Estrogen
| Estrogen | |
|---|---|
| External links | |
| MeSH | D004967 |
| Legal status | |
| In Wikidata | |
Estrogen (
Estrogens are synthesized in all vertebrates[4] and some insects.[5] Quantitatively, estrogens circulate at lower levels than androgens in both men and women.[6] While estrogen levels are significantly lower in males than in females, estrogens nevertheless have important physiological roles in males.[7]
Like all steroid hormones, estrogens readily diffuse across the cell membrane. Once inside the cell, they bind to and activate estrogen receptors (ERs) which in turn modulate the expression of many genes.[8] Additionally, estrogens bind to and activate rapid-signaling membrane estrogen receptors (mERs),[9][10] such as GPER (GPR30).[11]
In addition to their role as natural hormones, estrogens are used as
Synthetic and natural estrogens have been found in the environment and are referred to as xenoestrogens. Estrogens are among the wide range of endocrine-disrupting compounds (EDCs) and can cause health issues and reproductive disfunction in both wildlife and humans.[12][13]
Types and examples
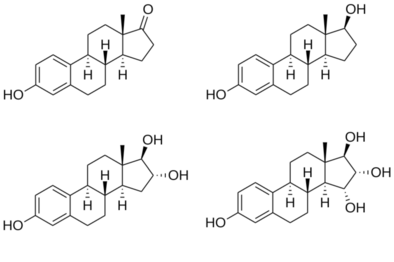
Structures of major endogenous estrogens
hydroxyl (–OH) groups : estrone (E1) has one, estradiol (E2) has two, estriol (E3) has three, and estetrol (E4) has four. |
The four major naturally occurring estrogens are
Minor endogenous estrogens, the biosyntheses of which do not involve
Biological function

The actions of estrogen are mediated by the
While estrogens are present in both
| Ligand | Other names | Relative binding affinities (RBA, %)a |
Absolute binding affinities (Ki, nM)a |
Action | ||
|---|---|---|---|---|---|---|
ERα |
ERβ |
ERα |
ERβ
| |||
| Estradiol | E2; 17β-Estradiol | 100 | 100 | 0.115 (0.04–0.24) | 0.15 (0.10–2.08) | Estrogen |
| Estrone | E1; 17-Ketoestradiol | 16.39 (0.7–60) | 6.5 (1.36–52) | 0.445 (0.3–1.01) | 1.75 (0.35–9.24) | Estrogen |
| Estriol | E3; 16α-OH-17β-E2 | 12.65 (4.03–56) | 26 (14.0–44.6) | 0.45 (0.35–1.4) | 0.7 (0.63–0.7) | Estrogen |
| Estetrol | E4; 15α,16α-Di-OH-17β-E2 | 4.0 | 3.0 | 4.9 | 19 | Estrogen |
| Alfatradiol | 17α-Estradiol | 20.5 (7–80.1) | 8.195 (2–42) | 0.2–0.52 | 0.43–1.2 | Metabolite |
16-Epiestriol |
16β-Hydroxy-17β-estradiol | 7.795 (4.94–63) | 50 | ? | ? | Metabolite |
17-Epiestriol |
16α-Hydroxy-17α-estradiol | 55.45 (29–103) | 79–80 | ? | ? | Metabolite |
16,17-Epiestriol |
16β-Hydroxy-17α-estradiol | 1.0 | 13 | ? | ? | Metabolite |
| 2-Hydroxyestradiol | 2-OH-E2 | 22 (7–81) | 11–35 | 2.5 | 1.3 | Metabolite |
| 2-Methoxyestradiol | 2-MeO-E2 | 0.0027–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Hydroxyestradiol | 4-OH-E2 | 13 (8–70) | 7–56 | 1.0 | 1.9 | Metabolite |
| 4-Methoxyestradiol | 4-MeO-E2 | 2.0 | 1.0 | ? | ? | Metabolite |
| 2-Hydroxyestrone | 2-OH-E1 | 2.0–4.0 | 0.2–0.4 | ? | ? | Metabolite |
| 2-Methoxyestrone | 2-MeO-E1 | <0.001–<1 | <1 | ? | ? | Metabolite |
| 4-Hydroxyestrone | 4-OH-E1 | 1.0–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestrone | 4-MeO-E1 | <1 | <1 | ? | ? | Metabolite |
| 16α-Hydroxyestrone | 16α-OH-E1; 17-Ketoestriol | 2.0–6.5 | 35 | ? | ? | Metabolite |
| 2-Hydroxyestriol | 2-OH-E3 | 2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestriol | 4-MeO-E3 | 1.0 | 1.0 | ? | ? | Metabolite |
| Estradiol sulfate | E2S; Estradiol 3-sulfate | <1 | <1 | ? | ? | Metabolite |
| Estradiol disulfate | Estradiol 3,17β-disulfate | 0.0004 | ? | ? | ? | Metabolite |
| Estradiol 3-glucuronide | E2-3G | 0.0079 | ? | ? | ? | Metabolite |
Estradiol 17β-glucuronide |
E2-17G | 0.0015 | ? | ? | ? | Metabolite |
| Estradiol 3-gluc. 17β-sulfate | E2-3G-17S | 0.0001 | ? | ? | ? | Metabolite |
| Estrone sulfate | E1S; Estrone 3-sulfate | <1 | <1 | >10 | >10 | Metabolite |
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | ? | ? | ? | Estrogen |
| Estradiol 17β-benzoate | E2-17B | 11.3 | 32.6 | ? | ? | Estrogen |
| Estrone methyl ether | Estrone 3-methyl ether | 0.145 | ? | ? | ? | Estrogen |
| ent-Estradiol | 1-Estradiol | 1.31–12.34 | 9.44–80.07 | ? | ? | Estrogen |
| Equilin | 7-Dehydroestrone | 13 (4.0–28.9) | 13.0–49 | 0.79 | 0.36 | Estrogen |
| Equilenin | 6,8-Didehydroestrone | 2.0–15 | 7.0–20 | 0.64 | 0.62 | Estrogen |
| 17β-Dihydroequilin | 7-Dehydro-17β-estradiol | 7.9–113 | 7.9–108 | 0.09 | 0.17 | Estrogen |
| 17α-Dihydroequilin | 7-Dehydro-17α-estradiol | 18.6 (18–41) | 14–32 | 0.24 | 0.57 | Estrogen |
| 17β-Dihydroequilenin | 6,8-Didehydro-17β-estradiol | 35–68 | 90–100 | 0.15 | 0.20 | Estrogen |
| 17α-Dihydroequilenin | 6,8-Didehydro-17α-estradiol | 20 | 49 | 0.50 | 0.37 | Estrogen |
| Δ8-Estradiol | 8,9-Dehydro-17β-estradiol | 68 | 72 | 0.15 | 0.25 | Estrogen |
| Δ8-Estrone | 8,9-Dehydroestrone | 19 | 32 | 0.52 | 0.57 | Estrogen |
| Ethinylestradiol | EE; 17α-Ethynyl-17β-E2 | 120.9 (68.8–480) | 44.4 (2.0–144) | 0.02–0.05 | 0.29–0.81 | Estrogen |
| Mestranol | EE 3-methyl ether | ? | 2.5 | ? | ? | Estrogen |
| Moxestrol | RU-2858; 11β-Methoxy-EE | 35–43 | 5–20 | 0.5 | 2.6 | Estrogen |
| Methylestradiol | 17α-Methyl-17β-estradiol | 70 | 44 | ? | ? | Estrogen |
| Diethylstilbestrol | DES; Stilbestrol | 129.5 (89.1–468) | 219.63 (61.2–295) | 0.04 | 0.05 | Estrogen |
| Hexestrol | Dihydrodiethylstilbestrol | 153.6 (31–302) | 60–234 | 0.06 | 0.06 | Estrogen |
| Dienestrol | Dehydrostilbestrol | 37 (20.4–223) | 56–404 | 0.05 | 0.03 | Estrogen |
| Benzestrol (B2) | – | 114 | ? | ? | ? | Estrogen |
| Chlorotrianisene | TACE | 1.74 | ? | 15.30 | ? | Estrogen |
| Triphenylethylene | TPE | 0.074 | ? | ? | ? | Estrogen |
| Triphenylbromoethylene | TPBE | 2.69 | ? | ? | ? | Estrogen |
| Tamoxifen | ICI-46,474 | 3 (0.1–47) | 3.33 (0.28–6) | 3.4–9.69 | 2.5 | SERM |
| Afimoxifene | 4-Hydroxytamoxifen; 4-OHT | 100.1 (1.7–257) | 10 (0.98–339) | 2.3 (0.1–3.61) | 0.04–4.8 | SERM |
| Toremifene | 4-Chlorotamoxifen; 4-CT | ? | ? | 7.14–20.3 | 15.4 | SERM |
| Clomifene | MRL-41 | 25 (19.2–37.2) | 12 | 0.9 | 1.2 | SERM |
| Cyclofenil | F-6066; Sexovid | 151–152 | 243 | ? | ? | SERM |
| Nafoxidine | U-11,000A | 30.9–44 | 16 | 0.3 | 0.8 | SERM |
| Raloxifene | – | 41.2 (7.8–69) | 5.34 (0.54–16) | 0.188–0.52 | 20.2 | SERM |
| Arzoxifene | LY-353,381 | ? | ? | 0.179 | ? | SERM |
| Lasofoxifene | CP-336,156 | 10.2–166 | 19.0 | 0.229 | ? | SERM |
| Ormeloxifene | Centchroman | ? | ? | 0.313 | ? | SERM |
| Levormeloxifene | 6720-CDRI; NNC-460,020 | 1.55 | 1.88 | ? | ? | SERM |
| Ospemifene | Deaminohydroxytoremifene | 0.82–2.63 | 0.59–1.22 | ? | ? | SERM |
| Bazedoxifene | – | ? | ? | 0.053 | ? | SERM |
| Etacstil | GW-5638 | 4.30 | 11.5 | ? | ? | SERM |
ICI-164,384 |
– | 63.5 (3.70–97.7) | 166 | 0.2 | 0.08 | Antiestrogen |
| Fulvestrant | ICI-182,780 | 43.5 (9.4–325) | 21.65 (2.05–40.5) | 0.42 | 1.3 | Antiestrogen |
| Propylpyrazoletriol | PPT | 49 (10.0–89.1) | 0.12 | 0.40 | 92.8 | ERα agonist |
| 16α-LE2 | 16α-Lactone-17β-estradiol | 14.6–57 | 0.089 | 0.27 | 131 | ERα agonist |
| 16α-Iodo-E2 | 16α-Iodo-17β-estradiol | 30.2 | 2.30 | ? | ? | ERα agonist |
| Methylpiperidinopyrazole | MPP | 11 | 0.05 | ? | ? | ERα antagonist |
| Diarylpropionitrile | DPN | 0.12–0.25 | 6.6–18 | 32.4 | 1.7 | ERβ agonist |
| 8β-VE2 | 8β-Vinyl-17β-estradiol | 0.35 | 22.0–83 | 12.9 | 0.50 | ERβ agonist |
| Prinaberel | ERB-041; WAY-202,041 | 0.27 | 67–72 | ? | ? | ERβ agonist |
| ERB-196 | WAY-202,196 | ? | 180 | ? | ? | ERβ agonist |
| Erteberel | SERBA-1; LY-500,307 | ? | ? | 2.68 | 0.19 | ERβ agonist |
| SERBA-2 | – | ? | ? | 14.5 | 1.54 | ERβ agonist |
| Coumestrol | – | 9.225 (0.0117–94) | 64.125 (0.41–185) | 0.14–80.0 | 0.07–27.0 | Xenoestrogen |
| Genistein | – | 0.445 (0.0012–16) | 33.42 (0.86–87) | 2.6–126 | 0.3–12.8 | Xenoestrogen |
| Equol | – | 0.2–0.287 | 0.85 (0.10–2.85) | ? | ? | Xenoestrogen |
| Daidzein | – | 0.07 (0.0018–9.3) | 0.7865 (0.04–17.1) | 2.0 | 85.3 | Xenoestrogen |
| Biochanin A | – | 0.04 (0.022–0.15) | 0.6225 (0.010–1.2) | 174 | 8.9 | Xenoestrogen |
| Kaempferol | – | 0.07 (0.029–0.10) | 2.2 (0.002–3.00) | ? | ? | Xenoestrogen |
| Naringenin | – | 0.0054 (<0.001–0.01) | 0.15 (0.11–0.33) | ? | ? | Xenoestrogen |
| 8-Prenylnaringenin | 8-PN | 4.4 | ? | ? | ? | Xenoestrogen |
| Quercetin | – | <0.001–0.01 | 0.002–0.040 | ? | ? | Xenoestrogen |
| Ipriflavone | – | <0.01 | <0.01 | ? | ? | Xenoestrogen |
| Miroestrol | – | 0.39 | ? | ? | ? | Xenoestrogen |
Deoxymiroestrol |
– | 2.0 | ? | ? | ? | Xenoestrogen |
β-Sitosterol |
– | <0.001–0.0875 | <0.001–0.016 | ? | ? | Xenoestrogen |
| Resveratrol | – | <0.001–0.0032 | ? | ? | ? | Xenoestrogen |
| α-Zearalenol | – | 48 (13–52.5) | ? | ? | ? | Xenoestrogen |
| β-Zearalenol | – | 0.6 (0.032–13) | ? | ? | ? | Xenoestrogen |
| Zeranol | α-Zearalanol | 48–111 | ? | ? | ? | Xenoestrogen |
| Taleranol | β-Zearalanol | 16 (13–17.8) | 14 | 0.8 | 0.9 | Xenoestrogen |
| Zearalenone | ZEN | 7.68 (2.04–28) | 9.45 (2.43–31.5) | ? | ? | Xenoestrogen |
| Zearalanone | ZAN | 0.51 | ? | ? | ? | Xenoestrogen |
| Bisphenol A | BPA | 0.0315 (0.008–1.0) | 0.135 (0.002–4.23) | 195 | 35 | Xenoestrogen |
| Endosulfan | EDS | <0.001–<0.01 | <0.01 | ? | ? | Xenoestrogen |
Kepone |
Chlordecone | 0.0069–0.2 | ? | ? | ? | Xenoestrogen |
o,p'-DDT |
– | 0.0073–0.4 | ? | ? | ? | Xenoestrogen |
p,p'-DDT |
– | 0.03 | ? | ? | ? | Xenoestrogen |
| Methoxychlor | p,p'-Dimethoxy-DDT | 0.01 (<0.001–0.02) | 0.01–0.13 | ? | ? | Xenoestrogen |
| HPTE | Hydroxychlor; p,p'-OH-DDT | 1.2–1.7 | ? | ? | ? | Xenoestrogen |
| Testosterone | T; 4-Androstenolone | <0.0001–<0.01 | <0.002–0.040 | >5000 | >5000 | Androgen |
| Dihydrotestosterone | DHT; 5α-Androstanolone | 0.01 (<0.001–0.05) | 0.0059–0.17 | 221–>5000 | 73–1688 | Androgen |
| Nandrolone | 19-Nortestosterone; 19-NT | 0.01 | 0.23 | 765 | 53 | Androgen |
| Dehydroepiandrosterone | DHEA; Prasterone | 0.038 (<0.001–0.04) | 0.019–0.07 | 245–1053 | 163–515 | Androgen |
5-Androstenediol |
A5; Androstenediol | 6 | 17 | 3.6 | 0.9 | Androgen |
| 4-Androstenediol | – | 0.5 | 0.6 | 23 | 19 | Androgen |
4-Androstenedione |
A4; Androstenedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| 3α-Androstanediol | 3α-Adiol | 0.07 | 0.3 | 260 | 48 | Androgen |
| 3β-Androstanediol | 3β-Adiol | 3 | 7 | 6 | 2 | Androgen |
| Androstanedione | 5α-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Etiocholanedione | 5β-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Methyltestosterone | 17α-Methyltestosterone | <0.0001 | ? | ? | ? | Androgen |
Ethinyl-3α-androstanediol |
17α-Ethynyl-3α-adiol | 4.0 | <0.07 | ? | ? | Estrogen |
Ethinyl-3β-androstanediol |
17α-Ethynyl-3β-adiol | 50 | 5.6 | ? | ? | Estrogen |
| Progesterone | P4; 4-Pregnenedione | <0.001–0.6 | <0.001–0.010 | ? | ? | Progestogen |
| Norethisterone | NET; 17α-Ethynyl-19-NT | 0.085 (0.0015–<0.1) | 0.1 (0.01–0.3) | 152 | 1084 | Progestogen |
Norethynodrel |
5(10)-Norethisterone | 0.5 (0.3–0.7) | <0.1–0.22 | 14 | 53 | Progestogen |
| Tibolone | 7α-Methylnorethynodrel | 0.5 (0.45–2.0) | 0.2–0.076 | ? | ? | Progestogen |
| Δ4-Tibolone | 7α-Methylnorethisterone | 0.069–<0.1 | 0.027–<0.1 | ? | ? | Progestogen |
| 3α-Hydroxytibolone | – | 2.5 (1.06–5.0) | 0.6–0.8 | ? | ? | Progestogen |
| 3β-Hydroxytibolone | – | 1.6 (0.75–1.9) | 0.070–0.1 | ? | ? | Progestogen |
| Footnotes: a = (1) ERβ proteins (except the ERβ values from Kuiper et al. (1997), which are rat ERβ). Sources: See template page.
| ||||||
| Estrogen | Relative binding affinities (%)
| ||||||
|---|---|---|---|---|---|---|---|
| ER | AR | PR | GR | MR | SHBG | CBG
| |
| Estradiol | 100 | 7.9 | 2.6 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Estradiol benzoate | ? | ? | ? | ? | ? | <0.1–0.16 | <0.1 |
| Estradiol valerate | 2 | ? | ? | ? | ? | ? | ? |
| Estrone | 11–35 | <1 | <1 | <1 | <1 | 2.7 | <0.1 |
| Estrone sulfate | 2 | 2 | ? | ? | ? | ? | ? |
| Estriol | 10–15 | <1 | <1 | <1 | <1 | <0.1 | <0.1 |
| Equilin | 40 | ? | ? | ? | ? | ? | 0 |
| Alfatradiol | 15 | <1 | <1 | <1 | <1 | ? | ? |
| Epiestriol | 20 | <1 | <1 | <1 | <1 | ? | ? |
| Ethinylestradiol | 100–112 | 1–3 | 15–25 | 1–3 | <1 | 0.18 | <0.1 |
| Mestranol | 1 | ? | ? | ? | ? | <0.1 | <0.1 |
| Methylestradiol | 67 | 1–3 | 3–25 | 1–3 | <1 | ? | ? |
| Moxestrol | 12 | <0.1 | 0.8 | 3.2 | <0.1 | <0.2 | <0.1 |
| Diethylstilbestrol | ? | ? | ? | ? | ? | <0.1 | <0.1 |
| Notes: Reference CBG . Sources: See template.
| |||||||
| Estrogen | Other names | RBA (%)a
|
REP (%)b | |||
|---|---|---|---|---|---|---|
| ER | ERα
|
ERβ
| ||||
| Estradiol | E2 | 100 | 100 | 100 | ||
Estradiol 3-sulfate
|
E2S; E2-3S | ? | 0.02 | 0.04 | ||
| Estradiol 3-glucuronide | E2-3G | ? | 0.02 | 0.09 | ||
Estradiol 17β-glucuronide
|
E2-17G | ? | 0.002 | 0.0002 | ||
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | 1.1 | 0.52 | ||
| Estradiol 17β-acetate | E2-17A | 31–45 | 24 | ? | ||
| Estradiol diacetate | EDA; Estradiol 3,17β-diacetate | ? | 0.79 | ? | ||
Estradiol propionate
|
EP; Estradiol 17β-propionate | 19–26 | 2.6 | ? | ||
| Estradiol valerate | EV; Estradiol 17β-valerate | 2–11 | 0.04–21 | ? | ||
| Estradiol cypionate | EC; Estradiol 17β-cypionate | ?c | 4.0 | ? | ||
| Estradiol palmitate | Estradiol 17β-palmitate | 0 | ? | ? | ||
| Estradiol stearate | Estradiol 17β-stearate | 0 | ? | ? | ||
| Estrone | E1; 17-Ketoestradiol | 11 | 5.3–38 | 14 | ||
| Estrone sulfate | E1S; Estrone 3-sulfate | 2 | 0.004 | 0.002 | ||
| Estrone glucuronide | E1G; Estrone 3-glucuronide | ? | <0.001 | 0.0006 | ||
| Ethinylestradiol | EE; 17α-Ethynylestradiol | 100 | 17–150 | 129 | ||
| Mestranol | EE 3-methyl ether | 1 | 1.3–8.2 | 0.16 | ||
| Quinestrol | EE 3-cyclopentyl ether | ? | 0.37 | ? | ||
| Footnotes: a = ERβ. Both mammalian cells and yeast have the capacity to hydrolyze estrogen esters. c = The affinities of estradiol cypionate for the ERs are similar to those of estradiol valerate and estradiol benzoate (figure ). Sources: See template page.
| ||||||
| Estrogen | RBA (%) |
Uterine weight (%) | Uterotrophy |
LH levels (%) | RBA (%)
|
|---|---|---|---|---|---|
| Control | – | 100 | – | 100 | – |
| Estradiol (E2) | 100 | 506 ± 20 | +++ | 12–19 | 100 |
| Estrone (E1) | 11 ± 8 | 490 ± 22 | +++ | ? | 20 |
| Estriol (E3) | 10 ± 4 | 468 ± 30 | +++ | 8–18 | 3 |
| Estetrol (E4) | 0.5 ± 0.2 | ? | Inactive | ? | 1 |
| 17α-Estradiol | 4.2 ± 0.8 | ? | ? | ? | ? |
| 2-Hydroxyestradiol | 24 ± 7 | 285 ± 8 | +b | 31–61 | 28 |
| 2-Methoxyestradiol | 0.05 ± 0.04 | 101 | Inactive | ? | 130 |
| 4-Hydroxyestradiol | 45 ± 12 | ? | ? | ? | ? |
| 4-Methoxyestradiol | 1.3 ± 0.2 | 260 | ++ | ? | 9 |
| 4-Fluoroestradiola | 180 ± 43 | ? | +++ | ? | ? |
| 2-Hydroxyestrone | 1.9 ± 0.8 | 130 ± 9 | Inactive | 110–142 | 8 |
| 2-Methoxyestrone | 0.01 ± 0.00 | 103 ± 7 | Inactive | 95–100 | 120 |
| 4-Hydroxyestrone | 11 ± 4 | 351 | ++ | 21–50 | 35 |
| 4-Methoxyestrone | 0.13 ± 0.04 | 338 | ++ | 65–92 | 12 |
| 16α-Hydroxyestrone | 2.8 ± 1.0 | 552 ± 42 | +++ | 7–24 | <0.5 |
| 2-Hydroxyestriol | 0.9 ± 0.3 | 302 | +b | ? | ? |
| 2-Methoxyestriol | 0.01 ± 0.00 | ? | Inactive | ? | 4 |
| Notes: Values are mean ± SD or range. ER RBA = endogenous ). b = Atypical uterotrophic effect which plateaus within 48 hours (estradiol's uterotrophy continues linearly up to 72 hours). Sources: See template.
| |||||
Overview of actions
This section is in prose. is available. (October 2019) |
- Musculoskeletal
- connective tissues, tendons, and ligaments, but also decreases stiffness of tendons and ligaments (especially during menstruation). Decreased stiffness of tendons gives women much lower predisposition to muscle strains but soft ligaments are much more prone to injuries (ACL tears are 2-8x more common among women than men).[26][27][28][29]
- Reduce bone resorption, increase bone formation
- In mice, estrogen has been shown to increase the proportion of the fastest-twitch (type IIX) muscle fibers by over 40%.[30]
- Metabolic
- Anti-inflammatory properties
- Accelerate metabolism
- Estradiol also regulates energy expenditure, body weight homeostasis, and seems to have much stronger anti-obesity effects than testosterone in general.[34]
- Other structural
- Maintenance of vessels and skin
- Protein synthesis
- Increase hepatic production of binding proteins
- Increase production of the hepatokine adropin.[35]
- Increase
- Coagulation
- Increase circulating level of plasminogen
- Decrease antithrombin III
- Increase platelet adhesiveness
- Increase vWF (estrogen -> Angiotensin II -> Vasopressin)
- Increase PAI-1 and PAI-2also through Angiotensin II
- Increase circulating level of
- Lipid
- Increase HDL, triglyceride
- Decrease LDL, fat deposition
- Increase
- Fluid balance
- Salt (sodium) and water retention
- Increase SHBG
- Gastrointestinal tract
- Reduce bowel motility
- Increase cholesterol in bile
- Melanin
- Estrogen is known to cause darkening of skin, especially in the face and areolae.[36] Pale skinned women will develop browner and yellower skin during pregnancy, as a result of the increase of estrogen, known as the "mask of pregnancy".[37]
- Estrogen is known to cause darkening of skin, especially in the face and
- Cancer
- Support hormone-sensitive breast cancers (see section below)
- Lung function
- Sexual
- Mediate formation of female secondary sex characteristics
- Stimulate endometrial growth
- Increase uterine growth
- Increase vaginal lubrication
- Thicken the vaginal wall
- Mediate formation of female
- Uterus lining
- Estrogen together with progesterone promotes and maintains the uterus lining in preparation for implantation of fertilized egg and maintenance of uterus function during gestation period, also upregulates oxytocin receptor in myometrium
- Ovulation
- Surge in estrogen level induces the release of Graafian follicle in the ovary.
- Surge in estrogen level induces the release of
- Sexual behavior
- Estrogen is required for female mammals to engage in ventromedial nucleus of the hypothalamus.[41]
- Sex drive is dependent on androgen levels[42] only in the presence of estrogen, but without estrogen, free testosterone level actually decreases sexual desire (instead of increasing sex drive), as demonstrated for those women who have hypoactive sexual desire disorder, and the sexual desire in these women can be restored by administration of estrogen (using oral contraceptive).[43]
- Estrogen is required for female mammals to engage in
Female pubertal development
Estrogens are responsible for the development of female
Breast development
Estrogen, in conjunction with
Androgens such as testosterone powerfully oppose estrogen action in the breasts, such as by reducing estrogen receptor expression in them.[53][54]
Female reproductive system
Estrogens are responsible for maturation and maintenance of the vagina and uterus, and are also involved in ovarian function, such as maturation of ovarian follicles. In addition, estrogens play an important role in regulation of gonadotropin secretion. For these reasons, estrogens are required for female fertility.
Neuroprotection and DNA repair
Estrogen regulated DNA repair mechanisms in the brain have neuroprotective effects.[55] Estrogen regulates the transcription of DNA base excision repair genes as well as the translocation of the base excision repair enzymes between different subcellular compartments.
Brain and behavior
Sex drive
Estrogens are involved in libido (sex drive) in both women and men.
Cognition
Verbal memory scores are frequently used as one measure of higher level cognition. These scores vary in direct proportion to estrogen levels throughout the menstrual cycle, pregnancy, and menopause. Furthermore, estrogens when administered shortly after natural or surgical menopause prevents decreases in verbal memory. In contrast, estrogens have little effect on verbal memory if first administered years after menopause.[56] Estrogens also have positive influences on other measures of cognitive function.[57] However the effect of estrogens on cognition is not uniformly favorable and is dependent on the timing of the dose and the type of cognitive skill being measured.[58]
The protective effects of estrogens on cognition may be mediated by estrogen's anti-inflammatory effects in the brain.[59] Studies have also shown that the Met allele gene and level of estrogen mediates the efficiency of prefrontal cortex dependent working memory tasks.[60][61] Researchers have urged for further research to illuminate the role of estrogen and its potential for improvement on cognitive function.[62]
Mental health
Estrogen is considered to play a significant role in women's
Compulsions in male lab mice, such as those in obsessive-compulsive disorder (OCD), may be caused by low estrogen levels. When estrogen levels were raised through the increased activity of the enzyme
Local application of estrogen in the rat hippocampus has been shown to inhibit the re-uptake of serotonin. Contrarily, local application of estrogen has been shown to block the ability of fluvoxamine to slow serotonin clearance, suggesting that the same pathways which are involved in SSRI efficacy may also be affected by components of local estrogen signaling pathways.[69]
Parenthood
Studies have also found that fathers had lower levels of cortisol and testosterone but higher levels of estrogen (estradiol) than did non-fathers.[70]
Binge eating
Estrogen may play a role in suppressing binge eating. Hormone replacement therapy using estrogen may be a possible treatment for binge eating behaviors in females. Estrogen replacement has been shown to suppress binge eating behaviors in female mice.[71] The mechanism by which estrogen replacement inhibits binge-like eating involves the replacement of serotonin (5-HT) neurons. Women exhibiting binge eating behaviors are found to have increased brain uptake of neuron 5-HT, and therefore less of the neurotransmitter serotonin in the cerebrospinal fluid.[72] Estrogen works to activate 5-HT neurons, leading to suppression of binge like eating behaviors.[71]
It is also suggested that there is an interaction between hormone levels and eating at different points in the female menstrual cycle. Research has predicted increased emotional eating during hormonal flux, which is characterized by high progesterone and estradiol levels that occur during the mid-luteal phase. It is hypothesized that these changes occur due to brain changes across the menstrual cycle that are likely a genomic effect of hormones. These effects produce menstrual cycle changes, which result in hormone release leading to behavioral changes, notably binge and emotional eating. These occur especially prominently among women who are genetically vulnerable to binge eating phenotypes.[73]
Binge eating is associated with decreased estradiol and increased progesterone.[74] Klump et al.[75] Progesterone may moderate the effects of low estradiol (such as during dysregulated eating behavior), but that this may only be true in women who have had clinically diagnosed binge episodes (BEs). Dysregulated eating is more strongly associated with such ovarian hormones in women with BEs than in women without BEs.[75]
The implantation of 17β-estradiol pellets in ovariectomized mice significantly reduced binge eating behaviors and injections of GLP-1 in ovariectomized mice decreased binge-eating behaviors.[71]
The associations between binge eating, menstrual-cycle phase and ovarian hormones correlated.[74][76][77]
Masculinization in rodents
In rodents, estrogens (which are locally aromatized from androgens in the brain) play an important role in psychosexual differentiation, for example, by masculinizing territorial behavior;[78] the same is not true in humans.[79] In humans, the masculinizing effects of prenatal androgens on behavior (and other tissues, with the possible exception of effects on bone) appear to act exclusively through the androgen receptor.[80] Consequently, the utility of rodent models for studying human psychosexual differentiation has been questioned.[81]
Skeletal system
Estrogens are responsible for both the pubertal growth spurt, which causes an acceleration in linear growth, and
Cardiovascular system
Women are less impacted by heart disease due to vasculo-protective action of estrogen which helps in preventing atherosclerosis.
| Absolute incidence of first VTE per 10,000 person–years during pregnancy and the postpartum period | ||||||||
|---|---|---|---|---|---|---|---|---|
| Swedish data A | Swedish data B | English data | Danish data | |||||
| Time period | N | Rate (95% CI) | N | Rate (95% CI) | N | Rate (95% CI) | N | Rate (95% CI) |
| Outside pregnancy | 1105 | 4.2 (4.0–4.4) | 1015 | 3.8 (?) | 1480 | 3.2 (3.0–3.3) | 2895 | 3.6 (3.4–3.7) |
| Antepartum | 995 | 20.5 (19.2–21.8) | 690 | 14.2 (13.2–15.3) | 156 | 9.9 (8.5–11.6) | 491 | 10.7 (9.7–11.6) |
| Trimester 1 | 207 | 13.6 (11.8–15.5) | 172 | 11.3 (9.7–13.1) | 23 | 4.6 (3.1–7.0) | 61 | 4.1 (3.2–5.2) |
| Trimester 2 | 275 | 17.4 (15.4–19.6) | 178 | 11.2 (9.7–13.0) | 30 | 5.8 (4.1–8.3) | 75 | 5.7 (4.6–7.2) |
| Trimester 3 | 513 | 29.2 (26.8–31.9) | 340 | 19.4 (17.4–21.6) | 103 | 18.2 (15.0–22.1) | 355 | 19.7 (17.7–21.9) |
| Around delivery | 115 | 154.6 (128.8–185.6) | 79 | 106.1 (85.1–132.3) | 34 | 142.8 (102.0–199.8) | –
| |
| Postpartum | 649 | 42.3 (39.2–45.7) | 509 | 33.1 (30.4–36.1) | 135 | 27.4 (23.1–32.4) | 218 | 17.5 (15.3–20.0) |
| Early postpartum | 584 | 75.4 (69.6–81.8) | 460 | 59.3 (54.1–65.0) | 177 | 46.8 (39.1–56.1) | 199 | 30.4 (26.4–35.0) |
| Late postpartum | 65 | 8.5 (7.0–10.9) | 49 | 6.4 (4.9–8.5) | 18 | 7.3 (4.6–11.6) | 319 | 3.2 (1.9–5.0) |
| Incidence rate ratios (IRRs) of first VTE during pregnancy and the postpartum period | ||||||||
| Swedish data A | Swedish data B | English data | Danish data | |||||
| Time period | IRR* (95% CI) | IRR* (95% CI) | IRR (95% CI)† | IRR (95% CI)† | ||||
| Outside pregnancy | Reference (i.e., 1.00)
| |||||||
| Antepartum | 5.08 (4.66–5.54) | 3.80 (3.44–4.19) | 3.10 (2.63–3.66) | 2.95 (2.68–3.25) | ||||
| Trimester 1 | 3.42 (2.95–3.98) | 3.04 (2.58–3.56) | 1.46 (0.96–2.20) | 1.12 (0.86–1.45) | ||||
| Trimester 2 | 4.31 (3.78–4.93) | 3.01 (2.56–3.53) | 1.82 (1.27–2.62) | 1.58 (1.24–1.99) | ||||
| Trimester 3 | 7.14 (6.43–7.94) | 5.12 (4.53–5.80) | 5.69 (4.66–6.95) | 5.48 (4.89–6.12) | ||||
| Around delivery | 37.5 (30.9–44.45) | 27.97 (22.24–35.17) | 44.5 (31.68–62.54) | –
| ||||
| Postpartum | 10.21 (9.27–11.25) | 8.72 (7.83–9.70) | 8.54 (7.16–10.19) | 4.85 (4.21–5.57) | ||||
| Early postpartum | 19.27 (16.53–20.21) | 15.62 (14.00–17.45) | 14.61 (12.10–17.67) | 8.44 (7.27–9.75) | ||||
| Late postpartum | 2.06 (1.60–2.64) | 1.69 (1.26–2.25) | 2.29 (1.44–3.65) | 0.89 (0.53–1.39) | ||||
| Notes: Swedish data A = Using any code for VTE regardless of confirmation. Swedish data B = Using only algorithm-confirmed VTE. Early postpartum = First 6 weeks after delivery. Late postpartum = More than 6 weeks after delivery. * = Adjusted for age and calendar year. † = Unadjusted ratio calculated based on the data provided. Source: [84] | ||||||||
Immune system
The effect of estrogen on the
On a molecular level estrogen induces the above-mentioned effects on cell via acting on intracellular
Associated conditions
Researchers have implicated estrogens in various
High estrogen can amplify stress-hormone responses in stressful situations.[90]
Biochemistry
Biosynthesis
Estrogens, in females, are produced primarily by the ovaries, and during pregnancy, the placenta.[92] Follicle-stimulating hormone (FSH) stimulates the ovarian production of estrogens by the granulosa cells of the ovarian follicles and corpora lutea. Some estrogens are also produced in smaller amounts by other tissues such as the liver, pancreas, bone, adrenal glands, skin, brain, adipose tissue,[93] and the breasts.[94] These secondary sources of estrogens are especially important in postmenopausal women.[95] The pathway of estrogen biosynthesis in extragonadal tissues is different. These tissues are not able to synthesize C19 steroids, and therefore depend on C19 supplies from other tissues[95] and the level of aromatase.[96]
In females, synthesis of estrogens starts in
Estrogen levels vary through the menstrual cycle, with levels highest near the end of the follicular phase just before ovulation.
Note that in males, estrogen is also produced by the Sertoli cells when FSH binds to their FSH receptors.
| Sex | Sex hormone | Reproductive phase |
Blood production rate |
Gonadal secretion rate |
Metabolic clearance rate |
Reference range (serum levels) | |
|---|---|---|---|---|---|---|---|
| SI units | Non-SI units | ||||||
| Men | Androstenedione | –
|
2.8 mg/day | 1.6 mg/day | 2200 L/day | 2.8–7.3 nmol/L | 80–210 ng/dL |
| Testosterone | –
|
6.5 mg/day | 6.2 mg/day | 950 L/day | 6.9–34.7 nmol/L | 200–1000 ng/dL | |
| Estrone | –
|
150 μg/day | 110 μg/day | 2050 L/day | 37–250 pmol/L | 10–70 pg/mL | |
| Estradiol | –
|
60 μg/day | 50 μg/day | 1600 L/day | <37–210 pmol/L | 10–57 pg/mL | |
| Estrone sulfate | –
|
80 μg/day | Insignificant | 167 L/day | 600–2500 pmol/L | 200–900 pg/mL | |
| Women | Androstenedione | –
|
3.2 mg/day | 2.8 mg/day | 2000 L/day | 3.1–12.2 nmol/L | 89–350 ng/dL |
| Testosterone | –
|
190 μg/day | 60 μg/day | 500 L/day | 0.7–2.8 nmol/L | 20–81 ng/dL | |
| Estrone | Follicular phase | 110 μg/day | 80 μg/day | 2200 L/day | 110–400 pmol/L | 30–110 pg/mL | |
| Luteal phase | 260 μg/day | 150 μg/day | 2200 L/day | 310–660 pmol/L | 80–180 pg/mL | ||
| Postmenopause | 40 μg/day | Insignificant | 1610 L/day | 22–230 pmol/L | 6–60 pg/mL | ||
| Estradiol | Follicular phase | 90 μg/day | 80 μg/day | 1200 L/day | <37–360 pmol/L | 10–98 pg/mL | |
| Luteal phase | 250 μg/day | 240 μg/day | 1200 L/day | 699–1250 pmol/L | 190–341 pg/mL | ||
| Postmenopause | 6 μg/day | Insignificant | 910 L/day | <37–140 pmol/L | 10–38 pg/mL | ||
| Estrone sulfate | Follicular phase | 100 μg/day | Insignificant | 146 L/day | 700–3600 pmol/L | 250–1300 pg/mL | |
| Luteal phase | 180 μg/day | Insignificant | 146 L/day | 1100–7300 pmol/L | 400–2600 pg/mL | ||
| Progesterone | Follicular phase | 2 mg/day | 1.7 mg/day | 2100 L/day | 0.3–3 nmol/L | 0.1–0.9 ng/mL | |
| Luteal phase | 25 mg/day | 24 mg/day | 2100 L/day | 19–45 nmol/L | 6–14 ng/mL | ||
Notes and sources
Notes: "The concentration of a steroid in the circulation is determined by the rate at which it is secreted from glands, the rate of metabolism of precursor or prehormones into the steroid, and the rate at which it is extracted by tissues and metabolized. The secretion rate of a steroid refers to the total secretion of the compound from a gland per unit time. Secretion rates have been assessed by sampling the venous effluent from a gland over time and subtracting out the arterial and peripheral venous hormone concentration. The metabolic clearance rate of a steroid is defined as the volume of blood that has been completely cleared of the hormone per unit time. The production rate of a steroid hormone refers to entry into the blood of the compound from all possible sources, including secretion from glands and conversion of prohormones into the steroid of interest. At steady state, the amount of hormone entering the blood from all sources will be equal to the rate at which it is being cleared (metabolic clearance rate) multiplied by blood concentration (production rate = metabolic clearance rate × concentration). If there is little contribution of prohormone metabolism to the circulating pool of steroid, then the production rate will approximate the secretion rate." Sources: See template. | |||||||
Distribution
Estrogens are plasma protein bound to albumin and/or sex hormone-binding globulin in the circulation.
Metabolism
Estrogens are
Estrogen metabolism in humans
hydroxyl (–OH) groups . Sources: See template page. |
Excretion
Estrogens are inactivated primarily by the
Medical use
Estrogens are used as medications, mainly in hormonal contraception, hormone replacement therapy,[99] and to treat gender dysphoria in transgender women and other transfeminine individuals as part of feminizing hormone therapy.[100]
Chemistry
The estrogen steroid hormones are estrane steroids.
History
In 1929,
The word estrogen derives from Ancient Greek. It is derived from "oestros"[102] (a periodic state of sexual activity in female mammals), and genos (generating).[102] It was first published in the early 1920s and referenced as "oestrin".[103] With the years, American English adapted the spelling of estrogen to fit with its phonetic pronunciation.
Society and culture
Etymology
The name estrogen is derived from the Greek οἶστρος (oîstros), literally meaning "verve" or "inspiration" but figuratively sexual passion or desire,[104] and the suffix -gen, meaning "producer of".
Environment
A range of synthetic and natural substances that possess estrogenic activity have been identified in the environment and are referred to xenoestrogens.[105]
- Synthetic substances such as bisphenol A as well as metalloestrogens (e.g., cadmium).
- Plant products with estrogenic activity are called phytoestrogens (e.g., coumestrol, daidzein, genistein, miroestrol).
- Those produced by fungi are known as mycoestrogens (e.g., zearalenone).
Estrogens are among the wide range of endocrine-disrupting compounds (EDCs) because they have high estrogenic potency. When an EDC makes its way into the environment, it may cause male reproductive dysfunction to wildlife and humans.[12][13] The estrogen excreted from farm animals makes its way into fresh water systems.[106][107] During the germination period of reproduction the fish are exposed to low levels of estrogen which may cause reproductive dysfunction to male fish.[108][109]
Cosmetics
Some hair
In addition to being considered misbranded drugs, products claiming to contain placental extract may also be deemed to be misbranded cosmetics if the extract has been prepared from placentas from which the hormones and other biologically active substances have been removed and the extracted substance consists principally of protein. The FDA recommends that this substance be identified by a name other than "placental extract" and describing its composition more accurately because consumers associate the name "placental extract" with a therapeutic use of some biological activity.[111]
See also
- List of steroid abbreviations
- Breastfeeding and fertility
References
- ^ ISBN 978-0-32-367281-8.
Estrogen is a generic term for any of three similar hormones derived from cholesterol: estradiol, estrone, and estriol.
- ISBN 978-8-13-124941-3.
The natural estrogens are steroids. However, typical estrogenic activity is also shown by chemicals which are not steroids. Hence, the term 'estrogen' is used as a generic term to describe all the compounds having estrogenic activity.
- PMID 30855848.
Estrogen is a steroid hormone associated with the female reproductive organs and is responsible for the development of female sexual characteristics. Estrogen is often referred to as estrone, estradiol, and estriol. ... Synthetic estrogen is also available for clinical use, designed to increase absorption and effectiveness by altering the estrogen chemical structure for topical or oral administration. Synthetic steroid estrogens include ethinyl estradiol, estradiol valerate, estropipate, conjugate esterified estrogen, and quinestrol.
- PMID 7083198.
- S2CID 31950471.
- PMID 12007895.
- S2CID 36834775.
- PMID 20821847.
- PMID 23756388.
- PMID 22538318.
- PMID 17222505.
- ^ S2CID 93975613.
- ^ OCLC 44957536.
- ISBN 978-3-642-96158-8.
- S2CID 11803629.
- PMID 23123738.
- PMID 10865186.
- ISSN 2002-4436.
- PMID 15345050.
- PMID 21117950.
- ISBN 978-0-7637-2765-9. Retrieved 12 October 2023.
- ^ Raloff J (6 December 1997). "Science News Online (12/6/97): Estrogen's Emerging Manly Alter Ego". Science News. Retrieved 4 March 2008.
- PMID 9393999.
- ^ "Estrogen Linked To Sperm Count, Male Fertility". Science Blog. Archived from the original on 7 May 2007. Retrieved 4 March 2008.
- S2CID 25280077.
- PMID 30697162.
- PMID 20335737.
- S2CID 24198956.
- S2CID 44446541.
- PMID 25559153. "Supplementation with estrogen increases the type-IIX percentage composition in the plantaris back to 42%. (70)"
- PMID 30097511.
- PMID 20441773.
- PMID 19696765.
- PMID 29224100.
- ^ PMID 35364299.
- ISBN 978-1-9751-8152-9. Retrieved 12 October 2023.
- ISBN 978-1-4496-4851-0. Retrieved 12 October 2023.
- S2CID 24642944. Archived from the original(PDF) on 25 February 2019.
- PMID 22438212.
- PMID 21851428.
- S2CID 28276218.
- S2CID 24557071.
- S2CID 20807391.
- PMID 20739412.
- S2CID 30440069.
- ^ ISBN 978-0-12-387584-6.
- ^ ISBN 978-0-08-091906-5.
- ^ ISBN 978-0-7020-3489-3.
- ISBN 978-1-58603-653-9.
- ISBN 978-3-13-149521-1.
- ISBN 978-0-7817-7427-7.
- ISBN 978-0-323-29296-2.
- ISBN 978-1-4557-2758-2.
- ISBN 978-0-323-24147-2.
- PMID 29311911.
- PMID 22004260.
- PMID 26109339.
- PMID 26149525.
- PMID 26774208.
- PMID 21471363.
- PMID 24778605.
- S2CID 10122688.
- S2CID 9172877.
- S2CID 8205014.
- S2CID 37965502.
- PMID 12085678.
- ^ "PMDD/PMS". The Massachusetts General Hospital Center for Women's Mental Health. Retrieved 12 January 2019.
- S2CID 22669945.
- PMID 22225849.
- PMID 11393496.
- ^ PMID 25157819.
- S2CID 31058047.
- PMID 22889242.
- ^ S2CID 36609028.
- ^ PMID 25343062.
- PMID 18307829.
- S2CID 21497515.
- PMID 19804754.
- S2CID 22116130.
- S2CID 20480423. Archived from the original(PDF) on 26 February 2019.
- S2CID 7465192.
- PMID 10500455.
- PMID 21836070.
- PMID 26560059.
- ^ S2CID 37931012.
- PMID 30210492.
- ^ S2CID 9137227.
- PMID 26779182.
- ^ PMID 25682174.
- ^
ISBN 9780973827521. Retrieved 24 July 2021.
[...] high estrogen amplifies your stress hormone responses to stressful things [...]
- ISSN 2002-4436.
- ISBN 978-0-321-88760-3.
- PMID 4815174.
- PMID 27530684.
- ^ PMID 11511861.
- S2CID 37344052.
- ISBN 978-3-642-96158-8. Retrieved 12 October 2023.
- PMID 31036290.
Physiologically, the metabolic conversion of estrogens allows their excretion from the body via urine, feces, and/or bile, along with the production of estrogen analogs, which have been shown to present antiproliferative effects (Tsuchiya et al., 2005).
- S2CID 24616324.
- PMID 28159148.
- PMID 15940278.
- ^ a b "Origin in Biomedical Terms: oestrogen or oestrogen". Bioetymology. Retrieved 24 January 2018.
- .
- Perseus Digital Library. Retrieved 28 December 2011.
- PMID 11258977.
- PMID 20977246.
- ^ Peach S. "Don't Blame The Pill | Latest News". Chemical & Engineering News. Retrieved 22 April 2023.
- PMID 16203238.
- PMID 16818244.
- ^ Sanghavi DM (17 October 2006). "Preschool Puberty, and a Search for the Causes". The New York Times. Retrieved 4 June 2008.
- ^ a b FDA (February 1995). "Products containing estrogenic hormones, placental extract or vitamins". Guide to Inspections of Cosmetic Product Manufacturers. Archived from the original on 14 October 2007. Retrieved 24 October 2006.
External links
- Nussey and Whitehead: Endocrinology, an integrated approach, Taylor and Francis 2001. Free online textbook.