Soil fertility


Soil fertility refers to the ability of
- The ability to supply essential plant nutrients and water in adequate amounts and proportions for plant growth and reproduction; and
- The absence of toxic substances which may inhibit plant growth e.g Fe2+ which leads to nutrient toxicity.
The following properties contribute to soil fertility in most situations:
- Sufficient soil depth for adequate root growth and water retention;
- Good internal drainage, allowing sufficient aeration for optimal root growth (although some plants, such as rice, tolerate waterlogging);
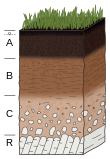
- Topsoil or horizon O is with sufficient soil organic matter for healthy soil structure and soil moisture retention;
- Soil pH in the range 5.5 to 7.0 (suitable for most plants but some prefer or tolerate more acid or alkaline conditions);
- Adequate concentrations of essential plant nutrients in plant-available forms;
- Presence of a range of microorganismsthat support plant growth.
In lands used for
Soil fertilization
Bioavailable phosphorus (available to soil life) is the element in soil that is most often lacking. Nitrogen and potassium are also needed in substantial amounts. For this reason these three elements are always identified on a commercial fertilizer analysis. For example, a 10-10-15 fertilizer has 10 percent nitrogen, 10 percent available phosphorus (P2O5) and 15 percent water-soluble potassium (K2O). Sulfur is the fourth element that may be identified in a commercial analysis—e.g. 21-0-0-24 which would contain 21% nitrogen and 24% sulfate.
Inorganic fertilizers are generally less expensive and have higher concentrations of nutrients than organic fertilizers. Also, since nitrogen, phosphorus and potassium generally must be in the inorganic forms to be taken up by plants, inorganic fertilizers are generally immediately bioavailable to plants without modification.[5] However, some have criticized the use of inorganic fertilizers, claiming that the water-soluble nitrogen does not provide for the long-term needs of the plant and creates water pollution. Slow-release fertilizers may reduce leaching loss of nutrients and may make the nutrients that they provide available over a longer period of time.
Soil fertility is a complex process that involves the constant cycling of nutrients between organic and inorganic forms. As plant material and animal wastes are decomposed by micro-organisms, they release inorganic nutrients to the soil solution, a process referred to as mineralization. Those nutrients may then undergo further transformations which may be aided or enabled by soil micro-organisms. Like plants, many micro-organisms require or preferentially use inorganic forms of nitrogen, phosphorus or potassium and will compete with plants for these nutrients, tying up the nutrients in microbial biomass, a process often called immobilization. The balance between immobilization and mineralization processes depends on the balance and availability of major nutrients and organic carbon to soil microorganisms.[6][7] Natural processes such as lightning strikes may fix atmospheric nitrogen by converting it to (NO2). Denitrification may occur under anaerobic conditions (flooding) in the presence of denitrifying bacteria. Nutrient cations, including potassium and many micronutrients, are held in relatively strong bonds with the negatively charged portions of the soil in a process known as cation exchange.
In 2008 the cost of phosphorus as fertilizer more than doubled, while the price of rock phosphate as base commodity rose eight-fold. Recently the term
A wide variety of materials have been described as soil conditioners due to their ability to improve soil quality, including biochar, offering multiple soil health benefits.[8]
Food waste compost was found to have better soil improvement than manure based compost.[9]
Light and CO2 limitations
Photosynthesis is the process whereby plants use light energy to drive chemical reactions which convert CO2 into sugars. As such, all plants require access to both light and carbon dioxide to produce energy, grow and reproduce.
While typically limited by nitrogen, phosphorus and potassium, low levels of carbon dioxide can also act as a limiting factor on plant growth. Peer-reviewed and published scientific studies have shown that increasing CO2 is highly effective at promoting plant growth up to levels over 300 ppm. Further increases in CO2 can, to a very small degree, continue to increase net photosynthetic output.[10]
Soil depletion
Soil depletion occurs when the components which contribute to fertility are removed and not replaced, and the conditions which support soil's fertility are not maintained. This leads to poor crop yields. In agriculture, depletion can be due to excessively intense cultivation and inadequate soil management.
Soil fertility can be severely challenged when
One of the most widespread occurrences of soil depletion as of 2008[update] is in tropical zones where nutrient content of soils is low. The combined effects of growing population densities, large-scale industrial logging, slash-and-burn agriculture and ranching, and other factors, have in some places depleted soils through rapid and almost total nutrient removal.
The depletion of soil has affected the state of plant life and crops in agriculture in many countries. In the Middle East for example, many countries find it difficult to grow produce because of droughts, lack of soil, and lack of irrigation. The
Topsoil depletion occurs when the nutrient-rich organic topsoil, which takes hundreds to thousands of years to build up under natural conditions, is eroded or depleted of its original organic material.[14] Historically, many past civilizations' collapses can be attributed to the depletion of the topsoil. Since the beginning of agricultural production in the Great Plains of North America in the 1880s, about one-half of its topsoil has disappeared.[15]
Depletion may occur through a variety of other effects, including overtillage (which damages soil structure), underuse of nutrient inputs which leads to mining of the soil nutrient bank, and salinization of soil.
Irrigation effects
The quality of irrigation water is very important to maintain soil fertility and
Top soil loss takes place in alkali soils due to erosion by rain water surface flows or drainage as they form colloids (fine mud) in contact with water. Plants absorb water-soluble inorganic salts only from the soil for their growth. Soil as such does not lose fertility just by growing crops but it lose its fertility due to accumulation of unwanted and depletion of wanted inorganic salts from the soil by improper irrigation and acid rain water (quantity and quality of water). The fertility of many soils which are not suitable for plant growth can be enhanced many times gradually by providing adequate irrigation water of suitable quality and good drainage from the soil.
Global distribution
See also
- Arable land
- Plaggen soil
- Shifting cultivation
- Soil contamination
- Soil life
- Terra preta
- Cation-exchange capacity
References
- ^ Kelly, Karina (13 September 1995). "A Chat with Tim Flannery on Population Control". Australian Broadcasting Corporation. Archived from the original on 13 January 2010. Retrieved 23 April 2010. "Well, Australia has by far the world's least fertile soils".
- ^ Grant, Cameron (August 2007). "Damaged Dirt" (PDF). The Advertiser. Archived from the original (PDF) on 6 July 2011. Retrieved 23 April 2010.
Australia has the oldest, most highly weathered soils on the planet.
- ^ Bodenfruchtbarkeit, Retrieved on 2015-11-09.
- ^ "Soil Fertility". www.fao.org. Archived from the original on 24 November 2017. Retrieved 18 June 2016.
- ^ Brady N., Weil R. 2002 Nitrogen and sulfur economy of soils. pp. 543–571 in Helba (ed.), The Nature and properties of soils. Pearson Education, NJ.
- ^ Sims, G. K., and M. M. Wander. 2002. Proteolytic activity under nitrogen or sulfur limitation. Appl. Soil Ecol. 568:1–5.
- ^ Sims, G.K. 2006. Nitrogen Starvation Promotes Biodegradation of N-Heterocyclic Compounds in Soil. Soil Biology & Biochemistry 38:2478–2480.
- S2CID 237725246.
- ISSN 2311-7524.
- ISBN 0387954392.
- ^ a b Cronon, William, Changes in the Land: Indians, Colonists, and the Ecology of New England, NY: Hill & Wang, 1983, pp. 145–152.
- ^ Scherr, Sara (1996). "Land degradation in the developing world: Implications for food, agriculture, and the environment to 2020" (PDF): 7–8.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Smaling, Eric (1997). "Soil Fertility in Africa is at Stake". Replenishing Soil Fertility in Africa Replenishingsoi: 49.
- ^ Bjonnes, R., 1997, Food vs Feed, People's News Agency; Frederiksberg C, Denmark
- ISBN 0963378457.
- ^ Managing Soil Tilth; Colorado state university garden notes, Retrieved on 2014-10-04.
- ^ Managing irrigation water quality, Oregon State University, US, Retrieved on 2012-10-04.