Melanin
| Melanin | |
|---|---|
 One possible structure of Eumelanin | |
| Material type | Heterogeneous Biopolymer |
Melanin (
There are five basic types of melanin:
In the human skin, melanogenesis is initiated by exposure to
Melanin types
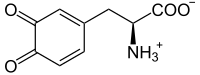
Eumelanin

Eumelanin has two forms linked to
In natural conditions, DHI and DHICA often co-polymerize, resulting in a range of eumelanin polymers. These polymers contribute to the variety of melanin components in human skin and hair, ranging from light yellow/red pheomelanin to light brown DHICA-enriched eumelanin and dark brown or black DHI-enriched eumelanin. These final polymers differ in solubility and color.[6]
Analysis of highly pigmented (Fitzpatrick type V and VI) skin finds that DHI-eumelanin comprises the largest portion, approximately 60–70%, followed by DHICA-eumelanin at 25–35%, and pheomelanin only 2–8%. Notably, while an enrichment of DHI-eumelanin occurs in during sun tanning, it is accompanied by a decrease in DHICA-eumelanin and pheomelanin.[6] A small amount of black eumelanin in the absence of other pigments causes grey hair. A small amount of eumelanin in the absence of other pigments causes blond hair.[7] Eumelanin is present in the skin and hair, etc.
Pheomelanin
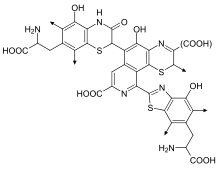
Pheomelanins (or phaeomelanins) impart a range of yellowish to reddish colors.[8] Pheomelanins are particularly concentrated in the lips, nipples, glans of the penis, and vagina.[9] When a small amount of eumelanin in hair (which would otherwise cause blond hair) is mixed with pheomelanin, the result is orange hair, which is typically called "red" or "ginger" hair. Pheomelanin is also present in the skin, and redheads consequently often have a more pinkish hue to their skin as well. Exposure of the skin to ultraviolet light increases pheomelanin content, as it does for eumelanin; but rather than absorbing light, pheomelanin within the hair and skin reflect yellow to red light, which may increase damage from UV radiation exposure.[10]
Pheomelanin production is highly dependent on
Neuromelanin
Neuromelanin (NM) is an insoluble polymer pigment produced in specific populations of
Other forms of melanins
Up until the 1960s, melanin was classified into eumelanin and pheomelanin. However in 1955 a melanin associated with nerve cells was discovered, neuromelanin, in 1972 a water-soluble form, pyomelanin. In 1976, allomelanin, the fifth form of the melanins was found in nature.[2]
Selenomelanin
It is possible to enrich melanin with
Trichochromes
Trichochromes (formerly called trichosiderins) are pigments produced from the same metabolic pathway as the eumelanins and pheomelanins, but unlike those molecules they have low molecular weight. They occur in some red human hair.[16]
Humans

In humans, melanin is the primary determinant of skin color. It is also found in hair, the pigmented tissue underlying the iris of the eye, and the stria vascularis of the inner ear. In the brain, tissues with melanin include the medulla and pigment-bearing neurons within areas of the brainstem, such as the locus coeruleus. It also occurs in the zona reticularis of the adrenal gland.[17]
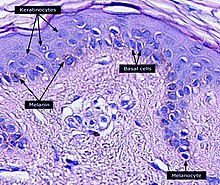
The melanin in the skin is produced by
Because melanin is an aggregate of smaller component molecules, there are many different types of melanin with different proportions and bonding patterns of these component molecules. Both pheomelanin and eumelanin are found in human skin and hair, but eumelanin is the most abundant melanin in humans, as well as the form most likely to be deficient in albinism.[19]
Other organisms
Melanins have very diverse roles and functions in various organisms. A form of melanin makes up the ink used by many
In
The darker feathers of birds owe their color to melanin and are less readily degraded by bacteria than unpigmented ones or those containing carotenoid pigments.[28] Feathers that contain melanin are also 39% more resistant to abrasion than those that do not because melanin granules help fill the space between the keratin strands that form feathers.[29][30] Pheomelanin synthesis in birds implies the consumption of cysteine, a semi‐essential amino acid that is necessary for the synthesis of the antioxidant glutathione (GSH) but that may be toxic if in excess in the diet. Indeed, many carnivorous birds, which have a high protein content in their diet, exhibit pheomelanin‐based coloration.[31]
Melanin is also important in
Melanin in
Arachnids are one of the few groups in which melanin has not been easily detected, though researchers found data suggesting spiders do in fact produce melanin.[38]
Some moth species, including the
Melanin protects
Plants

Melanin produced by plants are sometimes referred to as 'catechol melanins' as they can yield
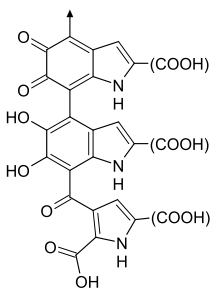
Interpretation as a single monomer
It is now understood that melanins do not have a single structure or stoichiometry. [citation needed] Nonetheless, chemical databases such as PubChem include structural and empirical formulae; typically 3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone. This can be thought of as a single monomer that accounts for the measured elemental composition and some properties of melanin, but is unlikely to be found in nature.[50] Solano[50] claims that this misleading trend stems from a report of an empirical formula in 1948,[51] but provides no other historical detail.
![3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone](http://upload.wikimedia.org/wikipedia/commons/thumb/2/20/Melanin.svg/240px-Melanin.svg.png)
| |
![3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone ball and stick model](http://upload.wikimedia.org/wikipedia/commons/thumb/7/7b/Melanin_ball_and_stick.png/240px-Melanin_ball_and_stick.png)
| |
| Names | |
|---|---|
| Preferred IUPAC name
3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone | |
| Identifiers | |
| ChemSpider | |
PubChem CID
|
|
| Properties | |
| C18H10N2O4 | |
| Molar mass | 318.288 g·mol−1 |
| Density | 1.6 to 1.8 g/cm3 |
| Melting point | < −20 °C (−4 °F; 253 K) |
| Boiling point | 450 to 550 °C (842 to 1,022 °F; 723 to 823 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Biosynthetic pathways
The first step of the biosynthetic pathway for both eumelanins and pheomelanins is catalysed by tyrosinase.[52]
- dopaquinone
Dopaquinone can combine with cysteine by two pathways to benzothiazines and pheomelanins
- Dopaquinone + cysteine → 5-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
- Dopaquinone + cysteine → 2-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
Also, dopaquinone can be converted to leucodopachrome and follow two more pathways to the eumelanins
- Dopaquinone → leucodopachrome → dopachrome → 5,6-dihydroxyindole-2-carboxylic acid → quinone → eumelanin
- Dopaquinone → leucodopachrome → dopachrome → 5,6-dihydroxyindole → quinone → eumelanin
Detailed metabolic pathways can be found in the KEGG database (see External links).
-
L-tyrosine
-
L-DOPA
-
L-dopaquinone
-
L-leucodopachrome
-
L-dopachrome
Microscopic appearance
Melanin is brown, non-refractile, and finely granular with individual granules having a diameter of less than 800 nanometers. This differentiates melanin from common blood breakdown pigments, which are larger, chunky, and refractile, and range in color from green to yellow or red-brown. In heavily pigmented lesions, dense aggregates of melanin can obscure histologic detail. A dilute solution of potassium permanganate is an effective melanin bleach.[53]
Genetic disorders and disease states
There are approximately nine types of
Ocular albinism affects not only eye pigmentation but visual acuity, as well. People with albinism typically test poorly, within the 20/60 to 20/400 range. In addition, two forms of albinism, with approximately 1 in 2,700 most prevalent among people of Puerto Rican origin, are associated with mortality beyond melanoma-related deaths.
The connection between albinism and
though why this is, is not fully understood.In Parkinson's disease, a disorder that affects neuromotor functioning, there is decreased neuromelanin in the substantia nigra and locus coeruleus as a consequence of specific dropping out of dopaminergic and noradrenergic pigmented neurons. This results in diminished dopamine and norepinephrine synthesis. While no correlation between race and the level of neuromelanin in the substantia nigra has been reported, the significantly lower incidence of Parkinson's in blacks than in whites has "prompt[ed] some to suggest that cutaneous melanin might somehow serve to protect the neuromelanin in substantia nigra from external toxins."[60]
In addition to melanin deficiency, the molecular weight of the melanin polymer may be decreased by various factors such as oxidative stress, exposure to light, perturbation in its association with melanosomal
Higher eumelanin levels also can be a disadvantage, however, beyond a higher disposition toward vitamin D deficiency. Dark skin is a complicating factor in the laser removal of port-wine stains. Effective in treating white skin, in general, lasers are less successful in removing port-wine stains in people of Asian or African descent. Higher concentrations of melanin in darker-skinned individuals simply diffuse and absorb the laser radiation, inhibiting light absorption by the targeted tissue. In a similar manner, melanin can complicate laser treatment of other dermatological conditions in people with darker skin.
Nicotine has an affinity for melanin-containing tissues because of its precursor function in melanin synthesis or its irreversible binding of melanin. This has been suggested to underlie the increased nicotine dependence and lower smoking cessation rates in darker pigmented individuals.[63]
Human adaptation
Physiology
Melanocytes insert granules of melanin into specialized cellular
Not all the effects of pigmentation are advantageous. Pigmentation increases the heat load in hot climates, and dark-skinned people absorb 30% more heat from sunlight than do very light-skinned people, although this factor may be offset by more profuse sweating. In cold climates dark skin entails more heat loss by radiation. Pigmentation also hinders synthesis of vitamin D. Since pigmentation appears to be not entirely advantageous to life in the tropics, other hypotheses about its biological significance have been advanced, for example a secondary phenomenon induced by adaptation to parasites and tropical diseases.[65]
Evolutionary origins
Effects
As with peoples having migrated northward, those with light skin migrating toward the equator acclimatize to the much stronger solar radiation. Nature selects for less melanin when ultraviolet radiation is weak. Most people's skin darkens when exposed to UV light, giving them more protection when it is needed. This is the physiological purpose of
Melanin in the eyes, in the
Recent research suggests that melanin may serve a protective role other than photoprotection.[72] Melanin is able to effectively chelate metal ions through its carboxylate and phenolic hydroxyl groups, in many cases much more efficiently than the powerful chelating ligand ethylenediaminetetraacetate (EDTA). Thus, it may serve to sequester potentially toxic metal ions, protecting the rest of the cell. This hypothesis is supported by the fact that the loss of neuromelanin observed in Parkinson's disease is accompanied by an increase in iron levels in the brain.
Physical properties and technological applications
Evidence exists in support of a highly cross-linked
Role in melanoma metastasis
The research done by Sarna's team proved that heavily pigmented melanoma cells have Young's modulus about 4.93 kPa, when in non-pigmented ones it was only 0.98 kPa.[77] In another experiment they found that elasticity of melanoma cells is important for its metastasis and growth: non-pigmented tumors were bigger than pigmented and it was much easier for them to spread. They also showed that there are both pigmented and non-pigmented cells in melanoma tumors, so that they can both be drug-resistant and metastatic.[77]
See also
- Albino
- Albinism in biology
- Griscelli syndrome, a syndrome characterised by hypopigmentation
- Human skin color
- Melanin theory
- Melanism
- Melanogenesis, melanin production
- Risks and benefits of sun exposure
- Skin whitening
- Vitamin D
References
- S2CID 3832753.
- ^ S2CID 231872855.
- PMID 28469642.
- S2CID 222101966.
- PMID 18435612.
- ^ PMID 11601655.
- S2CID 5121042.
- S2CID 21168932.
- ^ "pheomelanin". MetaCyc Metabolic Pathway Database. 2010. [full citation needed]
- PMID 2071942.
- PMID 21341762.
- S2CID 503902.
- S2CID 23096297.
- S2CID 220413025.
- ^ Mark Heiden (8 July 2020). "New biomaterial could shield against harmful radiation". Northwestern University. Retrieved 29 January 2023.
- PMID 22896083.
- .
- PMID 24278043.
- ^ "oculocutaneous albinism". Genetics Home Reference. Retrieved 25 September 2017.
- PMID 11871612.
- S2CID 10614298.
- .
- PMID 17520016.
- PMID 26811288.
- S2CID 8877527.
- PMID 25240282.
- .
- .
- JSTOR 1369048.
- PMID 27070583.
- S2CID 202018215.
- PMID 819593.
- ISBN 9780199681266.
- NCBI.[permanent dead link]
- PMID 7588057.
- ISBN 9783642809101.
- ISBN 978-163-081-797-8
- PMID 26449977.
- PMID 23363631.
- ^ a b Mosse, Irma B.; Dubovic, Boris V.; Plotnikova, Svetlana I.; Kostrova, Ludmila N.; Molophei, Vadim; Subbot, Svetlana T.; Maksimenya, Inna P. (20–25 May 2001). Obelic, B.; Ranogajev-Komor, M.; Miljanic, S.; Krajcar Bronic, I. (eds.). Melanin is Effective Radioprotector against Chronic Irradiation and Low Radiation Doses. IRPA Regional Congress on Radiation Protection in Central Europe: Radiation Protection and Health. INIS. Dubrovnik (Croatia): Croatian Radiation Protection Association. p. 35 (of 268).
- ^ S2CID 8570835.
- ^ PMID 24925363.
- ^ PMID 32274309.
- ^ PMID 22683898.
- ^ S2CID 59398089.
- ^ S2CID 245823003.
- ^ OCLC 711778164.
- PMID 27110763.
- S2CID 8280251.
- ^ .
- PMID 18920770.
- PMID 24895537.
- ^ "Melanin". pubchem.ncbi.nlm.nih.gov. Retrieved 25 September 2017.
- ^ "Oculocutaneous Albinism". Archived from the original on 23 December 2008.
- ^ a b Peracha, Mohammed O.; Cosgrove, Frances M.; Garcia-Valenzuela, Enrique; Eliott, Dean (5 November 2015). Roy, Sr, Hampton; Talavera, Francisco; Rowsey, J. James (eds.). "Ocular Manifestations of Albinism: Background, Pathophysiology, Epidemiology". Medscape. Additional contributions from Kilbourn Gordon, III. WebMD. Archived from the original on 28 March 2017. Retrieved 8 September 2022 – via eMedicine.
- ^ "Causes of Variability". Archived from the original on 23 September 2006. Retrieved 18 September 2006.
- ^ EntrezGene 300700
- ^ EntrezGene 606933
- PMID 7521050.
- ^ "Lewy Body Disease". Archived from the original on 21 July 2009.
- PMID 11434561.
- S2CID 30344869.
- S2CID 3070838.
- ^ "Human Skin Color Variation". The Smithsonian Institution's Human Origins Program. 20 June 2012. Retrieved 24 August 2019.
- ISBN 978-1-4051-6169-5
- ISSN 0362-4331. Retrieved 24 August 2019.
- PMID 19407144.
- ^ "A Single Migration From Africa Populated the World, Studies Find". The New York Times. 22 September 2016.
- PMID 10733465.
- S2CID 2245002.
- PMID 20445093.
- PMID 15140071.
- PMID 7556145.
- S2CID 27499198.
- S2CID 26079375.
- PMID 16030499.
- ^ PMID 31243305.
External links
- "Absorption spectrum of melanin". Department of Computer Science and Technology.
- "Tyrosine metabolism—Reference pathway". Kyoto Encyclopedia of Genes and Genomes. Archived from the original on 3 August 2020. Retrieved 24 August 2019.
- "Melanogenesis—Reference pathway". Kyoto Encyclopedia of Genes and Genomes. Archived from the original on 4 August 2020. Retrieved 24 August 2019.