Europium(III) chloride

| |
| |
| Names | |
|---|---|
| IUPAC names
Europium(III) chloride
Europium trichloride | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.030.025 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| EuCl3 | |
| Molar mass | 258.323 g/mol 366.41 g/mol (hexahydrate) |
| Melting point | 632 °C (1,170 °F; 905 K) decomposes |
| Solubility in other solvents | Soluble |
| Structure | |
UCl3 type), hP8
| |
| P63/m, No. 176 | |
| Tricapped trigonal prismatic (nine-coordinate) | |
| Related compounds | |
Other anions
|
Europium(III) oxide |
Other cations
|
Samarium(III) chloride Gadolinium(III) chloride |
Related compounds
|
Europium dichloride
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Europium(III) chloride is an
hygroscopic it rapidly absorbs water to form a white crystalline hexahydrate
, EuCl3·6H2O, which is colourless. The compound is used in research.
Preparation
Treating
oxychloride
.
Anhydrous EuCl3 is often prepared by the "ammonium chloride route," starting from either Eu2O3[1][2] or hydrated europium chloride (EuCl3·6H2O) by heating carefully to 230 °C.[3] These methods produce (NH4)2[EuCl5]:
- 10 NH4Cl + Eu2O3 → 2 (NH4)2[EuCl5] + 6 NH3 + 3 H2O
- EuCl3·6H2O + 2 NH4Cl → (NH4)2[EuCl5] + 6 H2O
The pentachloride decomposes thermally according to the following equation:
- (NH4)2[EuCl5] → 2 NH4Cl + EuCl3
The thermolysis reaction proceeds via the intermediary of (NH4)[Eu2Cl7].
Reactions
Europium(III) chloride is a precursor to other europium compounds. It can be converted to the corresponding
THF
and requires a period at reflux.
- EuCl3 + 3 LiN(SiMe3)2 → Eu(N(SiMe3)2)3 + 3 LiCl
Eu(N(SiMe3)2)3 is a starting material for the more complicated coordination complexes.
Reduction with
organometallic compounds of europium(II), such as bis(pentamethylcyclopentadienyl)europium(II) complexes.[5][6] Europium(III) chloride can be used as a starting point for the preparation of other europium salts
.
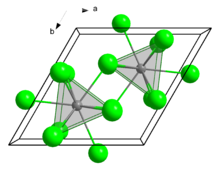
Structure
In the solid state, it crystallises in the UCl3 motif. The Eu centres are nine-coordinate.[7]
Bibliography
- Weast, R. C., ed. (1972). Handbook of Chemistry and Physics (53rd ed.). Cleveland, OH: Chemical Rubber Co.
References
- ^
Meyer, G. (1989). The Ammonium Chloride Route to Anhydrous Rare Earth Chlorides-The Example of YCl3. Inorganic Syntheses. Vol. 25. pp. 146–150. ISBN 978-0-470-13256-2.
- ISBN 3-13-103021-6.
- .
- .
- .
- .
- ISBN 978-0-08-037941-8.

