Essential gene
Essential genes are indispensable genes for organisms to grow and reproduce offspring under certain environment.[1] However, being essential is highly dependent on the circumstances in which an organism lives. For instance, a gene required to digest starch is only essential if starch is the only source of energy. Recently, systematic attempts have been made to identify those genes that are absolutely required to maintain life, provided that all nutrients are available.[2] Such experiments have led to the conclusion that the absolutely required number of genes for bacteria is on the order of about 250–300. Essential genes of single-celled organisms encode proteins for three basic functions including genetic information processing, cell envelopes and energy production.[1] Those gene functions are used to maintain a central metabolism, replicate DNA, translate genes into proteins, maintain a basic cellular structure, and mediate transport processes into and out of the cell. Compared with single-celled organisms, multicellular organisms have more essential genes related to communication and development. Most of the essential genes in viruses are related to the processing and maintenance of genetic information. In contrast to most single-celled organisms, viruses lack many essential genes for metabolism,[1] which forces them to hijack the host's metabolism. Most genes are not essential but convey selective advantages and increased fitness. Hence, the vast majority of genes are not essential and many can be deleted without consequences, at least under most circumstances.
Bacteria: genome-wide studies
Two main strategies have been employed to identify essential genes on a genome-wide basis: directed deletion of genes and random
| Organism | Mutagenesis | Method | Readout | ORFs | Non-ess. | Essential | % Ess. | Notes | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Mycoplasma genitalium/pneumoniae | Random | Population | Sequencing | 482 | 130 | 265–350 | 55–73% | --- | [5] |
| Mycoplasma genitalium | Random | Clones | Sequencing | 482 | 100 | 382 | 79% | b,c | [6] |
| Staphylococcus aureus WCUH29 | Random | Clones | Sequencing | 2,600 | n/a | 168 | n/a | b,c | [7] |
| Staphylococcus aureus RN4220 | Random | Clones | Sequencing | 2,892 | n/a | 658 | 23% | --- | [8] |
| Haemophilus influenzae Rd | Random | Population | Footprint-PCR | 1,657 | 602 | 670 | 40% | --- | [9] |
| Streptococcus pneumoniae Rx-1 | Targeted | Clones | Colony formation | 2,043 | 234 | 113 | n/a | c | [10] |
| Streptococcus pneumoniae D39 | Targeted | Clones | Colony formation | 2,043 | 560 | 133 | n/a | c | [11] |
| Streptococcus pyogenes 5448 | Random | Transposon | Tn-seq | 1,865 | ? | 227 | 12% | --- | [12] |
| Streptococcus pyogenes NZ131 | Random | Transposon | Tn-seq | 1,700 | ? | 241 | 14% | --- | [12] |
| Streptococcus sanguinis SK36 | Targeted | Clones | Colony formation | 2,270 | 2,052 | 218 | 10% | a,j | [1][13] |
| Mycobacterium tuberculosis H37Rv | Random | Population | Microarray | 3,989 | 2,567 | 614 | 15% | --- | [14] |
| Mycobacterium tuberculosis | Random | Transposon | ? | 3,989 | ? | 401 | 10% | --- | [15] |
| Mycobacterium tuberculosis H37Rv | Random | Transposon | NG-Sequencing | 3,989 | ? | 774 | 19% | --- | [16][17] |
| Mycobacterium tuberculosis H37Rv | Random | Transposon | NG-Sequencing | 3,989 | 3,364 | 625 | 16% | h,i | [18] |
| Mycobacterium tuberculosis | --- | Computational | Computational | 3,989 | ? | 283 | 7% | --- | [19] |
| Mycobacterium tuberculosis H37Rv | Targeted | CRISPRi | NG-Sequencing | 4,052 | 33,15 | 737 | 18% | --- | [20] |
| Bacillus subtilis 168 | Targeted | Clones | Colony formation | 4,105 | 3,830 | 261 | 7% | a,d,g | [21][22] |
| Escherichia coli K-12 MG1655 | Random | Population | Footprint-PCR | 4,308 | 3,126 | 620 | 14% | --- | [23] |
| Escherichia coli K-12 MG1655 | Targeted | Clones | Colony formation | 4,308 | 2,001 | n/a | n/a | a,e | [24] |
| Escherichia coli K-12 BW25113 | Targeted | Clones | Colony formation | 4,390 | 3,985 | 303 | 7% | a | [25] |
| Pseudomonas aeruginosa PAO1 | Random | Clones | Sequencing | 5,570 | 4,783 | 678 | 12% | a | [26] |
| Porphyromonas gingivalis | Random | Transposon | Sequencing | 1,990 | 1,527 | 463 | 23% | --- | [27] |
| Pseudomonas aeruginosa PA14 | Random | Clones | Sequencing | 5,688 | 4,469 | 335 | 6% | a,f | [28] |
Salmonella typhimurium |
Random | Clones | Sequencing | 4,425 | n/a | 257 | ~11% | b,c | [29] |
| Helicobacter pylori G27 | Random | Population | Microarray | 1,576 | 1,178 | 344 | 22% | --- | [30] |
| Corynebacterium glutamicum | Random | Population | ? | 3,002 | 2,352 | 650 | 22% | --- | [31] |
| Francisella novicida | Random | Transposon | ? | 1,719 | 1,327 | 392 | 23% | --- | [32] |
| Mycoplasma pulmonis UAB CTIP | Random | Transposon | ? | 782 | 472 | 310 | 40% | --- | [35] |
| Vibrio cholerae N16961 | Random | Transposon | ? | 3,890 | ? | 779 | 20% | --- | [36] |
Salmonella Typhi |
Random | Transposon | ? | 4,646 | ? | 353 | 8% | --- | [37] |
| Staphylococcus aureus | Random | Transposon | ? | ~2,600 | ? | 351 | 14% | --- | [38] |
| Caulobacter crescentus | Random | Transposon | Tn-Seq | 3,876 | 3,240 | 480 | 12.2% | --- | [39] |
| Neisseria meningitidis | Random | Transposon | ? | 2,158 | ? | 585 | 27% | --- | [40] |
Desulfovibrio alaskensis |
Random | Transposon | Sequencing | 3,258 | 2,871 | 387 | 12% | --- | [41] |
Table 1. Essential genes in bacteria. Mutagenesis: targeted mutants are gene deletions; random mutants are
On the basis of genome-wide experimental studies and systems biology analysis, an essential gene database has been developed by Kong et al. (2019) for predicting > 4000 bacterial species.[42]
Eukaryotes
In Saccharomyces cerevisiae (budding yeast) 15-20% of all genes are essential. In Schizosaccharomyces pombe (fission yeast) 4,836 heterozygous deletions covering 98.4% of the 4,914 protein coding open reading frames have been constructed. 1,260 of these deletions turned out to be essential.[43]
Similar screens are more difficult to carry out in other multicellular organisms, including mammals (as a model for humans), due to technical reasons, and their results are less clear. However, various methods have been developed for the nematode worm C. elegans,[44] the fruit fly,[45] and zebrafish[46] (see table). A recent study of 900 mouse genes concluded that 42% of them were essential although the selected genes were not representative.[47]
Gene knockout experiments are not possible or at least not ethical in humans. However, natural mutations have led to the identification of mutations that lead to early embryonic or later death.
While it may be difficult to prove that a gene is essential in humans, it can be demonstrated that a gene is not essential or not even causing disease. For instance, sequencing the genomes of 2,636 Icelandic citizens and the genotyping of 101,584 additional subjects found 8,041 individuals who had 1 gene completely knocked out (i.e. these people were homozygous for a non-functional gene).[50] Of the 8,041 individuals with complete knock-outs, 6,885 were estimated to be homozygotes, 1,249 were estimated to be compound heterozygotes (i.e. they had both alleles of a gene knocked out but the two alleles had different mutations). In these individuals, a total of 1,171 of the 19,135 human (RefSeq) genes (6.1%) were completely knocked out. It was concluded that these 1,171 genes are non-essential in humans — at least no associated diseases were reported.[50] Similarly, the exome sequences of 3222 British Pakistani-heritage adults with high parental relatedness revealed 1111 rare-variant homozygous genotypes with predicted loss of gene function (LOF = knockouts) in 781 genes.[51] This study found an average of 140 predicted LOF genotypes (per subject), including 16 rare (minor allele frequency <1%) heterozygotes, 0.34 rare homozygotes, 83.2 common heterozygotes and 40.6 common homozygotes. Nearly all rare homozygous LOF genotypes were found within autozygous segments (94.9%).[51] Even though most of these individuals had no obvious health issue arising from their defective genes, it is possible that minor health issues may be found upon more detailed examination.
A summary of essentiality screens is shown in the table below (mostly based on the Database of Essential Genes.[52]
| Organism | Method | Essential genes | Ref. |
| Arabidopsis thaliana | T-DNA insertion | 777 | [53] |
| Caenorhabditis elegans (worm) | RNA interference | 294 | [44] |
Danio rerio (zebrafish) |
Insertion mutagenesis | 288 | [46] |
| Drosophila melanogaster (fruit fly) | P-element insertion mutagenesis | 339 | [45] |
Homo sapiens (human) |
Literature search | 118 | [48] |
Homo sapiens (human) |
CRISPR/Cas9-based screen | 1,878 | [54] |
Homo sapiens (human) |
Haploid gene-trap screen | ~2,000 | [55] |
Homo sapiens (human)
|
mouse orthologs | 2,472 | [56] |
Mus musculus (mouse) |
Literature search | 2114 | [57] |
| Saccharomyces cerevisiae (yeast) | Single-gene deletions | 878 | [58] |
| Saccharomyces cerevisiae (yeast) | Single-gene deletions | 1,105 | [59] |
| Schizosaccharomyces pombe (yeast) | Single-gene deletions | 1,260 | [43] |
Viruses
Viruses lack many genes necessary for metabolism,
Tscharke and Dobson (2015) compiled a comprehensive survey of essential genes in Vaccinia Virus and assigned roles to each of the 223 ORFs of the Western Reserve (WR) strain and 207 ORFs of the Copenhagen strain, assessing their role in replication in cell culture. According to their definition, a gene is considered essential (i.e. has a role in cell culture) if its deletion results in a decrease in virus titre of greater than 10-fold in either a single or multiple step growth curve. All genes involved in wrapped virion production, actin tail formation, and extracellular virion release were also considered as essential. Genes that influence plaque size, but not replication were defined as non-essential. By this definition 93 genes are required for Vaccinia Virus replication in cell culture, while 108 and 94 ORFs, from WR and Copenhagen respectively, are non-essential.[61] Vaccinia viruses with deletions at either end of the genome behaved as expected, exhibiting only mild or host range defects. In contrast, combining deletions at both ends of the genome for VACV strain WR caused a devastating growth defect on all cell lines tested. This demonstrates that single gene deletions are not sufficient to assess the essentiality of genes and that more genes are essential in Vaccinia virus than originally thought.[61]
One of the bacteriophages screened for essential genes includes mycobacteriophage Giles. At least 35 of the 78 predicted Giles genes (45%) are non-essential for lytic growth. 20 genes were found to be essential.[62] A major problem with phage genes is that a majority of their genes remain functionally unknown, hence their role is difficult to assess. A screen of Salmonella enterica phage SPN3US revealed 13 essential genes although it remains a bit obscure how many genes were really tested.[63]
Quantitative gene essentiality analysis
In theory, essential genes are qualitative.[1] However, depending on the surrounding environment, certain essential gene mutants may show partial functions, which can be quantitatively determined in some studies. For instance, a particular gene deletion may reduce growth rate (or fertility rate or other characters) to 90% of the wild-type. If there are isozymes or alternative pathways for the essential genes, they can be deleted completely.[1] Using CRISPR interference, the expression of essential genes can be modulated or "tuned", leading to quantitative (or continuous) relationships between the level of gene-expression and the magnitude of fitness cost exhibited by a given mutant.[20]
Synthetic lethality
Two genes are synthetic lethal if neither one is essential but when both are mutated the double-mutant is lethal. Some studies have estimated that the number of synthetic lethal genes may be on the order of 45% of all genes.[64][65]
Conditionally essential genes

Many genes are essential only under certain circumstances. For instance, if the amino acid lysine is supplied to a cell any gene that is required to make lysine is non-essential. However, when there is no lysine supplied, genes encoding enzymes for lysine biosynthesis become essential, as no protein synthesis is possible without lysine.[4]
Streptococcus pneumoniae appears to require 147 genes for growth and survival in saliva,[66] more than the 113-133 that have been found in previous studies.
The deletion of a gene may result in death or in a block of cell division. While the latter case may implicate "survival" for some time, without cell division the cell may still die eventually. Similarly, instead of blocked cell division a cell may have reduced growth or metabolism ranging from nearly undetectable to almost normal. Thus, there is gradient from "essential" to completely non-essential, again depending on the condition. Some authors have thus distinguished between genes "essential for survival" and "essential for fitness".[4]
The role of genetic background. Similar to environmental conditions, the genetic background can determine the essentiality of a gene: a gene may be essential in one individual but not another, given his or her genetic background. Gene duplications are one possible explanation (see below).
Metabolic dependency. Genes involved in certain biosynthetic pathways, such as amino acid synthesis, can become non-essential if one or more amino acids are supplied by culture medium[1] or by another organism.[67] This is the main reason why many parasites (e.g. Cryptosporidium hominis)[68] or endosymbiontic bacteria lost many genes (e.g. Chlamydia). Such genes may be essential but only present in the host organism. For instance, Chlamydia trachomatis cannot synthesize purine and pyrimidine nucleotides de novo, so these bacteria are dependent on the nucleotide biosynthetic genes of the host.[69]
Another kind of metabolic dependency, unrelated to cross-species interactions, can be found when bacteria are grown under specific nutrient conditions. For example, more than 100 genes become essential when Escherichia coli is grown on nutrient-limited media. Specifically,
Gene duplications and alternative metabolic pathways
Many genes are duplicated within a genome and many organisms have different metabolic pathways (alternative metabolic pathway
In the worm C. elegans, non-essential genes are highly over-represented among duplicates, possibly because duplication of essential genes causes overexpression of these genes. Woods et al. found that non-essential genes are more often successfully duplicated (fixed) and lost compared to essential genes. By contrast, essential genes are less often duplicated but upon successful duplication are maintained over longer periods.[71]
Conservation
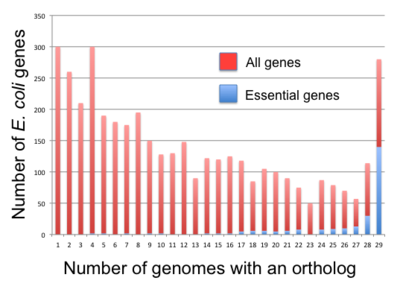
In bacteria, essential genes appear to be more conserved than nonessential genes
In eukaryotes, 83% of the one-to-one orthologs between Schizosaccharomyces pombe and Saccharomyces cerevisiae have conserved essentiality, that is, they are nonessential in both species or essential in both species. The remaining 17% of genes are nonessential in one species and essential in the other.[74] This is quite remarkable, given that S. pombe is separated from S. cerevisiae by approximately 400 million years of evolution.[75]
In general, highly conserved and thus older genes (i.e. genes with earlier phylogenetic origin) are more likely to be essential than younger genes - even if they have been duplicated.[76]
Study
The experimental study of essential genes is limited by the fact that, by definition, inactivation of an essential gene is lethal to the organism. Therefore, they cannot be simply deleted or mutated to analyze the resulting phenotypes (a common technique in genetics).
There are, however, some circumstances in which essential genes can be manipulated. In
Computational analysis can reveal many properties of proteins without analyzing them experimentally, e.g. by looking at homologous proteins, function, structure etc. (see also below, Predicting essential genes). The products of essential genes can also be studied when expressed in other organisms, or when purified and studied in vitro.
Conditionally essential genes are easier to study. Temperature-sensitive variants of essential genes have been identified which encode products that lose function at high temperatures, and so only show a phenotype at increased temperature.[77]
Reproducibility
If screens for essential genes are repeated in independent laboratories, they often result in different gene lists. For instance, screens in E. coli have yielded from ~300 to ~600 essential genes (see Table 1). Such differences are even more pronounced when different bacterial strains are used (see Figure 2). A common explanation is that the experimental conditions are different or that the nature of the mutation may be different (e.g. a complete gene deletion vs. a transposon mutant).[4] Transposon screens in particular are hard to reproduce, given that a transposon can insert at many positions within a gene. Insertions towards the 3' end of an essential gene may not have a lethal phenotype (or no phenotype at all) and thus may not be recognized as such. This can lead to erroneous annotations (here: false negatives).[78]
Comparison of
Different essential genes in different organisms
Different organisms may have different essential genes. For instance, Bacillus subtilis has 271 essential genes.[21] About one-half (150) of the orthologous genes in E. coli are also essential. Another 67 genes that are essential in E. coli are not essential in B. subtilis, while 86 E. coli essential genes have no B. subtilis ortholog.[25] In Mycoplasma genitalium at least 18 genes are essential that are not essential in M. bovis.[80] Many of these different essential genes are caused by paralogs or alternative metabolic pathways.[1]
Such different essential genes in bacteria can be used to develop targeted antibacterial therapies against certain specific pathogens to reduce
Prediction
Essential genes can be predicted computationally. However, most methods use experimental data ("training sets") to some extent. Chen et al.[83] determined four criteria to select training sets for such predictions: (1) essential genes in the selected training set should be reliable; (2) the growth conditions in which essential genes are defined should be consistent in training and prediction sets; (3) species used as training set should be closely related to the target organism; and (4) organisms used as training and prediction sets should exhibit similar phenotypes or lifestyles. They also found that the size of the training set should be at least 10% of the total genes to yield accurate predictions. Some approaches for predicting essential genes are:
Comparative genomics. Shortly after the first genomes (of Haemophilus influenzae and Mycoplasma genitalium) became available, Mushegian et al.[84] tried to predict the number of essential genes based on common genes in these two species. It was surmised that only essential genes should be conserved over the long evolutionary distance that separated the two bacteria. This study identified approximately 250 candidate essential genes.[84] As more genomes became available the number of predicted essential genes kept shrinking because more genomes shared fewer and fewer genes. As a consequence, it was concluded that the universal conserved core consists of less than 40 genes.[85][86] However, this set of conserved genes is not identical to the set of essential genes as different species rely on different essential genes.
A similar approach has been used to infer essential genes from the pan-genome of Brucella species. 42 complete Brucella genomes and a total of 132,143 protein-coding genes were used to predict 1252 potential essential genes, derived from the core genome by comparison with a prokaryote database of essential genes.[87]
Network analysis. After the first protein interaction networks of yeast had been published,[88] it was found that highly connected proteins (e.g. by protein-protein interactions) are more likely to be essential.[89] However, highly connected proteins may be experimental artifacts and high connectivity may rather represent pleiotropy instead of essentiality.[90] Nevertheless, network methods have been improved by adding other criteria and therefore do have some value in predicting essential genes.[91]
Machine Learning. Hua et al. used Machine Learning to predict essential genes in 25 bacterial species.[92]
Hurst index. Liu et al. (2015)[93] used the Hurst exponent, a characteristic parameter to describe long-range correlation in DNA to predict essential genes. In 31 out of 33 bacterial genomes the significance levels of the Hurst exponents of the essential genes were significantly higher than for the corresponding full-gene-set, whereas the significance levels of the Hurst exponents of the nonessential genes remained unchanged or increased only slightly.
Minimal genomes. It was also thought that essential genes could be inferred from
Metabolic modelling. Essential genes may be also predicted in completely sequenced genomes by metabolic reconstruction, that is, by reconstructing the complete metabolism from the gene content and then identifying those genes and pathways that have been found to be essential in other species. However, this method can be compromised by proteins of unknown function. In addition, many organisms have backup or alternative pathways which have to be taken into account (see figure 1). Metabolic modeling was also used by Basler (2015) to develop a method to predict essential metabolic genes.[95] Flux balance analysis, a method of metabolic modeling, has recently been used to predict essential genes in clear cell renal cell carcinoma metabolism.[96]
Genes of unknown function. Surprisingly, a significant number of essential genes has no known function. For instance, among the 385 essential candidates in M. genitalium, no function could be ascribed to 95 genes[6] even though this number had been reduced to 75 by 2011.[86] Most of unknown functionally essential genes have potential biological functions related to one of the three fundamental functions.[1]
ZUPLS. Song et al. presented a novel method to predict essential genes that only uses the Z-curve and other sequence-based features.[97] Such features can be calculated readily from the DNA/amino acid sequences. However, the reliability of this method remains a bit obscure.
Essential gene prediction servers. Guo et al. (2015) have developed three online services to predict essential genes in bacterial genomes. These freely available tools are applicable for single gene sequences without annotated functions, single genes with definite names, and complete genomes of bacterial strains.[98] Kong et al. (2019) have developed the ePath database, which can be used to search > 4000 bacterial species for predicting essential genes.[42]
Essential protein domains
Although most essential genes encode proteins, many essential proteins consist of a single domain. This fact has been used to identify essential protein domains. Goodacre et al. have identified hundreds of essential domains of unknown function (eDUFs).[99] Lu et al.[100] presented a similar approach and identified 3,450 domains that are essential in at least one microbial species.
See also
- Essential amino acid
- Essential proteins in protein complexes
- Gene
- Genome
- Minimal genome
- Mutation
References
- ^ PMID 22355642.
- ^ PMID 18974178.
- PMID 12657046.
- ^ PMID 16978855.
- S2CID 235447.
- ^ PMID 16407165.
- S2CID 24126939.
- PMID 11952893.
- PMID 11805338.
- PMID 12136097.
- PMID 15995353.
- ^ PMID 25996237.
- PMID 25636610.
- PMID 11606763.
- PMID 21285434.
- ^ PMID 21980284.
- PMID 25636614.
- PMID 28096490.
- PMID 24308365.
- ^ PMID 34297925.
- ^ PMID 12682299.
- ^ S2CID 23769853.
- PMID 13129938.
- PMID 15262929.
- ^ PMID 16738554.
- PMID 14617778.
- PMID 26358096.
- PMID 16477005.
- S2CID 45267476.
- PMID 15547264.
- PMID 21815106.
- PMID 17215359.
- S2CID 24054117.
- PMID 25636609.
- PMID 18452587.
- PMID 18574146.
- PMID 19826075.
- PMID 19570206.
- PMID 21878915.
- PMID 22208880.
- PMID 24865553.
- ^ PMID 31506471.
- ^ PMID 20473289.
- ^ S2CID 15745225.
- ^ PMID 10471706.
- ^ PMID 15256591.
- PMID 23870131.
- ^ PMID 18458337.
- PMID 23675308.
- ^ S2CID 205349719.
- ^ PMID 26940866.
- PMID 24243843.
- PMID 15266054.
- PMID 26472758.
- S2CID 26529733.
- PMID 23675308.
- PMID 17559966.
- PMID 11752246.
- ^ S2CID 4400400.
- PMID 14519856.
- ^ PMID 26290187.
- PMID 23560716.
- PMID 27605673.
- S2CID 4424895.
- PMID 25636612.
- PMID 24586856.
- PMID 27814362.
- S2CID 4394344.
- S2CID 46389854.
- PMID 27879333.
- PMID 23675306.
- ^ PMID 16014871.
- PMID 12045149.
- PMID 23661563.
- PMID 11178233.
- PMID 22319151.
- PMID 26175450.
- PMID 23520492.
- PMID 27159373.
- PMID 24897538.
- PMID 28609586.
- PMID 26544875.
- PMID 24466248.
- ^ PMID 8816789.
- PMID 15574825.
- ^ PMID 21889892.
- S2CID 14565579.
- S2CID 3009359.
- S2CID 258942.
- PMID 18719252.
- PMID 30776072.
- PMID 27660763.
- PMID 26067107.
- PMID 19609354.
- PMID 25636620.
- PMID 26040780.
- PMID 24603751.
- PMID 25636621.
- PMID 24381303.
- PMID 25636623.
Further reading
- Gao F, Luo H, Zhang CT, Zhang R (2015). "Gene Essentiality Analysis Based on DEG 10, an Updated Database of Essential Genes". Gene Essentiality. Methods in Molecular Biology. Vol. 1279. pp. 219–33. PMID 25636622.
- Long JL, ed. (2015). Gene Essentiality - Springer Methods and Protocols. Methods in Molecular Biology. Vol. 1279. Humana Press. p. 248. S2CID 27547825.
- Zhang R, ed. (2022). Essential Genes and Genomes. Methods in Molecular Biology. Vol. 2377. Humana Press. p. 434. S2CID 240006552.
