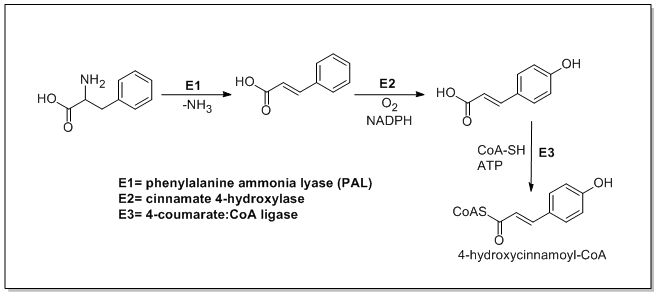
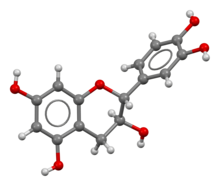
Catechin

| |
| |
| Names | |
|---|---|
| IUPAC name
(2R,3S)-2-(3,4-Dihydroxyphenyl)-3,4-dihydro-2H-chromene-3,5,7-triol
| |
| Other names
Cianidanol
Cyanidanol (+)-catechin D-Catechin Catechinic acid Catechuic acid Cianidol Dexcyanidanol (2R,3S)-Catechin 2,3-trans-Catechin (2R,3S)-Flavan-3,3′,4′,5,7-pentol | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.005.297 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H14O6 | |
| Molar mass | 290.271 g·mol−1 |
| Appearance | Colorless solid |
| Melting point | 175 to 177 °C (347 to 351 °F; 448 to 450 K) |
| UV-vis (λmax) | 276 nm |
Chiral rotation ([α]D)
|
+14.0° |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Mutagenic for mammalian somatic cells, mutagenic for bacteria and yeast |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
(+)-catechin : 10,000 mg/kg in rat (RTECS) 10,000 mg/kg in mouse 3,890 mg/kg in rat (other source) |
| Safety data sheet (SDS) | sciencelab AppliChem[permanent dead link] |
| Pharmacology | |
| Oral | |
| Pharmacokinetics: | |
| Urines | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Catechin
The name of the catechin chemical family derives from
Chemistry

Catechin possesses two
The most common catechin isomer is (+)-catechin. The other
The different epimers can be separated using chiral column chromatography.[2]
Making reference to no particular isomer, the molecule can just be called catechin. Mixtures of the different enantiomers can be called (±)-catechin or DL-catechin and (±)-epicatechin or DL-epicatechin.
Catechin and epicatechin are the building blocks of the proanthocyanidins, a type of condensed tannin.
- Diastereoisomers gallery
-
(+)-catechin (2R,3S)
-
(−)-catechin (2S,3R)
-
(−)-epicatechin (2R,3R)
-
(+)-epicatechin (2S,3S)

Moreover, the flexibility of the C-ring allows for two conformation isomers, putting the B-ring either in a pseudoequatorial position (E conformer) or in a pseudoaxial position (A conformer). Studies confirmed that (+)-catechin adopts a mixture of A- and E-conformers in aqueous solution and their conformational equilibrium has been evaluated to be 33:67.[3]
As flavonoids, catechins can act as
Oxidation
Electrochemical experiments show that (+)-catechin oxidation mechanism proceeds in sequential steps, related with the catechol and resorcinol groups and the oxidation is pH-dependent. The oxidation of the catechol 3′,4′-dihydroxyl electron-donating groups occurs first, at very low positive potentials, and is a reversible reaction. The hydroxyl groups of the resorcinol moiety oxidised afterwards were shown to undergo an irreversible oxidation reaction.[6]
The
Spectral data

UV-Vis
| |
|---|---|
Lambda-max :
|
276 nm |
Extinction coefficient (log ε)
|
4.01 |
| IR | |
| Major absorption bands | 1600 cm−1(benzene rings) |
NMR
| |
Proton NMR
|
δ : 2.49 (1H, dd, J = 16.0, 8.6 Hz, H-4a), |
Carbon-13 NMR
| |
| Other NMR data | |
MS
| |
| Masses of main fragments |
ESI-MS [M+H]+ m/z : 291.0
|
Natural occurrences
(+)-Catechin and (−)-epicatechin as well as their
In food
The main dietary sources of catechins in Europe and the United States are tea and pome fruits.[12][13]
Catechins and epicatechins are found in
Catechins are diverse among foods,[15] from peaches[17] to green tea and vinegar.[15][18] Catechins are found in barley grain where they are the main phenolic compound responsible for dough discoloration.[19] The taste associated with monomeric (+)-catechin or (−)-epicatechin is described as slightly astringent, but not bitter.[20]
Metabolism
Biosynthesis
The biosynthesis of catechin begins with ma
Biodegradation
Catechin oxygenase, a key enzyme in the degradation of catechin, is present in fungi and bacteria.[26]
Among bacteria, degradation of (+)-catechin can be achieved by
Among fungi, degradation of catechin can be achieved by Chaetomium cupreum.[29]
Metabolism in humans


Catechins are metabolised upon uptake from the
The stereochemical configuration of catechins has a strong impact on their uptake and metabolism as uptake is highest for (−)-epicatechin and lowest for (−)-catechin.[34]
Biotransformation
Biotransformation of (+)-catechin into taxifolin by a two-step oxidation can be achieved by Burkholderia sp.[35]
(+)-Catechin and (−)-epicatechin are transformed by the endophytic filamentous fungus
Leucoanthocyanidin reductase (LAR) uses (2R,3S)-catechin, NADP+ and H2O to produce 2,3-trans-3,4-cis-leucocyanidin, NADPH, and H+. Its gene expression has been studied in developing grape berries and grapevine leaves.[37]
Glycosides
- (2R,3S)-Catechin-7-O-β-D-glucopyranoside can be isolated from barley (Hordeum vulgare L.) and malt.[38]
- Epigeoside (catechin-3-O-α-L-rhamnopyranosyl-(1–4)-β-D-glucopyranosyl-(1–6)-β-D-glucopyranoside) can be isolated from the rhizomes of Epigynum auritum.[39]
Research

Vascular function
Only limited evidence from dietary studies indicates that catechins may affect
Due to extensive metabolism during digestion, the fate and activity of catechin metabolites responsible for this effect on blood vessels, as well as the actual mode of action, are unknown.[33][44]
Adverse events
Catechin and its metabolites can bind tightly to red blood cells and thereby induce the development of
Catechins from green tea can be hepatotoxic[48] and the European Food Safety Authority has recommended not to exceed 800 mg per day.[49]
Other
One limited meta-analysis showed that increasing consumption of green tea and its catechins to seven cups per day provided a small reduction in prostate cancer.[50] Nanoparticle methods are under preliminary research as potential delivery systems of catechins.[51]
Botanical effects
Catechins released into the ground by some plants may hinder the growth of their neighbors, a form of
Catechin acts as an infection-inhibiting factor in strawberry leaves.[54] Epicatechin and catechin may prevent coffee berry disease by inhibiting appressorial melanization of Colletotrichum kahawae.[55]
References
- ^ "Cutch and catechu plant origin". Food and Agriculture Organization of the United Nations. 5 November 2011. Archived from the original on 10 February 2019. Retrieved 26 July 2016.
- PMID 20143413.
- PMID 12956082.
- S2CID 23310671.
- PMID 8229463.
- hdl:10316/5128.
- .
- S2CID 28636157.
- PMID 25471637.
- .
- ^ "Michiyo Tsujimura (1888–1969)". Archived from the original on 21 November 2015. Retrieved 10 November 2015.
- PMID 17449588.
- PMID 26010916.
- PMID 18296356.
- ^ a b c "Polyphenols in green tea infusion". Phenol-Explorer, v 3.5. 2014. Retrieved 1 November 2014.
- PMID 18522407.
- .
- S2CID 91784893.
- PMID 17177530.
- .
- PMID 22226811.
- PMID 15464723.
- ISBN 978-0-470-74167-2.[page needed]
- JSTOR 2446026.
- PMID 20138891.
- ^ Arunachalam, M.; Mohan Raj, M.; Mohan, N.; Mahadevan, A. (2003). "Biodegradation of Catechin" (PDF). Proceedings of the Indian National Science Academy. B69 (4): 353–370. Archived from the original (PDF) on 2012-03-16.
- PMID 12787923.
- S2CID 41221044.
- S2CID 1257624.
- ^ PMID 27363516.
- PMID 23864538.
- PMID 22240152.
- ^ a b c "Flavonoids". Linus Pauling Institute, Oregon State University, Corvallis. 2016. Retrieved 24 July 2016.
- PMID 21074608.
- PMID 18068670.
- PMID 15997157.
- PMID 16169968.
- S2CID 84221785.
- PMID 1823978.
- PMID 22301923.
- PMID 22552030.
- S2CID 206969226.
- PMID 28933419.
- PMID 16418281.
- ISBN 978-0-470-57871-1.
- S2CID 3167832.
- ^ "Ruhen der Zulassung für Catergen" (PDF). Deutsches Ärzteblatt. 82 (38): 2706.
- ^ Health Canada (2017-11-15). "Summary Safety Review - Green tea extract-containing natural health products - Assessing the potential risk of liver injury (hepatotoxicity)". www.canada.ca. Retrieved 2022-05-06.
- PMID 32625874.
- PMID 28353571.
- S2CID 29522787.
- ^ a b Broz AK, Vivanco JM, Schultz MJ, Perry LG, Paschke MW (2006). "Secondary Metabolites and Allelopathy in Plant Invasions: A Case Study of Centaurea maculosa". In Taiz L, Zeiger E, Møller IM, Murphy A (eds.). Plant Physiology and Development (6th ed.). Sinauer Associates.
- S2CID 26483595.
- PMID 18944538.
- S2CID 30593181.
External links
 Media related to (+)-Catechin at Wikimedia Commons
Media related to (+)-Catechin at Wikimedia Commons