Ferrier rearrangement
The Ferrier rearrangement is an

Mechanism
In the first step, a delocalized allyloxocarbenium ion (2) is formed, typically with the aid of a
Lewis acid like indium(III) chloride or boron trifluoride. This ion reacts in situ with an alcohol, yielding a mixture of the α (3) and β (4) anomers of the 2-glycoside, with the double bond shifted to position 3,4.[3]
Examples
| Lewis acid | Alcohol | Conditions | Results |
|---|---|---|---|
| InCl3 | methanol | in dichloromethane | α:β = 7:1[4] |
dioxane
|
water | heating | 75% yield[5] |
| SnCl4 | methanol | in dichloromethane, –78 °C, 10 min | 83% yield, α:β = 86:14[6] |
O(C2H5)2
|
isopropanol
|
in dichloromethane, RT, 24 hr | 95% yield[7][8] |
| ZnCl2 | ethanol | in toluene, RT, 30–60 min | 65–95% yield, α:β = 89:11[9][10] |
| BF3·O(C2H5)2 | benzyl alcohol | in dichloromethane, –20 °C to RT, 1 hr | 98% yield[11] |
Modifications
Forming of C-glycosides
By replacing the alcohol with a silane, C-glycosides can be formed. With triethylsilane (R'=H), the reaction yields a 2,3-unsaturated deoxy sugar.[3]

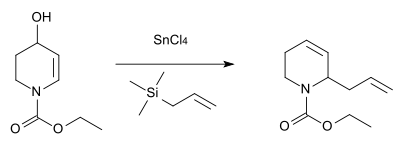
Nitrogen analogue
An analogous reaction with nitrogen as the heteroatom was described in 1984 for the synthesis of the antibiotic substance streptazolin.[12]
References
- .
- ISBN 0-471-26418-0.
- ^ .
- open accesspublication)
- .
- .
- .
- .
- doi:10.1039/b000661k.
- doi:10.1039/b000662i.
- doi:10.1039/a904991f.
- ^
Kozikowski, AP, Pyeong-uk Park (1984). "Synthesis of 2-substituted .DELTA.3-piperidines: the nitrogen analog of the Ferrier rearrangement. An approach to streptazolin". J. Org. Chem. 49 (9): 1674–1676. doi:10.1021/jo00183a044.)
{{cite journal}}: CS1 maint: multiple names: authors list (link
