Fatty acid metabolism
Fatty acid metabolism consists of various
In catabolism, fatty acids are metabolized to produce energy, mainly in the form of adenosine triphosphate (ATP). When compared to other macronutrient classes (carbohydrates and protein), fatty acids yield the most ATP on an energy per gram basis, when they are completely oxidized to CO2 and water by beta oxidation and the citric acid cycle.[2] Fatty acids (mainly in the form of triglycerides) are therefore the foremost storage form of fuel in most animals, and to a lesser extent in plants.
In anabolism, intact fatty acids are important precursors to triglycerides, phospholipids, second messengers, hormones and
Fatty acid catabolism
Fatty acids are stored as
- epinephrine and glucagon levels in the blood (or norepinephrine secreted by sympathetic nerves in adipose tissue), caused by declining blood glucose levels after meals, which simultaneously lowers the insulin level in the blood.[1]
- Once freed from glycerol, the free fatty acids enter the blood, which transports them, attached to plasma albumin, throughout the body.[4]
- Long-chain free fatty acids enter metabolizing cells (i.e. most living cells in the body except interstitial fluidsthat bathe these cells.
- Once inside the cell, long-chain-fatty-acid—CoA ligase catalyzes the reaction between a fatty acid molecule with ATP (which is broken down to AMP and inorganic pyrophosphate) to give a fatty acyl-adenylate, which then reacts with free coenzyme A to give a fatty acyl-CoA molecule.
- In order for the acyl-CoA to enter the mitochondrion the carnitine shuttle is used:[10][11][12]
- Acyl-CoA is transferred to the hydroxyl group of carnitine by outer and inner mitochondrial membranes.
- Acyl-carnitine is shuttled inside by a carnitine-acylcarnitine translocase, as a carnitine is shuttled outside.
- Acyl-carnitine is converted back to acyl-CoA by carnitine palmitoyltransferase II, located on the interior face of the inner mitochondrial membrane. The liberated carnitine is shuttled back to the cytosol, as an acyl-CoA is shuttled into the mitochondrial matrix.
- acetyl CoA, which condense with oxaloacetate to form citrate at the "beginning" of the citric acid cycle.[2] It is convenient to think of this reaction as marking the "starting point" of the cycle, as this is when fuel - acetyl-CoA - is added to the cycle, which will be dissipated as CO2 and H2O with the release of a substantial quantity of energy captured in the form of ATP, during the course of each turn of the cycle and subsequent oxidative phosphorylation.
- Briefly, the steps in beta oxidation are as follows:[2]
- Dehydrogenation by acyl-CoA dehydrogenase, yielding 1 FADH2
- Hydration by enoyl-CoA hydratase
- Dehydrogenation by 3-hydroxyacyl-CoA dehydrogenase, yielding 1 NADH + H+
- Cleavage by thiolase, yielding 1 acetyl-CoA and a fatty acid that has now been shortened by 2 carbons (forming a new, shortened acyl-CoA)
- This beta oxidation reaction is repeated until the fatty acid has been completely reduced to acetyl-CoA or, in, the case of fatty acids with odd numbers of carbon atoms, acetyl-CoA and 1 molecule of propionyl-CoA per molecule of fatty acid. Each beta oxidative cut of the acyl-CoA molecule eventually yields 5 ATP molecules in oxidative phosphorylation.[13][14]
- The acetyl-CoA produced by beta oxidation enters the citric acid cycle in the mitochondrion by combining with oxaloacetate to form citrate. Coupled to oxidative phosphorylation this results in the complete combustion of the acetyl-CoA to CO2 and water. The energy released in this process is captured in the form of 1 GTP and 11 ATP molecules per acetyl-CoA molecule oxidized.[2][10] This is the fate of acetyl-CoA wherever beta oxidation of fatty acids occurs, except under certain circumstances in the liver.
- The propionyl-CoA is later converted into succinyl-CoA through biotin-dependant propionyl-CoA carboxylase (PCC) and Vitamin B12-dependant methylmalonyl-CoA mutase (MCM), sequentially.[15][16] Succinyl-CoA is first converted to malate, and then to pyruvate where it is then transported to the matrix to enter the citric acid cycle.
In the liver oxaloacetate can be wholly or partially diverted into the
- The glycerol released by lipase action is triose phosphate isomerase converts this compound to glyceraldehyde 3-phosphate, which is oxidized via glycolysis, or converted to glucose via gluconeogenesis.
Fatty acids as an energy source
Fatty acids, stored as triglycerides in an organism, are a concentrated
Hibernating animals provide a good example for utilization of fat reserves as fuel. For example, bears hibernate for about 7 months, and during this entire period, the energy is derived from degradation of fat stores. Migrating birds similarly build up large fat reserves before embarking on their intercontinental journeys.[17]
The fat stores of young adult humans average between about 10–20 kg, but vary greatly depending on gender and individual disposition.[18] By contrast, the human body stores only about 400 g of glycogen, of which 300 g is locked inside the skeletal muscles and is unavailable to the body as a whole. The 100 g or so of glycogen stored in the liver is depleted within one day of starvation.[10] Thereafter the glucose that is released into the blood by the liver for general use by the body tissues has to be synthesized from the glucogenic amino acids and a few other gluconeogenic substrates, which do not include fatty acids.[1] Nonetheless, lipolysis releases glycerol which can enter the pathway of gluconeogenesis.
Carbohydrate synthesis from glycerol and fatty acids
Fatty acids are broken down to acetyl-CoA by means of beta oxidation inside the mitochondria, whereas
However, acetyl-CoA can be converted to acetoacetate, which can decarboxylate to
The glycerol released into the blood during the
Other functions and uses of fatty acids
Intracellular signaling

Fatty acids are an integral part of the phospholipids that make up the bulk of the
Eicosanoid paracrine hormones

One product of fatty acid metabolism are the
The prostaglandins are synthesized in the cell membrane by the cleavage of arachidonate from the phospholipids that make up the membrane. This is catalyzed either by
If arachidonate is acted upon by a
Prostaglandins have two derivatives:
Dietary sources of fatty acids, their digestion, absorption, transport in the blood and storage



A significant proportion of the fatty acids in the body are obtained from the diet, in the form of triglycerides of either animal or plant origin. The fatty acids in the fats obtained from land animals tend to be saturated, whereas the fatty acids in the triglycerides of fish and plants are often polyunsaturated and therefore present as oils.
These
The contents of these micelles (but not the bile salts) enter the

The chylomicrons circulate throughout the body, giving the blood plasma a milky or creamy appearance after a fatty meal.[citation needed] Lipoprotein lipase on the endothelial surfaces of the capillaries, especially in adipose tissue, but to a lesser extent also in other tissues, partially digests the chylomicrons into free fatty acids, glycerol and chylomicron remnants. The fatty acids are absorbed by the adipocytes[citation needed], but the glycerol and chylomicron remnants remain in the blood plasma, ultimately to be removed from the circulation by the liver. The free fatty acids released by the digestion of the chylomicrons are absorbed by the adipocytes[citation needed], where they are resynthesized into triglycerides using glycerol derived from glucose in the glycolytic pathway[citation needed]. These triglycerides are stored, until needed for the fuel requirements of other tissues, in the fat droplet of the adipocyte.
The
Adipose tissue and lactating mammary glands also take up glucose from the blood for conversion into triglycerides. This occurs in the same way as in the liver, except that these tissues do not release the triglycerides thus produced as VLDL into the blood. Adipose tissue cells store the triglycerides in their fat droplets, ultimately to release them again as free fatty acids and glycerol into the blood (as described above), when the plasma concentration of insulin is low, and that of glucagon and/or epinephrine is high.[33] Mammary glands discharge the fat (as cream fat droplets) into the milk that they produce under the influence of the anterior pituitary hormone prolactin.
All cells in the body need to manufacture and maintain their membranes and the membranes of their organelles. Whether they rely entirely on free fatty acids absorbed from the blood, or are able to synthesize their own fatty acids from blood glucose, is not known. The cells of the
Fatty acid synthesis

Much like
The diagrams presented show how fatty acids are synthesized in microorganisms and list the enzymes found in
In animals as well as some fungi such as yeast, these same reactions occur on fatty acid synthase I (FASI), a large dimeric protein that has all of the enzymatic activities required to create a fatty acid. FASI is less efficient than FASII; however, it allows for the formation of more molecules, including "medium-chain" fatty acids via early chain termination.[37] Enzymes, acyltransferases and transacylases, incorporate fatty acids in phospholipids, triacylglycerols, etc. by transferring fatty acids between an acyl acceptor and donor. They also have the task of synthesizing bioactive lipids as well as their precursor molecules.[38]
Once a 16:0 carbon fatty acid has been formed, it can undergo a number of modifications, resulting in desaturation and/or elongation. Elongation, starting with stearate (18:0), is performed mainly in the endoplasmic reticulum by several membrane-bound enzymes. The enzymatic steps involved in the elongation process are principally the same as those carried out by fatty acid synthesis, but the four principal successive steps of the elongation are performed by individual proteins, which may be physically associated.[39][40]
| Step | Enzyme | Reaction | Description |
|---|---|---|---|
| (a) | Acetyl CoA:ACP transacylase | Activates acetyl CoA for reaction with malonyl-ACP | |
| (b) | Malonyl CoA:ACP transacylase | Activates malonyl CoA for reaction with acetyl-ACP | |
| (c) | 3-ketoacyl-ACP synthase
|
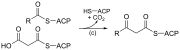 |
Reacts ACP-bound acyl chain with chain-extending malonyl-ACP |
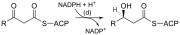
| (d) | 3-ketoacyl-ACP reductase | Reduces the carbon 3 ketone to a hydroxyl group | |
| (e) | 3-Hydroxyacyl ACP dehydrase | Eliminates water | |
| (f) | Enoyl-ACP reductase | Reduces the C2-C3 double bond. |
Abbreviations: ACP – Acyl carrier protein, CoA – Coenzyme A, NADP – Nicotinamide adenine dinucleotide phosphate.
Note that during fatty synthesis the reducing agent is
Glycolytic end products are used in the conversion of carbohydrates into fatty acids
In humans, fatty acids are formed from carbohydrates predominantly in the liver and
Regulation of fatty acid synthesis
Acetyl-CoA is formed into
High plasma levels of
Disorders
Disorders of fatty acid metabolism can be described in terms of, for example, hypertriglyceridemia (too high level of triglycerides), or other types of hyperlipidemia. These may be familial or acquired.
Familial types of disorders of fatty acid metabolism are generally classified as
Moreover, cancer cells can display irregular fatty acid metabolism with regard to both
References
- ^ ISBN 0-7167-2009-4.
- ^ a b c d Oxidation of fatty acids
- S2CID 35349649.
- ^ Mobilization and cellular uptake of stored fats (triacylglycerols) (with animation)
- S2CID 2769738.
- PMID 23506886.
- PMID 12843297.
- PMID 23072752.
- ISBN 0-7167-2009-4.
- ^ ISBN 0-7167-2009-4.
- ^ Activation and transportation of fatty acids to the mitochondria via the carnitine shuttle (with animation)
- S2CID 28692799.
- ^ Oxidation of odd carbon chain length fatty acids
- ^ Oxidation of unsaturated fatty acids
- PMID 29033250.
- PMID 2647392.
- ISBN 0-7167-2009-4.
- S2CID 39812342.
- PMID 4553872.
- ^ a b c Glew, Robert H. "You Can Get There From Here: Acetone, Anionic Ketones and Even-Carbon Fatty Acids can Provide Substrates for Gluconeogenesis". Nigerian Journal of Physiological Science. 25 (1). Invited review: 2–4. Archived from the original on 26 September 2013. Retrieved 7 August 2016.
- ^ PMID 9921151.
- S2CID 37769342.
- PMID 1537826.
- ISBN 0-7167-2009-4.
- ^ ISBN 0-7167-2009-4.
- ISBN 978-0-87893-617-5.
- ^ a b Digestion of fats (triacylglycerols)
- PMID 14097367.
- ISBN 0-7167-2009-4.
- ISBN 978-1133104056.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ISBN 0443-041776.
- ISBN 0-7167-2009-4.
- PMID 2661229.
- ^ ISBN 0-7167-2009-4.
- ^ a b Dijkstra, Albert J., R. J. Hamilton, and Wolf Hamm. "Fatty Acid Biosynthesis." Trans Fatty Acids. Oxford: Blackwell Pub., 2008. 12. Print.
- ^ "MetaCyc pathway: superpathway of fatty acids biosynthesis". MetaCyc Metabolic Pathway Database. BioCyc. (E. coli).
- ^ a b Christie, William W. (20 April 2011). "Fatty Acids: Straight-chain Saturated, Structure, Occurrence and Biosynthesis". In American Oil Chemists' Society (ed.). AOCS Lipid Library. Archived from the original on 2011-07-21. Retrieved 2011-05-02.
- PMID 24125941.
- ^ "MetaCyc pathway: stearate biosynthesis I (animals)". MetaCyc Metabolic Pathway Database. BioCyc.
- ^ "MetaCyc pathway: very long chain fatty acid biosynthesis II". MetaCyc Metabolic Pathway Database. BioCyc.
- ^ PMID 17344645. Retrieved 2010-08-30.
this process is outlined graphically in page 73
- ^ ISBN 978-0-471-21495-3.
- ^ Diwan, Joyce J. "Fatty Acid Synthesis." Rensselaer Polytechnic Institute (RPI) :: Architecture, Business, Engineering, IT, Humanities, Science. Web. 30 Apr. 2011. <"Fatty Acid Synthesis". Archived from the original on 2011-06-07. Retrieved 2011-05-02.>.
- S2CID 85532042.
- S2CID 232300877.