost.
See also
- Journal of Environmental Radioactivity
- Naturally occurring radioactive material
- Radioecology
- Radium in the environment
- Uranium in the environment
References
- ^ Generic Procedures for Assessment and Response during a Radiological Emergency, IAEA TECDOC Series number 1162, published in 2000 [1]
- ^ Z. Vukovic, V. Sipka, D. Todorovic and S. Stankovic, Journal of Radioanalytical and Nuclear Chemistry, 2006, 268, 129–131.
- ^ United Nations Scientific Committee on the Effects of Atomic Radiation, 1993, Report to the General Assembly, with scientific annexes, New York
- ISBN 80-7302-053-X.
- ^ P.P. Parekh, T.M. Semkow, M.A. Torres, D.K. Haines, J.M. Cooper, P.M. Rosenberg and M.E. Kitto, Journal of Environmental Radioactivity, 2006, 85, 103-120
- ^ A. Preston, J.W.R. Dutton and B.R. Harvey, Nature, 1968, 218, 689-690.
- ^ P. Soudek, Š. Valenová, Z. Vavříková and T. Vaněk, Journal of Environmental Radioactivity, 2006, 88, 236-250
- ^ M. Yamamoto et al., Journal of Environmental Radioactivity, 2006, 86, 110-131)
References about cosmogenic isotope dating
- Gosse, John C., and Phillips, Fred M. (2001). "Terrestrial in situ cosmogenic nuclides: Theory and application". Quaternary Science Reviews 20, 1475–1560.
- Granger, Darryl E., Fabel, Derek, and Palmer, Arthur N. (2001). "Pliocene-Pleistocene incision of the Green River, Kentucky, determined from radioactive decay of cosmogenic 26Al and 10Be in Mammoth Cave sediments". Geological Society of America Bulletin 113 (7), 825–836.
Further reading
- Radioactivity, Ionizing Radiation and Nuclear Energy, by J. Hala and J.D. Navratil
- A review of the subject has been published by Scientific Committee on Problems of the Environment (SCOPE) in the report SCOPE 50 Radioecology after chernobyl.
