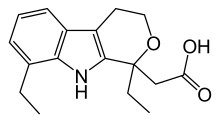
Etodolac
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692015 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 100% |
| Metabolism | liver |
| Elimination half-life | 7.3 ± 4.0 hours |
| Excretion | renal |
| Identifiers | |
| |
JSmol) | |
| Melting point | 145 to 148 °C (293 to 298 °F) |
| Solubility in water | 3.92e-02 mg/mL [ALOGPS] mg/mL (20 °C) |
| |
| |
| (verify) | |
Etodolac is a nonsteroidal anti-inflammatory drug (NSAID).
It was patented in 1971 and approved for medical use in 1985.[2] It was approved in the U.S. in 1991.[3]
Medical uses

NSAIDs are used for the management of mild to moderate pain, fever, and inflammation. They work by reducing the levels of prostaglandins, which are chemicals that are responsible for pain and the fever and tenderness that occur with inflammation. Etodolac blocks the cyclooxygenase (abbrev. COX) enzymes which form prostanoids, resulting in lower concentrations of prostaglandins. As a consequence, inflammation, pain and fever are reduced.
Post-marketing studies demonstrated that etodolac inhibition of cyclooxygenase is somewhat
In the UK, Etodolac is licensed for the treatment of inflammation and pain caused by osteoarthritis and rheumatoid arthritis.[6]
Interactions
Etodolac should be avoided by patients with a history of

Pregnancy and nursing
Etodolac is generally avoided during
In October 2020, the U.S. Food and Drug Administration (FDA) required the drug label to be updated for all nonsteroidal anti-inflammatory medications to describe the risk of kidney problems in unborn babies that result in low amniotic fluid.[8][9] They recommend avoiding NSAIDs in pregnant women at 20 weeks or later in pregnancy.[8][9]
Brand names
Etodolac is manufactured by Almirall Limited under the trade name Lodine SR[10] and by Meda Pharmaceuticals under the name Eccoxolac.[11] Generic etodolac is also available.[12]
The drug is also sold under several other brand names, including:
- "Etogesic" (India)
- Etova (India)
- Dualgan (Portugal)
- Etodin (South Korea)
- Etofree (India)
- Etopan (Israel)
- Flancox®[13] (Brazil)
- Haipen (Japan)
- Lodine (France, Switzerland, United States)
- Proxym (S Etodolac) (India)
- Dolarit, Edolar, Etol (Turkey)
- Lonine (Taiwan)
- Etodine (Egypt)
- "Etodin Fort" (Bulgaria)
References
- FDA. Retrieved 22 Oct 2023.
- ISBN 9783527607495.
- ^ a b Ogbru O. "etodolac, Lodine (Discontinued): Drug Facts, Side Effects and Dosing". MedicineNet.
- PMID 10377455.
- PMID 17275129.
- ^ BNF 55 - Etodolac
- S2CID 31642564.
- ^ a b "FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S. Food and Drug Administration (FDA) (Press release). 15 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "NSAIDs may cause rare kidney problems in unborn babies". U.S. Food and Drug Administration. 21 July 2017. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Lodine SR". medicines.org.uk. Archived from the original on 2008-03-18. Retrieved 2008-08-07.
- ^ "Eccoxolac". medicines.org.uk. Archived from the original on 2012-12-24.
- ^ "Etodolac preparations". BNF 55.[permanent dead link]
- ^ "Flancox". apsen.com.br. 2 April 2020.
