Iron(III) oxide
 | |

| |
| |
| Names | |
|---|---|
| IUPAC name
Iron(III) oxide
| |
| Other names | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.013.790 |
| EC Number |
|
| E number | E172(ii) (colours) |
| 11092 | |
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| Fe2O3 | |
| Molar mass | 159.687 g·mol−1 |
| Appearance | Red solid |
| Odor | Odorless |
| Density | 5.25 g/cm3[1] |
| Melting point | 1,539 °C (2,802 °F; 1,812 K)[1] decomposes 105 °C (221 °F; 378 K) β-dihydrate, decomposes 150 °C (302 °F; 423 K) β-monohydrate, decomposes 50 °C (122 °F; 323 K) α-dihydrate, decomposes 92 °C (198 °F; 365 K) α-monohydrate, decomposes[2] |
| Insoluble | |
| Solubility | Soluble in diluted acids,[1] barely soluble in sugar solution[2] Trihydrate slightly soluble in aq. tartaric acid, citric acid, CH3COOH[2] |
| +3586.0x10−6 cm3/mol | |
Refractive index (nD)
|
n1 = 2.91, n2 = 3.19 (α, hematite)[3] |
| Structure | |
Rhombohedral, hR30 (α-form)[4]
Cubic bixbyite, cI80 (β-form) Cubic spinel (γ-form) Orthorhombic (ε-form)[5] | |
| R3c, No. 161 (α-form)[4] Ia3, No. 206 (β-form) Pna21, No. 33 (ε-form)[5] | |
| 3m (α-form)[4] 2/m 3 (β-form) mm2 (ε-form)[5] | |
| Octahedral (Fe3+, α-form, β-form)[4] | |
| Thermochemistry[6] | |
Heat capacity (C)
|
103.9 J/mol·K[6] |
Std molar
entropy (S⦵298) |
87.4 J/mol·K[6] |
Std enthalpy of (ΔfH⦵298)formation |
−824.2 kJ/mol[6] |
Gibbs free energy (ΔfG⦵)
|
−742.2 kJ/mol[6] |
| Hazards | |
| GHS labelling: | |
 [7] [7]
| |
| Warning | |
| H315, H319, H335[7] | |
| P261, P305+P351+P338[7] | |
| NFPA 704 (fire diamond) | |
Threshold limit value (TLV)
|
5 mg/m3[1] (TWA) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
10 g/kg (rats, oral)[9] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 10 mg/m3[8] |
REL (Recommended)
|
TWA 5 mg/m3[8] |
IDLH (Immediate danger) |
2500 mg/m3[8] |
| Related compounds | |
Other anions
|
Iron(III) fluoride |
Other cations
|
Manganese(III) oxide Cobalt(III) oxide |
Related
iron oxides |
Iron(II) oxide Iron(II,III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Iron(III) oxide or ferric oxide is the inorganic compound with the formula Fe2O3. It is one of the three main oxides of iron, the other two being iron(II) oxide (FeO), which is rare; and iron(II,III) oxide (Fe3O4), which also occurs naturally as the mineral magnetite. As the mineral known as hematite, Fe2O3 is the main source of iron for the steel industry. Fe2O3 is readily attacked by acids. Iron(III) oxide is often called rust, since rust shares several properties and has a similar composition; however, in chemistry, rust is considered an ill-defined material, described as hydrous ferric oxide.[10]
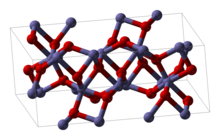
Structure
Fe2O3 can be obtained in various
Alpha phase
α-Fe2O3 has the
Gamma phase
Other solid phases
Several other phases have been identified or claimed. The β-phase is cubic body-centered (space group Ia3), metastable, and at temperatures above 500 °C (930 °F) converts to alpha phase. It can be prepared by reduction of hematite by carbon,[clarification needed] pyrolysis of iron(III) chloride solution, or thermal decomposition of iron(III) sulfate.[13]
The epsilon (ε) phase is rhombic, and shows properties intermediate between alpha and gamma, and may have useful magnetic properties applicable for purposes such as high density
Additionally, at high pressure an
Liquid phase
Molten Fe2O3 is expected to have a coordination number of close to 5 oxygen atoms about each iron atom, based on measurements of slightly oxygen deficient supercooled liquid iron oxide droplets, where supercooling circumvents the need for the high oxygen pressures required above the melting point to maintain stoichiometry.[16]
Hydrated iron(III) oxides
Several hydrates of Iron(III) oxide exist. When alkali is added to solutions of soluble Fe(III) salts, a red-brown gelatinous precipitate forms. This is not Fe(OH)3, but Fe2O3·H2O (also written as Fe(O)OH). Several forms of the hydrated oxide of Fe(III) exist as well. The red lepidocrocite (γ-Fe(O)OH) occurs on the outside of rusticles, and the orange goethite (α-Fe(O)OH) occurs internally in rusticles. When Fe2O3·H2O is heated, it loses its water of hydration. Further heating at 1670 kelvin converts Fe2O3 to black Fe3O4 (FeIIFeIII2O4), which is known as the mineral magnetite. Fe(O)OH is soluble in acids, giving [Fe(H2O)6]3+. In concentrated aqueous alkali, Fe2O3 gives [Fe(OH)6]3−.[12]
Reactions
The most important reaction is its
- Fe2O3 + 3 CO → 2 Fe + 3 CO2
Another redox reaction is the extremely
- 2 Al + Fe2O3 → 2 Fe + Al2O3
This process is used to weld thick metals such as rails of train tracks by using a ceramic container to funnel the molten iron in between two sections of rail. Thermite is also used in weapons and making small-scale cast-iron sculptures and tools.
Partial reduction with hydrogen at about 400 °C produces magnetite, a black magnetic material that contains both Fe(III) and Fe(II):[18]
- 3 Fe2O3 + H2 → 2 Fe3O4 + H2O
Iron(III) oxide is insoluble in water but dissolves readily in strong acid, e.g., hydrochloric and
Heating iron(III) oxides with other metal oxides or carbonates yields materials known as ferrates (ferrate (III)):[18]
- ZnO + Fe2O3 → Zn(FeO2)2
Preparation
Iron(III) oxide is a product of the oxidation of iron. It can be prepared in the laboratory by electrolyzing a solution of sodium bicarbonate, an inert electrolyte, with an iron anode:
- 4 Fe + 3 O2 + 2 H2O → 4 FeO(OH)
The resulting hydrated iron(III) oxide, written here as FeO(OH), dehydrates around 200 °C.[18][19]
- 2 FeO(OH) → Fe2O3 + H2O
Uses
Iron industry
The overwhelming application of iron(III) oxide is as the feedstock of the steel and iron industries, e.g., the production of iron, steel, and many alloys.[19]
Polishing
A very fine powder of ferric oxide is known as "jeweler's rouge", "red rouge", or simply rouge. It is used to put the final polish on metallic
Pigment
Iron(III) oxide is also used as a
Hematite is the characteristic component of the Swedish paint color Falu red.
Magnetic recording
Iron(III) oxide was the most common magnetic particle used in all types of magnetic storage and recording media, including magnetic disks (for data storage) and magnetic tape (used in audio and video recording as well as data storage). Its use in computer disks was superseded by cobalt alloy, enabling thinner magnetic films with higher storage density.[22]
Photocatalysis
α-Fe2O3 has been studied as a
Medicine
Calamine lotion, used to treat mild itchiness, is chiefly composed of a combination of zinc oxide, acting as astringent, and about 0.5% iron(III) oxide, the product's active ingredient, acting as antipruritic. The red color of iron(III) oxide is also mainly responsible for the lotion's pink color.
See also
References
- ^ a b c d Haynes, p. 4.69
- ^ a b c d Comey, Arthur Messinger; Hahn, Dorothy A. (February 1921). A Dictionary of Chemical Solubilities: Inorganic (2nd ed.). New York: The MacMillan Company. p. 433.
- ^ Haynes, p. 4.141
- ^ ISBN 978-1-118-07191-5.
- ^ a b c d Vujtek, Milan; Zboril, Radek; Kubinek, Roman; Mashlan, Miroslav. "Ultrafine Particles of Iron(III) Oxides by View of AFM – Novel Route for Study of Polymorphism in Nano-world" (PDF). Univerzity Palackého. Retrieved 12 July 2014.
- ^ a b c d e Haynes, p. 5.12
- ^ a b c Sigma-Aldrich Co., Iron(III) oxide. Retrieved on 2014-07-12.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0344". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "SDS of Iron(III) oxide" (PDF). KJLC. England: Kurt J Lesker Company Ltd. 5 January 2012. Retrieved 12 July 2014.
- ^ PubChem. "Iron oxide (Fe2O3), hydrate". pubchem.ncbi.nlm.nih.gov. Retrieved 11 November 2020.
- ISBN 978-0-471-93620-6.
- ^ ISBN 978-0-13-175553-6.
- ^ "Mechanism of Oxidation & Thermal Decomposition of Iron Sulphides" (PDF).
- S2CID 230482821. Retrieved 25 January 2021.
- PMID 24820819.
- .
- ^ Adlam; Price (1945). Higher School Certificate Inorganic Chemistry. Leslie Slater Price.
- ^ a b c Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1661.
- ^ ISBN 978-0-7506-3365-9.
- ISBN 978-1-884207-73-0.
- ISBN 978-0-19-957915-0.
- .
- ^ PMID 17147381.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - doi:10.1149/1.2131532.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 24437340.
- PMID 20201513.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 25490778.
Cited sources
- Haynes, William M., ed. (2011). ISBN 978-1439855119.



