Epinephrine (medication)
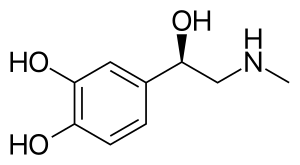 Skeletal formula of adrenaline | |
 | |
| Clinical data | |
|---|---|
| Trade names | EpiPen, Adrenaclick, others |
| Other names | Epinephrine, adrenaline, adrenalin |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603002 |
| License data |
|
| Pregnancy category |
|
COMT) | |
| Legal status | |
| Legal status | |
COMT) | |
| Metabolites | Metanephrine[4] |
| Onset of action | Rapid[5] |
| Elimination half-life | 2 minutes |
| Duration of action | Few minutes[6] |
| Excretion | Urine |
| Identifiers | |
| |
JSmol) | |
| Density | 1.283±0.06 g/cm3 @ 20 °C, 760 Torr |
| |
| |
Epinephrine, also known as adrenaline, is a
Common side effects include shakiness,
Epinephrine is normally produced by both the
Medical uses
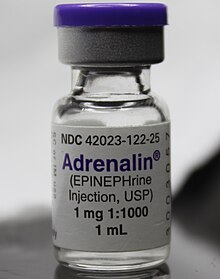
Epinephrine is used to treat a number of conditions, including cardiac arrest, anaphylaxis, and superficial bleeding.[21] It has been used historically for bronchospasm and low blood sugar, but newer treatments for these that are selective for β2 adrenoceptors, such as salbutamol, are currently preferred.[citation needed]
Heart problems
While epinephrine is often used to treat
In cases of IV infusion for bradycardia the dose is 1 mg epinephrine is mixed with 500ml of NS or D5W. The infusion should run at 2-10 micrograms/min (titrated to effect). IV infusion for post-cardiac arrest hypotension: The dosing is 2 to 10 micrograms/min.
Endotracheal Tube: 2-2.5 mg epinephrine is diluted in 10cc NS and given directly into the ET tube.[26]
Anaphylaxis
Epinephrine is the drug of choice for treating allergic reaction anaphylaxis. The commonly used epinephrine autoinjector delivers a 0.3 mg epinephrine injection (0.3 mL, 1:1000). It is indicated in the emergency treatment of allergic reactions, including anaphylaxis to stings, contrast agents, medicines, or people with a history of anaphylactic reactions to known triggers. A single dose is recommended for people who weigh 30 kg or more, repeated if necessary. A lower-strength product is available for children.[27][28][29][30]
Intramuscular injection can be complicated in that the depth of subcutaneous fat varies and may result in
Asthma
Epinephrine is also used as a bronchodilator for asthma if specific β2 agonists are unavailable or ineffective.[33]
When given by the subcutaneous or intramuscular routes for asthma, an appropriate dose is 0.3 to 0.5 mg.[34][35]
Because of the high intrinsic efficacy (receptor binding ability) of epinephrine, high drug concentrations cause adverse side effects when treating asthma. The value of using nebulized epinephrine in acute asthma is unclear.[36]
Croup
Racemic epinephrine has historically been used for the
Bronchiolitis
There is a lack of consensus as to whether inhaled nebulized epinephrine is beneficial in the treatment of bronchiolitis, with most guidelines recommending against its use.[40]
Local anesthetics
When epinephrine is mixed with local anesthetics, such as bupivacaine or lidocaine, and used for local anesthesia or intrathecal injection, it prolongs the numbing effect and motor block effect of the anesthetic by up to an hour.[41] Epinephrine is frequently combined with local anesthetic and can cause panic attacks.[42]
Epinephrine is mixed with cocaine to form Moffett's solution, used in nasal surgery.[43]
Upper airway obstruction
Upper airway obstruction with edema and stridor can be treated with racemic epinephrine.[44]
Adverse effects
Adverse reactions to adrenaline include
Rarely, exposure to medically administered epinephrine may cause Takotsubo cardiomyopathy.[45]
Use is contraindicated in people on nonselective
Mechanism of action
| Organ | Effects |
|---|---|
| Heart | Increases heart rate; contractility; conduction across AV node |
| Lungs | Increases respiratory rate; bronchodilation |
| Liver | Stimulates glycogenolysis |
| Brain | |
| Systemic | Vasoconstriction and vasodilation |
| Triggers lipolysis | |
| Muscle contraction |
As a hormone, epinephrine acts on nearly all body tissues. Its actions vary by tissue type and tissue expression of adrenergic receptors. For example, high epinephrine levels cause smooth muscle relaxation in the airways but cause contraction of the smooth muscle that lines most arterioles.[citation needed]
Epinephrine acts by binding to a variety of
Its actions increase peripheral resistance via α1 receptor-dependent vasoconstriction and increase cardiac output via its binding to β1 receptors. The goal of reducing peripheral circulation is to increase coronary and cerebral perfusion pressures and therefore increase oxygen exchange at the cellular level.[51] While epinephrine does increase aortic, cerebral, and carotid circulation pressure, it lowers carotid blood flow and end-tidal CO2 or ETCO2 levels. It appears that epinephrine may improve macrocirculation at the expense of the capillary beds where perfusion takes place.[52]
History
Extracts of the adrenal gland were first obtained by Polish physiologist Napoleon Cybulski in 1895. These extracts, which he called nadnerczyna, contained adrenaline and other catecholamines.[53] American ophthalmologist William H. Bates discovered adrenaline's usage for eye surgeries prior to 20 April 1896.[54] Japanese chemist Jōkichi Takamine and his assistant Keizo Uenaka independently discovered adrenaline in 1900.[55][56] In 1901, Takamine successfully isolated and purified the hormone from the adrenal glands of sheep and oxen.[57] Adrenaline was first synthesized in the laboratory by Friedrich Stolz and Henry Drysdale Dakin, independently, in 1904.[56]
Society and culture
Brand names
Common brand names include Asthmanefrin, Micronefrin, Nephron, VapoNefrin, and Primatene Mist.
Delivery forms
Epinephrine is available in an autoinjector delivery system.
There is an epinephrine
A common concentration for epinephrine is 2.25% w/v epinephrine in solution, which contains 22.5 mg/mL, while a 1% solution is typically used for aerosolization.
- Adults: 0.5–0.75 ml of a 2.25% solution in 2.0 ml normal saline.[61]
- Pediatrics: 0.25–0.75 ml of a 2.25% solution in 2.0 ml normal saline.[61][62]
References
- PMID 1136652.
- S2CID 9054777.
- PMID 5468075.
- S2CID 2330329.
- ^ a b c d e f g h i "Epinephrine". The American Society of Health-System Pharmacists. Archived from the original on 6 September 2015. Retrieved 15 August 2015.
- ISBN 9781449645861. Archivedfrom the original on 8 September 2017.
- ^ ISBN 9781608315727. Archivedfrom the original on 8 September 2017.
- ^ "(-)-adrenaline". Guide to Pharmacology. IUPS/BPS. Archived from the original on 1 September 2015. Retrieved 21 August 2015.
- PMID 19135584.
- ISBN 9780071481274.
Epinephrine occurs in only a small number of central neurons, all located in the medulla. Epinephrine is involved in visceral functions, such as the control of respiration. It is also produced by the adrenal medulla.
- ISBN 9780781768528. Archivedfrom the original on 8 September 2017.
- ^ ISBN 9788131215661. Archivedfrom the original on 8 September 2017.
- ISBN 9781483262888. Archivedfrom the original on 8 September 2017.
- ISBN 9781107268500. Archivedfrom the original on 8 September 2017.
- ^ "Epinephrine".
- ISBN 9780080568775. Archivedfrom the original on 8 September 2017.
- ISBN 9783527607495.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Epinephrine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "Epinephrine". The American Society of Health-System Pharmacists. Archived from the original on 7 March 2011. Retrieved 3 April 2011.
- S2CID 58580872. Archived from the originalon 28 August 2021. Retrieved 27 January 2019.
- PMID 25215099.
- ^ PMID 24642404.
- PMID 26472995.
- ^ "ACLS and epinephrine".
- ^ Mylan Specialty L.P. "EPIPEN®- epinephrine injection, EPIPEN Jr®- epinephrine injection" (PDF). FDA Product Label. Archived (PDF) from the original on 1 February 2014. Retrieved 22 January 2014.
- .
- PMID 20956224.
- PMID 20692689.
- ^ Pennsylvania Patient Advisory. "Let's Stop this "Epi"demic!—Preventing Errors with Epinephrine". Archived from the original on 2 February 2014. Retrieved 22 January 2014.
- PMID 14656845.
- PMID 23578933.
- ^ Soar, Perkins, et al (2010) European Resuscitation Council Guidelines for Resuscitation 2010 Section 8. Cardiac arrest in special circumstances: Electrolyte abnormalities, poisoning, drowning, accidental hypothermia, hyperthermia, asthma, anaphylaxis, cardiac surgery, trauma, pregnancy, electrocution. Resuscitation. Oct. pp.1400–1433
- ^ Fisher, Brown, Cooke (Eds) (2006) Joint Royal Colleges Ambulance Liaison Committee. UK Ambulance Clinical Practice Guidelines.
- S2CID 37328426.
- PMID 18295000.
- ^ PMID 9451322.
- ^ S2CID 41978318.
- PMID 31541233.
- S2CID 29154283.
- ISBN 978-3-00-008562-8.
- PMID 15533141.
- ^ Markovchick V (2007). Critical Care Secrets (fourth ed.).
- PMID 27889211.
- ^ ISBN 978-1-59541-101-3.
- PMID 3521311.
- PMID 1900669.
- ^ ISBN 978-3-13-144061-7.
- PMID 12147535.
- ^ "Guideline 11.5: Medications in Adult Cardiac Arrest" (PDF). Australian Resuscitation Council. December 2010. Archived from the original on 12 March 2015. Retrieved 7 March 2015.
- PMID 22445865.
- from the original on 10 March 2011.
- ^ Bates WH (16 May 1896). "The Use of Extract of Suprarenal Capsule in the Eye". New York Medical Journal: 647–650. Archived from the original on 3 April 2015. Retrieved 7 March 2015.
- S2CID 32540165.
- ^ S2CID 20999106.
- ^ Takamine J (1901). The isolation of the active principle of the suprarenal gland. Great Britain: Cambridge University Press. pp. xxix–xxx.
{{cite book}}:|work=ignored (help) - ^ "Background". Armstrong Pharmaceuticals. Archived from the original on 13 September 2013. Retrieved 12 December 2016.
- ^ "Statement from FDA Commissioner Scott Gottlieb, M.D., and Janet Woodcock, M.D., director of the Center for Drug Evaluation and Research, on approval of OTC Primatene Mist to treat mild asthma". FDA archive. 24 March 2020.
- ^ "Frequent Asked Questions". Armstrong Pharmaceuticals. Archived from the original on 25 September 2011. Retrieved 22 September 2011.
Primatene® Mist was launched in 1963. The Primatene® Mist brand has built a long-time heritage for over-the-counter relief of bronchial asthma.
- ^ PMID 17626698.
- PMID 11869578.
