Farnesyl-diphosphate farnesyltransferase
| Squalene synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
ExPASy NiceZyme view | | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| farnesyl-diphosphate farnesyltransferase 1 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | FDFT1 | ||||||
Chr. 8 p23.1-p22 | |||||||
| |||||||
Squalene synthase (SQS) or farnesyl-diphosphate:farnesyl-diphosphate farnesyl transferase is an enzyme localized to the membrane of the endoplasmic reticulum. SQS participates in the isoprenoid biosynthetic pathway, catalyzing a two-step reaction in which two identical molecules of farnesyl pyrophosphate (FPP) are converted into squalene, with the consumption of NADPH.[2] Catalysis by SQS is the first committed step in sterol synthesis, since the squalene produced is converted exclusively into various sterols, such as cholesterol, via a complex, multi-step pathway. SQS belongs to squalene/phytoene synthase family of proteins.
Diversity
Squalene synthase has been characterized in animals, plants, and yeast.[3] In terms of structure and mechanics, squalene synthase closely resembles phytoene synthase (PHS), another prenyltransferase. PHS serves a similar role to SQS in plants and bacteria, catalyzing the synthesis of phytoene, a precursor of carotenoid compounds.[4]
Structure
Squalene synthase (SQS) is localized exclusively to the

Mechanism

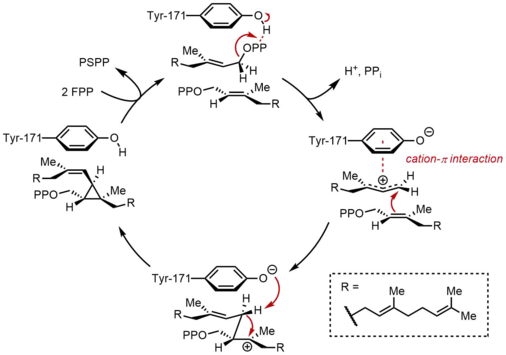
Squalene synthase (SQS) catalyzes the reductive dimerization of farnesyl pyrophosphate (FPP), in which two identical molecules of FPP are converted into one molecule of squalene. The reaction occurs in two steps, proceeding through the intermediate presqualene pyrophosphate (PSPP). FPP is a
FPP condensation
In the first half-reaction, two identical molecules of farnesyl pyrophosphate (FPP) are bound to squalene synthase (SQS) in a sequential manner. The FPP molecules bind to distinct regions of the enzyme, and with different binding affinities.
PSPP rearrangement and reduction
In the second half-reaction of SQS, presqualene pyrophosphate (PSPP) moves to a second reaction site within SQS. Keeping PSPP in the central channel of SQS is thought to protect the reactive intermediate from reacting with water.[5] From PSPP, squalene is formed by a series of carbocation rearrangements.[12][13] The process begins with ionization of pyrophosphate, giving a cyclopropylcarbinyl cation. The cation rearranges by a 1,2-migration of a cyclopropane C–C bond to the carbocation, forming the bond shown in blue to give a cyclobutyl carbocation. Subsequently, a second 1,2-migration occurs to form another cyclopropylcarbinyl cation, with the cation resting on a tertiary carbon. This resulting carbocation is then ring-opened by a hydride delivered by NADPH, giving squalene, which is then released by SQS into the membrane of the endoplasmic reticulum.[2]
While cyclopropylcarbinyl-cyclopropylcarbinyl rearrangements can proceed through discrete cyclobutyl cation intermediates, the supposed cyclobutyl cation could not be trapped in model studies. Thus, the cyclobutyl cation may actually be a transition state between the two cyclopropylcarbinyl cations, rather than a discrete intermediate. The stereochemistry of the intermediates and the olefin geometry in the final product is dictated by the suprafacial nature of the 1,2-shifts and stereoelectronic requirements. While other mechanisms have been proposed, the mechanism shown above is supported by isolation of rillingol, which is the alcohol formed from trapping the second cyclopropylcarbinyl cation with water.
Regulation

FPP is an important metabolic intermediate in the mevalonate pathway that represents a major branch point in terpenoid pathways.[2][14] FPP is used to form several important classes of compounds in addition to sterols (via squalene), including ubiquinone[15] and dolichols.[16] SQS catalyzes the first committed step in sterol biosynthesis from FPP, and is therefore important for controlling the flux towards sterol vs. non-sterol products. The activity of SQS is intimately related to the activity of HMG-CoA reductase, which catalyzes the rate-limiting step of the mevalonate pathway. High levels of LDL-derived cholesterol inhibit HMG-CoA reductase activity significantly, since mevalonate is no longer needed for sterol production. However, residual HMG-CoA reductase activity is observed even with very high LDL levels, such that FPP can be made for forming non-sterol products essential for cell growth.[17] To prevent this residual FPP from being used for sterol synthesis when sterols are abundant, SQS activity declines significantly when LDL levels are high.[18] This suppression of SQS activity is better thought of as a flux control mechanism, rather than a way to regulate cholesterol levels. This is since HMG-CoA reductase is the more significant control factor for regulating cholesterol synthesis (its activity is 98% inhibited when LDL levels are high).[17]
Regulation by sterols
SQS regulation occurs primarily at the level of SQS
Aside from SREBPs, accessory transcription factors are needed for maximal activation of the SQS promoter. Promoter studies using
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
Biological Function
Squalene synthase (SQS) is an enzyme participating in the isoprenoid biosynthetic pathway. SQS synthase catalyzes the branching point between sterol and nonsterol biosynthesis, and commits farnesyl pyrophosphate (FPP) exclusively to production of sterols.[2] An important sterol produced by this pathway is cholesterol, which is used in cell membranes and for the synthesis of hormones.[22] SQS competes with several other enzymes for use of FPP, since it is a precursor for a variety of terpenoids. Decreases in SQS activity limit flux of FPP to the sterol pathway, and increase the production of nonsterol products. Important nonsterol products include ubiquinone, dolichols, heme A, and farnesylated proteins [23]
Development of squalene synthase knockout mice has demonstrated that loss of squalene synthase is lethal, and that the enzyme is essential for development of the central nervous system.[24]
Disease Relevance
Squalene synthase is a target for the regulation of cholesterol levels. Increased
Squalene synthase inhibitors
Squalene synthase inhibitors have been shown to decrease cholesterol synthesis, as well as to decrease plasma triglyceride levels.[22][27] SQS inhibitors may provide an alternative to HMG-CoA reductase inhibitors (statins), which have problematic side effects for some patients.[28] Squalene synthase inhibitors that have been investigated for use in the prevention of cardiovascular disease include lapaquistat (TAK-475), zaragozic acid, and RPR 107393.[29][30] Despite reaching phase II clinical trials, lapaquistat was discontinued by 2008.[31][32]
Squalene synthase homolog inhibition in
References
- PMID 21353782.
- ^ PMID 11111077.
- PMID 7892265.
- ^ PMID 11008488.
- ^ PMID 10896663.
- PMID 2068081.
- ^ PMID 9575210.
- .
- PMID 21098670.
- ^ PMID 4348553.
- PMID 8157649.
- PMID 12137537.
- .
- PMID 6995544.
- PMID 5340877.
- PMID 4319540.
- ^ PMID 219777.
- PMID 228272.
- PMID 7665618.
- PMID 9092581.
- PMID 9575211.
- ^ PMID 21864285.
- S2CID 23878922.
- ^ PMID 16741291.
- S2CID 28176904.
- S2CID 205406994.
- PMID 11730728.
- S2CID 33130333.
- S2CID 45715717.
- PMID 9152381.
- ^ Gibbs, Edwina (29 October 2007). "UPDATE 2-US FDA tells Takeda to stop some TAK-475 trials". Reuters. Retrieved 5 March 2013.
- ^ "Discontinuation of Development of TAK-475, A Compound for Treatment of Hypercholesterolemia". Takeda Pharmaceutical Company Limited. 28 March 2008. Retrieved 5 March 2013.
- PMID 18276850.
External links
- Farnesyl-Diphosphate+Farnesyltransferase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)